Estrogen receptor
| estrogen receptor 1 (ER-alpha) | |||||||
|---|---|---|---|---|---|---|---|
 | |||||||
| Identifiers | |||||||
| Symbol | ESR1 | ||||||
| Alt. symbols | ER-α, NR3A1 | ||||||
| NCBI gene | 2099 | ||||||
| HGNC | 3467 | ||||||
| OMIM | 133430 | ||||||
| PDB | 1ERE | ||||||
| RefSeq | NM_000125 | ||||||
| UniProt | P03372 | ||||||
| Other data | |||||||
| Locus | Chr. 6 q24-q27 | ||||||
| |||||||
| estrogen receptor 2 (ER-beta) | |||||||
|---|---|---|---|---|---|---|---|
 | |||||||
| Identifiers | |||||||
| Symbol | ESR2 | ||||||
| Alt. symbols | ER-β, NR3A2 | ||||||
| NCBI gene | 2100 | ||||||
| HGNC | 3468 | ||||||
| OMIM | 601663 | ||||||
| PDB | 1QKM | ||||||
| RefSeq | NM_001040275 | ||||||
| UniProt | Q92731 | ||||||
| Other data | |||||||
| Locus | Chr. 14 q21-q22 | ||||||
| |||||||
Estrogen receptors (ERs) are proteins found in cells that function as receptors for the hormone estrogen (17β-estradiol).[1] There are two main classes of ERs. The first includes the intracellular estrogen receptors, namely ERα and ERβ, which belong to the nuclear receptor family. The second class consists of membrane estrogen receptors (mERs), such as GPER (GPR30), ER-X, and Gq-mER, which are primarily G protein-coupled receptors. This article focuses on the nuclear estrogen receptors (ERα and ERβ).
Upon activation by estrogen, intracellular ERs undergo translocation to the nucleus where they bind to specific DNA sequences. As DNA-binding transcription factors, they regulate the activity of various genes. However, ERs also exhibit functions that are independent of their DNA-binding capacity.[2] These non-genomic actions contribute to the diverse effects of estrogen signaling in cells.
Estrogen receptors (ERs) belong to the family of steroid hormone receptors, which are hormone receptors for sex steroids. Along with androgen receptors (ARs) and progesterone receptors (PRs), ERs play crucial roles in regulating sexual maturation and gestation. These receptors mediate the effects of their respective hormones, contributing to the development and maintenance of reproductive functions and secondary sexual characteristics.
Genes
[edit]In humans, the two forms of the estrogen receptor are encoded by different genes, ESR1 and ESR2 on the sixth and fourteenth chromosome (6q25.1 and 14q23.2), respectively.
Structure
[edit]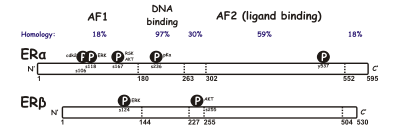
| Estrogen receptor alpha N-terminal AF1 domain | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| Symbol | Oest_recep | ||||||||
| Pfam | PF02159 | ||||||||
| InterPro | IPR001292 | ||||||||
| SCOP2 | 1hcp / SCOPe / SUPFAM | ||||||||
| |||||||||
| Estrogen and estrogen related receptor C-terminal domain | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| Symbol | ESR1_C | ||||||||
| Pfam | PF12743 | ||||||||
| |||||||||
There are two different forms of the estrogen receptor, usually referred to as α and β, each encoded by a separate gene (ESR1 and ESR2, respectively). Hormone-activated estrogen receptors form dimers, and, since the two forms are coexpressed in many cell types, the receptors may form ERα (αα) or ERβ (ββ) homodimers or ERαβ (αβ) heterodimers.[3] Estrogen receptor alpha and beta show significant overall sequence homology, and both are composed of five domains designated A/B through F (listed from the N- to C-terminus; amino acid sequence numbers refer to human ER).[citation needed]
The N-terminal A/B domain is able to transactivate gene transcription in the absence of bound ligand (e.g., the estrogen hormone). While this region is able to activate gene transcription without ligand, this activation is weak and more selective compared to the activation provided by the E domain. The C domain, also known as the DNA-binding domain, binds to estrogen response elements in DNA. The D domain is a hinge region that connects the C and E domains. The E domain contains the ligand binding cavity as well as binding sites for coactivator and corepressor proteins. The E-domain in the presence of bound ligand is able to activate gene transcription. The C-terminal F domain function is not entirely clear and is variable in length.[citation needed]
Due to alternative RNA splicing, several ER isoforms are known to exist. At least three ERα and five ERβ isoforms have been identified. The ERβ isoforms receptor subtypes can transactivate transcription only when a heterodimer with the functional ERß1 receptor of 59 kDa is formed. The ERß3 receptor was detected at high levels in the testis. The two other ERα isoforms are 36 and 46kDa.[4][5]
Only in fish, but not in humans, an ERγ receptor has been described.[6]
Tissue distribution
[edit]Both ERs are widely expressed in different tissue types, however there are some notable differences in their expression patterns:[7]
- The ERα is found in endometrium, breast cancer cells, ovarian stromal cells, and the hypothalamus.[8] In males, ERα protein is found in the epithelium of the efferent ducts.[9]
- The expression of the ERβ protein has been documented in ovarian granulosa cells, kidney, brain, bone, heart,[10] lungs, intestinal mucosa, prostate, and endothelial cells.
The ERs are regarded to be cytoplasmic receptors in their unliganded state, but visualization research has shown that only a small fraction of the ERs reside in the cytoplasm, with most ER constitutively in the nucleus.[11] The "ERα" primary transcript gives rise to several alternatively spliced variants of unknown function.[12]
Signal transduction
[edit]Since estrogen is a steroidal hormone, it can readily diffuse through the phospholipid membranes of cells due to its lipophilic nature. As a result, estrogen receptors can be located intracellularly and do not necessarily need to be membrane-bound to interact with estrogen.[13] However, both intracellular and membrane-bound estrogen receptors exist, each mediating different cellular responses to estrogen.[14]
Genomic
[edit]In the absence of hormone, estrogen receptors are predominantly located in the cytoplasm.[15] Hormone binding triggers a series of events, beginning with the migration of the receptor from the cytoplasm to the nucleus. This is followed by the dimerization of the receptor, where two receptor molecules join together. Finally, the receptor dimer binds to specific DNA sequences known as hormone response elements, initiating the process of gene regulation.
The DNA/receptor complex then recruits other proteins responsible for transcription of downstream DNA into mRNA and ultimately protein, resulting in changes in cell function.[15] Estrogen receptors are also present within the cell nucleus, and both estrogen receptor subtypes (ERα and ERβ) contain a DNA-binding domain, allowing them to function as transcription factors regulating protein production.[16]
The receptor also interacts with transcription factors such as activator protein 1 and Sp-1 to promote transcription, via several coactivators including PELP-1.[15] Tumor suppressor kinase LKB1 coactivates ERα in the cell nucleus through direct binding, recruiting it to the promoter of ERα-responsive genes. LKB1's catalytic activity enhances ERα transactivation compared to catalytically deficient LKB1 mutants.[17] Direct acetylation of estrogen receptor alpha at lysine residues in the hinge region by p300 regulates transactivation and hormone sensitivity.[18]
Non-genomic
[edit]Nuclear estrogen receptors can also associate with the cell surface membrane and undergo rapid activation upon cellular exposure to estrogen.[19][20]
Some ERs interact with cell membranes by binding to caveolin-1 and forming complexes with G proteins, striatin, receptor tyrosine kinases (e.g., EGFR and IGF-1), and non-receptor tyrosine kinases (e.g., Src).[2][19] Membrane-bound ERs associated with striatin can increase levels of Ca2+ and nitric oxide (NO).[21] Interactions with receptor tyrosine kinases trigger signaling to the nucleus via the mitogen-activated protein kinase (MAPK/ERK) and phosphoinositide 3-kinase (Pl3K/AKT) pathways.[22]
Glycogen synthase kinase-3 (GSK)-3β inhibits nuclear ER transcription by preventing phosphorylation of serine 118 on nuclear ERα. The PI3K/AKT and MAPK/ERK pathways can phosphorylate GSK-3β, thereby removing its inhibitory effect, with the latter pathway acting via rsk.
17β-Estradiol has been shown to activate the G protein-coupled receptor GPR30.[23] However, the subcellular localization and precise role of this receptor remain controversial.[24]
Clinical significance
[edit]

Cancer
[edit]Estrogen receptors are over-expressed in around 70% of breast cancer cases, referred to as "ER-positive", and can be demonstrated in such tissues using immunohistochemistry. Two hypotheses have been proposed to explain why this causes tumorigenesis, and the available evidence suggests that both mechanisms contribute:
- First, binding of estrogen to the ER stimulates proliferation of mammary cells, with the resulting increase in cell division and DNA replication, leading to mutations.
- Second, estrogen metabolism produces genotoxic waste.
The result of both processes is disruption of cell cycle, apoptosis and DNA repair, which increases the chance of tumour formation. ERα is certainly associated with more differentiated tumours, while evidence that ERβ is involved is controversial. Different versions of the ESR1 gene have been identified (with single-nucleotide polymorphisms) and are associated with different risks of developing breast cancer.[25]
Estrogen and the ERs have also been implicated in breast cancer, ovarian cancer, colon cancer, prostate cancer, and endometrial cancer. Advanced colon cancer is associated with a loss of ERβ, the predominant ER in colon tissue, and colon cancer is treated with ERβ-specific agonists.[26]
Endocrine therapy for breast cancer involves selective estrogen receptor modulators (SERMS), such as tamoxifen, which behave as ER antagonists in breast tissue, or aromatase inhibitors, such as anastrozole. ER status is used to determine sensitivity of breast cancer lesions to tamoxifen and aromatase inhibitors.[27] Another SERM, raloxifene, has been used as a preventive chemotherapy for women judged to have a high risk of developing breast cancer.[28] Another chemotherapeutic anti-estrogen, ICI 182,780 (Faslodex), which acts as a complete antagonist, also promotes degradation of the estrogen receptor.
However, de novo resistance to endocrine therapy undermines the efficacy of using competitive inhibitors like tamoxifen. Hormone deprivation through the use of aromatase inhibitors is also rendered futile.[29] Massively parallel genome sequencing has revealed the common presence of point mutations on ESR1 that are drivers for resistance, and promote the agonist conformation of ERα without the bound ligand. Such constitutive, estrogen-independent activity is driven by specific mutations, such as the D538G or Y537S/C/N mutations, in the ligand binding domain of ESR1 and promote cell proliferation and tumor progression without hormone stimulation.[30]
Menopause
[edit]The metabolic effects of estrogen in postmenopausal women has been linked to the genetic polymorphism of estrogen receptor beta (ER-β).[31]
Aging
[edit]Studies in female mice have shown that estrogen receptor-alpha declines in the pre-optic hypothalamus as they grow old. Female mice that were given a calorically restricted diet during the majority of their lives maintained higher levels of ERα in the pre-optic hypothalamus than their non-calorically restricted counterparts.[8]
Obesity
[edit]A dramatic demonstration of the importance of estrogens in the regulation of fat deposition comes from transgenic mice that were genetically engineered to lack a functional aromatase gene. These mice have very low levels of estrogen and are obese.[32] Obesity was also observed in estrogen deficient female mice lacking the follicle-stimulating hormone receptor.[33] The effect of low estrogen on increased obesity has been linked to estrogen receptor alpha.[34]
SERMs for other treatment purposes
[edit]SERMs are also being studied for the treatment of uterine fibroids[35] and endometriosis.[36] The evidence supporting the use of SERMs for treating uterine fibroids (reduction in size of fibroids and improving other clinical outcomes) is inconclusive and more research is needed.[35] It is also not clear if SERMs is effective for treating endometriosis.[36]
Estrogen insensitivity syndrome
[edit]Estrogen insensitivity syndrome is a rare intersex condition with 5 reported cases, in which estrogen receptors do not function. The phenotype results in extensive masculinization. Unlike androgen insensitivity syndrome, EIS does not result in phenotype sex reversal. It is incredibly rare and is anologious to the AIS, and forms of adrenal hyperplasia. The reason why AIS is common and EIS is exceptionally rare is that XX AIS does not result in infertility, and therefore can be maternally inheirented, while EIS always results in infertility regardless of karyotype. A negative feedback loop between the endocrine system also occurs in EIS, in which the gonads produce markedly higher levels of estrogen for individuals with EIS (119–272 pg/mL XY and 750–3,500 pg/mL XX, see average levels) however no feminizing effects occur.[37][38]
Ligands
[edit]Agonists
[edit]- Endogenous estrogens (e.g., estradiol, estrone, estriol, estetrol)
- Natural estrogens (e.g., conjugated estrogens)
- Synthetic estrogens (e.g., ethinylestradiol, diethylstilbestrol)
Mixed (agonist and antagonist mode of action)
[edit]- Phytoestrogens (e.g., coumestrol, daidzein, genistein, miroestrol)
- Selective estrogen receptor modulators (e.g., tamoxifen, clomifene, raloxifene)
Antagonists
[edit]- Antiestrogens (e.g., fulvestrant, ICI-164384, ethamoxytriphetol)
Affinities
[edit]| Ligand | Other names | Relative binding affinities (RBA, %)a | Absolute binding affinities (Ki, nM)a | Action | ||
|---|---|---|---|---|---|---|
| ERα | ERβ | ERα | ERβ | |||
| Estradiol | E2; 17β-Estradiol | 100 | 100 | 0.115 (0.04–0.24) | 0.15 (0.10–2.08) | Estrogen |
| Estrone | E1; 17-Ketoestradiol | 16.39 (0.7–60) | 6.5 (1.36–52) | 0.445 (0.3–1.01) | 1.75 (0.35–9.24) | Estrogen |
| Estriol | E3; 16α-OH-17β-E2 | 12.65 (4.03–56) | 26 (14.0–44.6) | 0.45 (0.35–1.4) | 0.7 (0.63–0.7) | Estrogen |
| Estetrol | E4; 15α,16α-Di-OH-17β-E2 | 4.0 | 3.0 | 4.9 | 19 | Estrogen |
| Alfatradiol | 17α-Estradiol | 20.5 (7–80.1) | 8.195 (2–42) | 0.2–0.52 | 0.43–1.2 | Metabolite |
| 16-Epiestriol | 16β-Hydroxy-17β-estradiol | 7.795 (4.94–63) | 50 | ? | ? | Metabolite |
| 17-Epiestriol | 16α-Hydroxy-17α-estradiol | 55.45 (29–103) | 79–80 | ? | ? | Metabolite |
| 16,17-Epiestriol | 16β-Hydroxy-17α-estradiol | 1.0 | 13 | ? | ? | Metabolite |
| 2-Hydroxyestradiol | 2-OH-E2 | 22 (7–81) | 11–35 | 2.5 | 1.3 | Metabolite |
| 2-Methoxyestradiol | 2-MeO-E2 | 0.0027–2.0 | 1.0 | ? | ? | Metabolite |
| 4-Hydroxyestradiol | 4-OH-E2 | 13 (8–70) | 7–56 | 1.0 | 1.9 | Metabolite |
| 4-Methoxyestradiol | 4-MeO-E2 | 2.0 | 1.0 | ? | ? | Metabolite |
| 2-Hydroxyestrone | 2-OH-E1 | 2.0–4.0 | 0.2–0.4 | ? | ? | Metabolite |
| 2-Methoxyestrone | 2-MeO-E1 | <0.001–<1 | <1 | ? | ? | Metabolite |
| 4-Hydroxyestrone | 4-OH-E1 | 1.0–2.0 | 1.0 | ? | ? | Metabolite |
| 4-Methoxyestrone | 4-MeO-E1 | <1 | <1 | ? | ? | Metabolite |
| 16α-Hydroxyestrone | 16α-OH-E1; 17-Ketoestriol | 2.0–6.5 | 35 | ? | ? | Metabolite |
| 2-Hydroxyestriol | 2-OH-E3 | 2.0 | 1.0 | ? | ? | Metabolite |
| 4-Methoxyestriol | 4-MeO-E3 | 1.0 | 1.0 | ? | ? | Metabolite |
| Estradiol sulfate | E2S; Estradiol 3-sulfate | <1 | <1 | ? | ? | Metabolite |
| Estradiol disulfate | Estradiol 3,17β-disulfate | 0.0004 | ? | ? | ? | Metabolite |
| Estradiol 3-glucuronide | E2-3G | 0.0079 | ? | ? | ? | Metabolite |
| Estradiol 17β-glucuronide | E2-17G | 0.0015 | ? | ? | ? | Metabolite |
| Estradiol 3-gluc. 17β-sulfate | E2-3G-17S | 0.0001 | ? | ? | ? | Metabolite |
| Estrone sulfate | E1S; Estrone 3-sulfate | <1 | <1 | >10 | >10 | Metabolite |
| Estradiol benzoate | EB; Estradiol 3-benzoate | 10 | ? | ? | ? | Estrogen |
| Estradiol 17β-benzoate | E2-17B | 11.3 | 32.6 | ? | ? | Estrogen |
| Estrone methyl ether | Estrone 3-methyl ether | 0.145 | ? | ? | ? | Estrogen |
| ent-Estradiol | 1-Estradiol | 1.31–12.34 | 9.44–80.07 | ? | ? | Estrogen |
| Equilin | 7-Dehydroestrone | 13 (4.0–28.9) | 13.0–49 | 0.79 | 0.36 | Estrogen |
| Equilenin | 6,8-Didehydroestrone | 2.0–15 | 7.0–20 | 0.64 | 0.62 | Estrogen |
| 17β-Dihydroequilin | 7-Dehydro-17β-estradiol | 7.9–113 | 7.9–108 | 0.09 | 0.17 | Estrogen |
| 17α-Dihydroequilin | 7-Dehydro-17α-estradiol | 18.6 (18–41) | 14–32 | 0.24 | 0.57 | Estrogen |
| 17β-Dihydroequilenin | 6,8-Didehydro-17β-estradiol | 35–68 | 90–100 | 0.15 | 0.20 | Estrogen |
| 17α-Dihydroequilenin | 6,8-Didehydro-17α-estradiol | 20 | 49 | 0.50 | 0.37 | Estrogen |
| Δ8-Estradiol | 8,9-Dehydro-17β-estradiol | 68 | 72 | 0.15 | 0.25 | Estrogen |
| Δ8-Estrone | 8,9-Dehydroestrone | 19 | 32 | 0.52 | 0.57 | Estrogen |
| Ethinylestradiol | EE; 17α-Ethynyl-17β-E2 | 120.9 (68.8–480) | 44.4 (2.0–144) | 0.02–0.05 | 0.29–0.81 | Estrogen |
| Mestranol | EE 3-methyl ether | ? | 2.5 | ? | ? | Estrogen |
| Moxestrol | RU-2858; 11β-Methoxy-EE | 35–43 | 5–20 | 0.5 | 2.6 | Estrogen |
| Methylestradiol | 17α-Methyl-17β-estradiol | 70 | 44 | ? | ? | Estrogen |
| Diethylstilbestrol | DES; Stilbestrol | 129.5 (89.1–468) | 219.63 (61.2–295) | 0.04 | 0.05 | Estrogen |
| Hexestrol | Dihydrodiethylstilbestrol | 153.6 (31–302) | 60–234 | 0.06 | 0.06 | Estrogen |
| Dienestrol | Dehydrostilbestrol | 37 (20.4–223) | 56–404 | 0.05 | 0.03 | Estrogen |
| Benzestrol (B2) | – | 114 | ? | ? | ? | Estrogen |
| Chlorotrianisene | TACE | 1.74 | ? | 15.30 | ? | Estrogen |
| Triphenylethylene | TPE | 0.074 | ? | ? | ? | Estrogen |
| Triphenylbromoethylene | TPBE | 2.69 | ? | ? | ? | Estrogen |
| Tamoxifen | ICI-46,474 | 3 (0.1–47) | 3.33 (0.28–6) | 3.4–9.69 | 2.5 | SERM |
| Afimoxifene | 4-Hydroxytamoxifen; 4-OHT | 100.1 (1.7–257) | 10 (0.98–339) | 2.3 (0.1–3.61) | 0.04–4.8 | SERM |
| Toremifene | 4-Chlorotamoxifen; 4-CT | ? | ? | 7.14–20.3 | 15.4 | SERM |
| Clomifene | MRL-41 | 25 (19.2–37.2) | 12 | 0.9 | 1.2 | SERM |
| Cyclofenil | F-6066; Sexovid | 151–152 | 243 | ? | ? | SERM |
| Nafoxidine | U-11,000A | 30.9–44 | 16 | 0.3 | 0.8 | SERM |
| Raloxifene | – | 41.2 (7.8–69) | 5.34 (0.54–16) | 0.188–0.52 | 20.2 | SERM |
| Arzoxifene | LY-353,381 | ? | ? | 0.179 | ? | SERM |
| Lasofoxifene | CP-336,156 | 10.2–166 | 19.0 | 0.229 | ? | SERM |
| Ormeloxifene | Centchroman | ? | ? | 0.313 | ? | SERM |
| Levormeloxifene | 6720-CDRI; NNC-460,020 | 1.55 | 1.88 | ? | ? | SERM |
| Ospemifene | Deaminohydroxytoremifene | 0.82–2.63 | 0.59–1.22 | ? | ? | SERM |
| Bazedoxifene | – | ? | ? | 0.053 | ? | SERM |
| Etacstil | GW-5638 | 4.30 | 11.5 | ? | ? | SERM |
| ICI-164,384 | – | 63.5 (3.70–97.7) | 166 | 0.2 | 0.08 | Antiestrogen |
| Fulvestrant | ICI-182,780 | 43.5 (9.4–325) | 21.65 (2.05–40.5) | 0.42 | 1.3 | Antiestrogen |
| Propylpyrazoletriol | PPT | 49 (10.0–89.1) | 0.12 | 0.40 | 92.8 | ERα agonist |
| 16α-LE2 | 16α-Lactone-17β-estradiol | 14.6–57 | 0.089 | 0.27 | 131 | ERα agonist |
| 16α-Iodo-E2 | 16α-Iodo-17β-estradiol | 30.2 | 2.30 | ? | ? | ERα agonist |
| Methylpiperidinopyrazole | MPP | 11 | 0.05 | ? | ? | ERα antagonist |
| Diarylpropionitrile | DPN | 0.12–0.25 | 6.6–18 | 32.4 | 1.7 | ERβ agonist |
| 8β-VE2 | 8β-Vinyl-17β-estradiol | 0.35 | 22.0–83 | 12.9 | 0.50 | ERβ agonist |
| Prinaberel | ERB-041; WAY-202,041 | 0.27 | 67–72 | ? | ? | ERβ agonist |
| ERB-196 | WAY-202,196 | ? | 180 | ? | ? | ERβ agonist |
| Erteberel | SERBA-1; LY-500,307 | ? | ? | 2.68 | 0.19 | ERβ agonist |
| SERBA-2 | – | ? | ? | 14.5 | 1.54 | ERβ agonist |
| Coumestrol | – | 9.225 (0.0117–94) | 64.125 (0.41–185) | 0.14–80.0 | 0.07–27.0 | Xenoestrogen |
| Genistein | – | 0.445 (0.0012–16) | 33.42 (0.86–87) | 2.6–126 | 0.3–12.8 | Xenoestrogen |
| Equol | – | 0.2–0.287 | 0.85 (0.10–2.85) | ? | ? | Xenoestrogen |
| Daidzein | – | 0.07 (0.0018–9.3) | 0.7865 (0.04–17.1) | 2.0 | 85.3 | Xenoestrogen |
| Biochanin A | – | 0.04 (0.022–0.15) | 0.6225 (0.010–1.2) | 174 | 8.9 | Xenoestrogen |
| Kaempferol | – | 0.07 (0.029–0.10) | 2.2 (0.002–3.00) | ? | ? | Xenoestrogen |
| Naringenin | – | 0.0054 (<0.001–0.01) | 0.15 (0.11–0.33) | ? | ? | Xenoestrogen |
| 8-Prenylnaringenin | 8-PN | 4.4 | ? | ? | ? | Xenoestrogen |
| Quercetin | – | <0.001–0.01 | 0.002–0.040 | ? | ? | Xenoestrogen |
| Ipriflavone | – | <0.01 | <0.01 | ? | ? | Xenoestrogen |
| Miroestrol | – | 0.39 | ? | ? | ? | Xenoestrogen |
| Deoxymiroestrol | – | 2.0 | ? | ? | ? | Xenoestrogen |
| β-Sitosterol | – | <0.001–0.0875 | <0.001–0.016 | ? | ? | Xenoestrogen |
| Resveratrol | – | <0.001–0.0032 | ? | ? | ? | Xenoestrogen |
| α-Zearalenol | – | 48 (13–52.5) | ? | ? | ? | Xenoestrogen |
| β-Zearalenol | – | 0.6 (0.032–13) | ? | ? | ? | Xenoestrogen |
| Zeranol | α-Zearalanol | 48–111 | ? | ? | ? | Xenoestrogen |
| Taleranol | β-Zearalanol | 16 (13–17.8) | 14 | 0.8 | 0.9 | Xenoestrogen |
| Zearalenone | ZEN | 7.68 (2.04–28) | 9.45 (2.43–31.5) | ? | ? | Xenoestrogen |
| Zearalanone | ZAN | 0.51 | ? | ? | ? | Xenoestrogen |
| Bisphenol A | BPA | 0.0315 (0.008–1.0) | 0.135 (0.002–4.23) | 195 | 35 | Xenoestrogen |
| Endosulfan | EDS | <0.001–<0.01 | <0.01 | ? | ? | Xenoestrogen |
| Kepone | Chlordecone | 0.0069–0.2 | ? | ? | ? | Xenoestrogen |
| o,p'-DDT | – | 0.0073–0.4 | ? | ? | ? | Xenoestrogen |
| p,p'-DDT | – | 0.03 | ? | ? | ? | Xenoestrogen |
| Methoxychlor | p,p'-Dimethoxy-DDT | 0.01 (<0.001–0.02) | 0.01–0.13 | ? | ? | Xenoestrogen |
| HPTE | Hydroxychlor; p,p'-OH-DDT | 1.2–1.7 | ? | ? | ? | Xenoestrogen |
| Testosterone | T; 4-Androstenolone | <0.0001–<0.01 | <0.002–0.040 | >5000 | >5000 | Androgen |
| Dihydrotestosterone | DHT; 5α-Androstanolone | 0.01 (<0.001–0.05) | 0.0059–0.17 | 221–>5000 | 73–1688 | Androgen |
| Nandrolone | 19-Nortestosterone; 19-NT | 0.01 | 0.23 | 765 | 53 | Androgen |
| Dehydroepiandrosterone | DHEA; Prasterone | 0.038 (<0.001–0.04) | 0.019–0.07 | 245–1053 | 163–515 | Androgen |
| 5-Androstenediol | A5; Androstenediol | 6 | 17 | 3.6 | 0.9 | Androgen |
| 4-Androstenediol | – | 0.5 | 0.6 | 23 | 19 | Androgen |
| 4-Androstenedione | A4; Androstenedione | <0.01 | <0.01 | >10000 | >10000 | Androgen |
| 3α-Androstanediol | 3α-Adiol | 0.07 | 0.3 | 260 | 48 | Androgen |
| 3β-Androstanediol | 3β-Adiol | 3 | 7 | 6 | 2 | Androgen |
| Androstanedione | 5α-Androstanedione | <0.01 | <0.01 | >10000 | >10000 | Androgen |
| Etiocholanedione | 5β-Androstanedione | <0.01 | <0.01 | >10000 | >10000 | Androgen |
| Methyltestosterone | 17α-Methyltestosterone | <0.0001 | ? | ? | ? | Androgen |
| Ethinyl-3α-androstanediol | 17α-Ethynyl-3α-adiol | 4.0 | <0.07 | ? | ? | Estrogen |
| Ethinyl-3β-androstanediol | 17α-Ethynyl-3β-adiol | 50 | 5.6 | ? | ? | Estrogen |
| Progesterone | P4; 4-Pregnenedione | <0.001–0.6 | <0.001–0.010 | ? | ? | Progestogen |
| Norethisterone | NET; 17α-Ethynyl-19-NT | 0.085 (0.0015–<0.1) | 0.1 (0.01–0.3) | 152 | 1084 | Progestogen |
| Norethynodrel | 5(10)-Norethisterone | 0.5 (0.3–0.7) | <0.1–0.22 | 14 | 53 | Progestogen |
| Tibolone | 7α-Methylnorethynodrel | 0.5 (0.45–2.0) | 0.2–0.076 | ? | ? | Progestogen |
| Δ4-Tibolone | 7α-Methylnorethisterone | 0.069–<0.1 | 0.027–<0.1 | ? | ? | Progestogen |
| 3α-Hydroxytibolone | – | 2.5 (1.06–5.0) | 0.6–0.8 | ? | ? | Progestogen |
| 3β-Hydroxytibolone | – | 1.6 (0.75–1.9) | 0.070–0.1 | ? | ? | Progestogen |
| Footnotes: a = (1) Binding affinity values are of the format "median (range)" (# (#–#)), "range" (#–#), or "value" (#) depending on the values available. The full sets of values within the ranges can be found in the Wiki code. (2) Binding affinities were determined via displacement studies in a variety of in-vitro systems with labeled estradiol and human ERα and ERβ proteins (except the ERβ values from Kuiper et al. (1997), which are rat ERβ). Sources: See template page. | ||||||
Binding and functional selectivity
[edit]The ER's helix 12 domain plays a crucial role in determining interactions with coactivators and corepressors and, therefore, the respective agonist or antagonist effect of the ligand.[39][40]
Different ligands may differ in their affinity for alpha and beta isoforms of the estrogen receptor:
- estradiol binds equally well to both receptors[41]
- estrone, and raloxifene bind preferentially to the alpha receptor[41]
- estriol, and genistein to the beta receptor[41]
Subtype selective estrogen receptor modulators preferentially bind to either the α- or the β-subtype of the receptor. In addition, the different estrogen receptor combinations may respond differently to various ligands, which may translate into tissue selective agonistic and antagonistic effects.[42] The ratio of α- to β- subtype concentration has been proposed to play a role in certain diseases.[43]
The concept of selective estrogen receptor modulators is based on the ability to promote ER interactions with different proteins such as transcriptional coactivator or corepressors. Furthermore, the ratio of coactivator to corepressor protein varies in different tissues.[44] As a consequence, the same ligand may be an agonist in some tissue (where coactivators predominate) while antagonistic in other tissues (where corepressors dominate). Tamoxifen, for example, is an antagonist in breast and is, therefore, used as a breast cancer treatment[25] but an ER agonist in bone (thereby preventing osteoporosis) and a partial agonist in the endometrium (increasing the risk of uterine cancer).
Discovery
[edit]Estrogen receptors were first identified by Elwood V. Jensen at the University of Chicago in 1958,[45][46] for which Jensen was awarded the Lasker Award.[47] The gene for a second estrogen receptor (ERβ) was identified in 1996 by Kuiper et al. in rat prostate and ovary using degenerate ERalpha primers.[48]
See also
[edit]- Membrane estrogen receptor
- Estrogen insensitivity syndrome
- Aromatase deficiency
- Aromatase excess syndrome
References
[edit]- ^ Dahlman-Wright K, Cavailles V, Fuqua SA, Jordan VC, Katzenellenbogen JA, Korach KS, et al. (December 2006). "International Union of Pharmacology. LXIV. Estrogen receptors". Pharmacological Reviews. 58 (4): 773–781. doi:10.1124/pr.58.4.8. PMID 17132854. S2CID 45996586.
- ^ a b Levin ER (August 2005). "Integration of the extranuclear and nuclear actions of estrogen". Molecular Endocrinology. 19 (8): 1951–1959. doi:10.1210/me.2004-0390. PMC 1249516. PMID 15705661.
- ^ Li X, Huang J, Yi P, Bambara RA, Hilf R, Muyan M (September 2004). "Single-chain estrogen receptors (ERs) reveal that the ERalpha/beta heterodimer emulates functions of the ERalpha dimer in genomic estrogen signaling pathways". Molecular and Cellular Biology. 24 (17): 7681–7694. doi:10.1128/MCB.24.17.7681-7694.2004. PMC 506997. PMID 15314175.
- ^ Nilsson S, Mäkelä S, Treuter E, Tujague M, Thomsen J, Andersson G, et al. (October 2001). "Mechanisms of estrogen action". Physiological Reviews. 81 (4): 1535–1565. doi:10.1152/physrev.2001.81.4.1535. PMID 11581496. S2CID 10223568.
- ^ Leung YK, Mak P, Hassan S, Ho SM (August 2006). "Estrogen receptor (ER)-beta isoforms: a key to understanding ER-beta signaling". Proceedings of the National Academy of Sciences of the United States of America. 103 (35): 13162–13167. Bibcode:2006PNAS..10313162L. doi:10.1073/pnas.0605676103. PMC 1552044. PMID 16938840.
- ^ Hawkins MB, Thornton JW, Crews D, Skipper JK, Dotte A, Thomas P (September 2000). "Identification of a third distinct estrogen receptor and reclassification of estrogen receptors in teleosts". Proceedings of the National Academy of Sciences of the United States of America. 97 (20): 10751–10756. Bibcode:2000PNAS...9710751H. doi:10.1073/pnas.97.20.10751. PMC 27095. PMID 11005855.
- ^ Couse JF, Lindzey J, Grandien K, Gustafsson JA, Korach KS (November 1997). "Tissue distribution and quantitative analysis of estrogen receptor-alpha (ERalpha) and estrogen receptor-beta (ERbeta) messenger ribonucleic acid in the wild-type and ERalpha-knockout mouse". Endocrinology. 138 (11): 4613–4621. doi:10.1210/en.138.11.4613. PMID 9348186.
- ^ a b Yaghmaie F, Saeed O, Garan SA, Freitag W, Timiras PS, Sternberg H (June 2005). "Caloric restriction reduces cell loss and maintains estrogen receptor-alpha immunoreactivity in the pre-optic hypothalamus of female B6D2F1 mice" (PDF). Neuro Endocrinology Letters. 26 (3): 197–203. PMID 15990721.
- ^ Hess RA (July 2003). "Estrogen in the adult male reproductive tract: a review". Reproductive Biology and Endocrinology. 1 (52): 52. doi:10.1186/1477-7827-1-52. PMC 179885. PMID 12904263.
- ^ Babiker FA, De Windt LJ, van Eickels M, Grohe C, Meyer R, Doevendans PA (February 2002). "Estrogenic hormone action in the heart: regulatory network and function". Cardiovascular Research. 53 (3): 709–719. doi:10.1016/S0008-6363(01)00526-0. PMID 11861041.
- ^ Htun H, Holth LT, Walker D, Davie JR, Hager GL (February 1999). "Direct visualization of the human estrogen receptor alpha reveals a role for ligand in the nuclear distribution of the receptor". Molecular Biology of the Cell. 10 (2): 471–486. doi:10.1091/mbc.10.2.471. PMC 25181. PMID 9950689.
- ^ Pfeffer U, Fecarotta E, Vidali G (May 1995). "Coexpression of multiple estrogen receptor variant messenger RNAs in normal and neoplastic breast tissues and in MCF-7 cells". Cancer Research. 55 (10): 2158–2165. PMID 7743517.
- ^ Yaşar P, Ayaz G, User SD, Güpür G, Muyan M (January 2017). "Molecular mechanism of estrogen-estrogen receptor signaling". Reproductive Medicine and Biology. 16 (1): 4–20. doi:10.1002/rmb2.12006. PMC 5715874. PMID 29259445.
- ^ Fuentes N, Silveyra P (2019). "Estrogen receptor signaling mechanisms". Advances in Protein Chemistry and Structural Biology. 116: 135–170. doi:10.1016/bs.apcsb.2019.01.001. PMC 6533072. PMID 31036290.
- ^ a b c Dahlman-Wright K, Cavailles V, Fuqua SA, Jordan VC, Katzenellenbogen JA, Korach KS, et al. (December 2006). "International Union of Pharmacology. LXIV. Estrogen receptors". Pharmacological Reviews. 58 (4): 773–781. doi:10.1124/pr.58.4.8. PMID 17132854. S2CID 45996586.
- ^ Levin ER (August 2005). "Integration of the extranuclear and nuclear actions of estrogen". Molecular Endocrinology. 19 (8): 1951–1959. doi:10.1210/me.2004-0390. PMC 1249516. PMID 15705661.
- ^ Nath-Sain S, Marignani PA (June 2009). "LKB1 catalytic activity contributes to estrogen receptor alpha signaling". Molecular Biology of the Cell. 20 (11): 2785–2795. doi:10.1091/MBC.E08-11-1138. PMC 2688557. PMID 19369417.
- ^ Wang C, Fu M, Angeletti RH, Siconolfi-Baez L, Reutens AT, Albanese C, et al. (May 2001). "Direct acetylation of the estrogen receptor alpha hinge region by p300 regulates transactivation and hormone sensitivity". The Journal of Biological Chemistry. 276 (21): 18375–83. doi:10.1074/jbc.M100800200. PMID 11279135.
- ^ a b Zivadinovic D, Gametchu B, Watson CS (2005). "Membrane estrogen receptor-alpha levels in MCF-7 breast cancer cells predict cAMP and proliferation responses". Breast Cancer Research. 7 (1): R101–R112. doi:10.1186/bcr958. PMC 1064104. PMID 15642158.
- ^ Björnström L, Sjöberg M (June 2004). "Estrogen receptor-dependent activation of AP-1 via non-genomic signalling". Nuclear Receptor. 2 (1): 3. doi:10.1186/1478-1336-2-3. PMC 434532. PMID 15196329.
- ^ Lu Q, Pallas DC, Surks HK, Baur WE, Mendelsohn ME, Karas RH (December 2004). "Striatin assembles a membrane signaling complex necessary for rapid, nongenomic activation of endothelial NO synthase by estrogen receptor alpha". Proceedings of the National Academy of Sciences of the United States of America. 101 (49): 17126–17131. Bibcode:2004PNAS..10117126L. doi:10.1073/pnas.0407492101. PMC 534607. PMID 15569929.
- ^ Kato S, Endoh H, Masuhiro Y, Kitamoto T, Uchiyama S, Sasaki H, et al. (December 1995). "Activation of the estrogen receptor through phosphorylation by mitogen-activated protein kinase". Science. 270 (5241): 1491–1494. Bibcode:1995Sci...270.1491K. doi:10.1126/science.270.5241.1491. PMID 7491495. S2CID 4662264.
- ^ Prossnitz ER, Arterburn JB, Sklar LA (February 2007). "GPR30: A G protein-coupled receptor for estrogen". Molecular and Cellular Endocrinology. 265–266: 138–142. doi:10.1016/j.mce.2006.12.010. PMC 1847610. PMID 17222505.
- ^ Otto C, Rohde-Schulz B, Schwarz G, Fuchs I, Klewer M, Brittain D, et al. (October 2008). "G protein-coupled receptor 30 localizes to the endoplasmic reticulum and is not activated by estradiol". Endocrinology. 149 (10): 4846–4856. doi:10.1210/en.2008-0269. PMID 18566127.
- ^ a b Deroo BJ, Korach KS (March 2006). "Estrogen receptors and human disease". The Journal of Clinical Investigation. 116 (3): 561–570. doi:10.1172/JCI27987. PMC 2373424. PMID 16511588.
- ^ Harris HA, Albert LM, Leathurby Y, Malamas MS, Mewshaw RE, Miller CP, et al. (October 2003). "Evaluation of an estrogen receptor-beta agonist in animal models of human disease". Endocrinology. 144 (10): 4241–4249. doi:10.1210/en.2003-0550. PMID 14500559.
- ^ Clemons M, Danson S, Howell A (August 2002). "Tamoxifen ("Nolvadex"): a review". Cancer Treatment Reviews. 28 (4): 165–180. doi:10.1016/s0305-7372(02)00036-1. PMID 12363457.
- ^ Fabian CJ, Kimler BF (March 2005). "Selective estrogen-receptor modulators for primary prevention of breast cancer". Journal of Clinical Oncology. 23 (8): 1644–1655. doi:10.1200/JCO.2005.11.005. PMID 15755972.
- ^ Oesterreich S, Davidson NE (December 2013). "The search for ESR1 mutations in breast cancer". Nature Genetics. 45 (12): 1415–1416. doi:10.1038/ng.2831. PMC 4934882. PMID 24270445.
- ^ Li S, Shen D, Shao J, Crowder R, Liu W, Prat A, et al. (September 2013). "Endocrine-therapy-resistant ESR1 variants revealed by genomic characterization of breast-cancer-derived xenografts". Cell Reports. 4 (6): 1116–1130. doi:10.1016/j.celrep.2013.08.022. PMC 3881975. PMID 24055055.
- ^ Darabi M, Ani M, Panjehpour M, Rabbani M, Movahedian A, Zarean E (2011). "Effect of estrogen receptor β A1730G polymorphism on ABCA1 gene expression response to postmenopausal hormone replacement therapy". Genetic Testing and Molecular Biomarkers. 15 (1–2): 11–15. doi:10.1089/gtmb.2010.0106. PMID 21117950.
- ^ Hewitt KN, Boon WC, Murata Y, Jones ME, Simpson ER (September 2003). "The aromatase knockout mouse presents with a sexually dimorphic disruption to cholesterol homeostasis". Endocrinology. 144 (9): 3895–3903. doi:10.1210/en.2003-0244. PMID 12933663.
- ^ Danilovich N, Babu PS, Xing W, Gerdes M, Krishnamurthy H, Sairam MR (November 2000). "Estrogen deficiency, obesity, and skeletal abnormalities in follicle-stimulating hormone receptor knockout (FORKO) female mice". Endocrinology. 141 (11): 4295–4308. doi:10.1210/endo.141.11.7765. PMID 11089565.
- ^ Ohlsson C, Hellberg N, Parini P, Vidal O, Bohlooly-Y M, Rudling M, et al. (November 2000). "Obesity and disturbed lipoprotein profile in estrogen receptor-alpha-deficient male mice". Biochemical and Biophysical Research Communications. 278 (3): 640–645. doi:10.1006/bbrc.2000.3827. PMID 11095962.
- ^ a b Deng L, Wu T, Chen XY, Xie L, Yang J (October 2012). "Selective estrogen receptor modulators (SERMs) for uterine leiomyomas". The Cochrane Database of Systematic Reviews. 10: CD005287. doi:10.1002/14651858.CD005287.pub4. PMID 23076912.
- ^ a b van Hoesel MH, Chen YL, Zheng A, Wan Q, Mourad SM, et al. (Cochrane Gynaecology and Fertility Group) (May 2021). "Selective oestrogen receptor modulators (SERMs) for endometriosis". The Cochrane Database of Systematic Reviews. 2021 (5): CD011169. doi:10.1002/14651858.CD011169.pub2. PMC 8130989. PMID 33973648.
- ^ Lemke TL, Williams DA (24 January 2012). Foye's Principles of Medicinal Chemistry. Lippincott Williams & Wilkins. pp. 1392–. ISBN 978-1-60913-345-0.
- ^ Smith EP, Boyd J, Frank GR, Takahashi H, Cohen RM, Specker B, et al. (October 1994). "Estrogen resistance caused by a mutation in the estrogen-receptor gene in a man". The New England Journal of Medicine. 331 (16): 1056–1061. doi:10.1056/NEJM199410203311604. PMID 8090165.
- ^ Ascenzi P, Bocedi A, Marino M (August 2006). "Structure-function relationship of estrogen receptor alpha and beta: impact on human health". Molecular Aspects of Medicine. 27 (4): 299–402. doi:10.1016/j.mam.2006.07.001. PMID 16914190.
- ^ Bourguet W, Germain P, Gronemeyer H (October 2000). "Nuclear receptor ligand-binding domains: three-dimensional structures, molecular interactions and pharmacological implications". Trends in Pharmacological Sciences. 21 (10): 381–388. doi:10.1016/S0165-6147(00)01548-0. PMID 11050318.
- ^ a b c Zhu BT, Han GZ, Shim JY, Wen Y, Jiang XR (September 2006). "Quantitative structure-activity relationship of various endogenous estrogen metabolites for human estrogen receptor alpha and beta subtypes: Insights into the structural determinants favoring a differential subtype binding". Endocrinology. 147 (9): 4132–4150. doi:10.1210/en.2006-0113. PMID 16728493.
- ^ Kansra S, Yamagata S, Sneade L, Foster L, Ben-Jonathan N (July 2005). "Differential effects of estrogen receptor antagonists on pituitary lactotroph proliferation and prolactin release". Molecular and Cellular Endocrinology. 239 (1–2): 27–36. doi:10.1016/j.mce.2005.04.008. PMID 15950373. S2CID 42052008.
- ^ Bakas P, Liapis A, Vlahopoulos S, Giner M, Logotheti S, Creatsas G, et al. (November 2008). "Estrogen receptor alpha and beta in uterine fibroids: a basis for altered estrogen responsiveness". Fertility and Sterility. 90 (5): 1878–1885. doi:10.1016/j.fertnstert.2007.09.019. hdl:10442/7330. PMID 18166184.
- ^ Shang Y, Brown M (March 2002). "Molecular determinants for the tissue specificity of SERMs". Science. 295 (5564): 2465–2468. Bibcode:2002Sci...295.2465S. doi:10.1126/science.1068537. PMID 11923541. S2CID 30634073.
- ^ Jensen EV, Jordan VC (June 2003). "The estrogen receptor: a model for molecular medicine" (abstract). Clinical Cancer Research. 9 (6): 1980–1989. PMID 12796359.
- ^ Jensen E (2011). "A conversation with Elwood Jensen. Interview by David D. Moore". Annual Review of Physiology. 74: 1–11. doi:10.1146/annurev-physiol-020911-153327. PMID 21888507.
- ^ Bracey D (2004). "UC Scientist Wins 'American Nobel' Research Award". University of Cincinnati press release.
- ^ Kuiper GG, Enmark E, Pelto-Huikko M, Nilsson S, Gustafsson JA (June 1996). "Cloning of a novel receptor expressed in rat prostate and ovary". Proceedings of the National Academy of Sciences of the United States of America. 93 (12): 5925–5930. doi:10.1073/pnas.93.12.5925. PMC 39164. PMID 8650195.
External links
[edit]- Estrogen Receptors at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Goodsell DS (September 2003). "Estrogen Receptor". Protein Data Bank, Research Collaboratory for Structural Bioinformatics (RCSB). Archived from the original on March 11, 2006. Retrieved 2008-03-15.
