Melperone
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Buronil |
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | Oral, intramuscular injection |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 87% (IM), 54% (Oral via syrup), 65% (Oral, tablet)[1] |
| Protein binding | 50% |
| Metabolism | Hepatic |
| Elimination half-life | 3–4 hours (oral)[1] 6 hours (IM) |
| Excretion | Renal (70% as metabolites, 5.5–10.4% as unchanged drug)[1][2] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.107.027 |
| Chemical and physical data | |
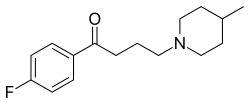
| Formula | C16H22FNO |
| Molar mass | 263.356 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Melperone (Bunil (PT), Buronil (AT, BE, CZ, DK, FI†, NL†, NO†, SE), Eunerpan (DE))[3] is an atypical antipsychotic of the butyrophenone chemical class, making it structurally related to the typical antipsychotic haloperidol. It first entered clinical use in 1960s.[4]
Marketing and indications
[edit]It has been tried in treatment-resistant cases of schizophrenia with some (albeit limited) success.[4][5][6][7] It has also been reported effective in the treatment of L-DOPA and other forms of psychosis in Parkinson's disease[8] (although a multicentre, double-blind, placebo-controlled study conducted in 2012 failed to support these findings[9]). It is also known to possess anxiolytic properties.[10] It is marketed in the following countries:[3][11]
Adverse effects
[edit]Melperone is reported to produce significantly less weight gain than clozapine and approximately as much weight gain as typical antipsychotics.[12] It is also purported to produce around as much prolactin secretion as clozapine (which is virtually nil).[13] It is also purported to produce sedative effects[14] and QT interval prolongation.[15] It is also known to produce less extrapyramidal side effects than the first-generation (typical) antipsychotic, thiothixene.[16] It can also produce (usually relatively mild) dry mouth.[17]
- Constipation
- Diarrhea
- Nausea
- Vomiting
- Appetite loss
- Hypersalivation (drooling)
- Extrapyramidal side effects (e.g. tremor, dystonia, hypokinesis, akathisia, dyskinesias)
- Insomnia
- Agitation
- Headache
- Dizziness
- Fatigue
- Miosis
- Mydriasis
- Blurred vision
- Elevated liver enzymes (esp. ALT and GGTP)
- Tardive dyskinesia
- Neuroleptic malignant syndrome
- Blood dyscrasias (pancytopenia, agranulocytosis, leukopenia, thrombocytopenia, etc.)
- Seizures (probably rare/uncommon)
- Increased intraocular pressure
- Intrahepatic cholestasis (probably rare)
- Orthostatic hypotension (probably common)
- Arrhythmias
- Rash
- Hyperprolactinemia (which can lead to e.g. galactorrhea, gynecomastia)
- Weight gain
- Increased appetite
Interactions
[edit]Melperone is reported to be a CYP2D6 inhibitor.[20][21][22]
Pharmacology
[edit]Melperone binds to the dopamine D2 receptor, just like all other clinically utilized antipsychotics, but it does so with a very low affinity and hence may be liable to rapidly dissociate from the D2 receptor hence potentially giving it the profile of an atypical antipsychotic.[23]
| Receptor | Ki [nM][24] |
|---|---|
| 5-HT1A | 2,200 |
| 5-HT1D | 3,400 |
| 5-HT2A | 230 |
| 5-HT2C | 2,100 |
| 5-HT6 | 1,254 |
| 5-HT7 | 578 |
| α1 | 180 |
| α2 | 150 |
| M1 | >10,000 |
| M2 | 2,400 |
| M3 | >10,000 |
| M4 | 4,400 |
| M5 | >10,000 |
| D2 | 194 |
| D3 | 347 |
| D4 | 555 |
| H1 | 580 |
Synthesis
[edit]
For the last step of the synthesis the sidechain 4-Chloro-4'-Fluorobutyrophenone [3874-54-2] (1) is attached to 4-Methylpiperidine (4-Pipecoline) [626-58-4] (2).
See also
[edit]References
[edit]- ^ a b c Borgström L, Larsson H, Molander L (1982). "Pharmacokinetics of parenteral and oral melperone in man". European Journal of Clinical Pharmacology. 23 (2): 173–6. doi:10.1007/BF00545974. PMID 7140807. S2CID 36697288.
- ^ a b c d Product Information: Eunerpan(R), Melperonhydrochlorid (Report). Knoll Deutschland GmbH, Ludwigshafen. 1995.
- ^ a b Melperone Hydrochloride. The Royal Pharmaceutical Society of Great Britain. 30 January 2013. Archived from the original on 14 January 2021. Retrieved 3 November 2013.
{{cite book}}:|work=ignored (help) - ^ a b Röhricht F, Gadhia S, Alam R, Willis M (2012). "Auditing clinical outcomes after introducing off-licence prescribing of atypical antipsychotic melperone for patients with treatment refractory schizophrenia". TheScientificWorldJournal. 2012: 512047. doi:10.1100/2012/512047. PMC 3330679. PMID 22566771.
- ^ Whiskey E, Vavrova M, Gaughran F, Taylor D (February 2011). "Melperone in treatment-refractory schizophrenia: a case series". Therapeutic Advances in Psychopharmacology. 1 (1): 19–23. doi:10.1177/2045125311399800. PMC 3736899. PMID 23983923.
- ^ Meltzer HY, Sumiyoshi T, Jayathilake K (December 2001). "Melperone in the treatment of neuroleptic-resistant schizophrenia". Psychiatry Research. 105 (3): 201–9. doi:10.1016/s0165-1781(01)00346-8. PMID 11814539. S2CID 23311317.
- ^ Sumiyoshi T, Meltzer HY, Jayathilake K (2004). "Melperone, an atypical antipsychotic drug, in the treatment of schizophrenia: dose-response analysis on effectiveness and tolerability, and efficacy for treatment-resistant schizophrenia and cognitive function". International Clinical Psychopharmacology. 19 (3): 184. doi:10.1097/00004850-200405000-00039.
- ^ Barbato L, Monge A, Stocchi F, Nordera G. Melperone in the treatment of iatrogenic psychosis in Parkinson’s disease. Funct Neurol. 1996 Aug;11(4):201–7.
- ^ Friedman JH (May 2012). "Melperone is ineffective in treating Parkinson's disease psychosis". Movement Disorders. 27 (6): 803–4. doi:10.1002/mds.24942. PMID 22362330. S2CID 41211677.
- ^ Pöldinger WJ (1984). "Melperone in low doses in anxious neurotic patients. A double-blind placebo-controlled clinical study". Neuropsychobiology. 11 (3): 181–6. doi:10.1159/000118074. PMID 6147789.
- ^ "Buronil generic. Price of buronil. Uses, Dosage, Side effects".
- ^ Bobo WV, Jayathilake K, Lee MA, Meltzer HY (April 2010). "Changes in weight and body mass index during treatment with melperone, clozapine and typical neuroleptics". Psychiatry Research. 176 (2–3): 114–9. doi:10.1016/j.psychres.2009.03.026. PMID 20199813. S2CID 25366120.
- ^ Bobo WV, Jayathilake K, Lee MA, Meltzer HY (July 2009). "Melperone, an aytpical antipsychotic drug with clozapine-like effect on plasma prolactin: contrast with typical neuroleptics". Human Psychopharmacology. 24 (5): 415–22. doi:10.1002/hup.1036. PMID 19551763. S2CID 25812368.
- ^ Molander L, Borgström L (1983). "Sedative effects and prolactin response to single oral doses of melperone". Psychopharmacology. 79 (2–3): 142–7. doi:10.1007/bf00427801. PMID 6133301. S2CID 392818.
- ^ Hui WK, Mitchell LB, Kavanagh KM, Gillis AM, Wyse DG, Manyari DE, Duff HJ (January 1990). "Melperone: electrophysiologic and antiarrhythmic activity in humans". Journal of Cardiovascular Pharmacology. 15 (1): 144–9. doi:10.1097/00005344-199001000-00023. PMID 1688972. S2CID 40589560.
- ^ Bjerkenstedt L (1989). "Melperone in the treatment of schizophrenia". Acta Psychiatrica Scandinavica. Supplementum. 352: 35–9. doi:10.1111/j.1600-0447.1989.tb06434.x. PMID 2479227. S2CID 7828966.
- ^ Molander L, Birkhed D (1981). "Effect of single oral doses of various neuroleptic drugs on salivary secretion rate, pH, and buffer capacity in healthy subjects". Psychopharmacology. 75 (2): 114–8. doi:10.1007/bf00432171. PMID 6119724. S2CID 780924.
- ^ a b c Kirkegaard A, Kirkegaard G, Geismar L, Christensen I (1981). "Additional studies on side effects of melperone in long-term therapy for 1 to 15 years in psychiatric patients". Arzneimittel-Forschung. 31 (4): 737–40. PMID 6113835.
- ^ a b c Christensen I, Geismar L, Kirkegaard A, Kirkegaard G (May 1986). "Additional studies on side effects of melperone in long-term therapy for 1-20 years in psychiatric patients". Arzneimittel-Forschung. 36 (5): 855–60. PMID 2873821.
- ^ Gahr M, Gastl R, Kölle MA, Schönfeldt-Lecuona C, Freudenmann RW (February 2012). "Successful treatment of schizophrenia with melperone augmentation in a patient with phenotypic CYP2D6 ultrarapid metabolization: a case report". Journal of Medical Case Reports. 6 (1): 49. doi:10.1186/1752-1947-6-49. PMC 3298719. PMID 22309430.
- ^ Köhnke MD, Lutz U, Wiatr G, Schwärzler F, Weller B, Schott K, Buchkremer G (April 2006). "Cytochrome P450 2D6 dependent metabolization of risperidone is inhibited by melperone". European Journal of Clinical Pharmacology. 62 (4): 333–4. doi:10.1007/s00228-006-0098-y. PMID 16534635. S2CID 13168439.
- ^ Grözinger M, Dragicevic A, Hiemke C, Shams M, Müller MJ, Härtter S (January 2003). "Melperone is an inhibitor of the CYP2D6 catalyzed O-demethylation of venlafaxine". Pharmacopsychiatry. 36 (1): 3–6. doi:10.1055/s-2003-38084. PMID 12649767. S2CID 260242201.
- ^ Seeman P (January 2004). "Atypical Antipsychotics: Mechanism of Action" (PDF). FOCUS: The Journal of Lifelong Learning in Psychiatry. 2 (1): 48–58. doi:10.1176/foc.2.1.48. PMID 11873706. Archived from the original (PDF) on 2013-11-27.
- ^ Roth BL, Driscol J. "PDSP Ki Database". Psychoactive Drug Screening Program (PDSP). University of North Carolina at Chapel Hill and the United States National Institute of Mental Health. Archived from the original on 2013-11-08. Retrieved 2013-10-14.
- ^ J Lassen, S Hernestam, N Sterner, U.S. patent 3,816,433 (1974 to Ferrosan Ab).
- ^ BE651144 idem Erik Harry Hernestam Sven, et al.GB 1029220 (1966 to Ferrosan); CA, 63, 13244c
- ^ Leyva-Pérez, Antonio; Cabrero-Antonino, Jose R.; Rubio-Marqués, Paula; Al-Resayes, Saud I.; Corma, Avelino (2014). "Synthesis of the ortho/meta/para Isomers of Relevant Pharmaceutical Compounds by Coupling a Sonogashira Reaction with a Regioselective Hydration". ACS Catalysis. 4 (3): 722–731. doi:10.1021/cs401075z.
External links
[edit]- PubChem Substance
- Clinical trial number NCT00125138 for "Melperone (an Anti-Psychotic) in Patients With Psychosis Associated With Parkinson's Disease" at ClinicalTrials.gov
