From Wikipedia, the free encyclopedia
Chemical compound
7α-Methylestradiol Other names 7α-Methyl-E2; 7α-Me-E2; 7α-Methylestra-1,3,5(10)-triene-3,17β-diol Drug class Estrogen
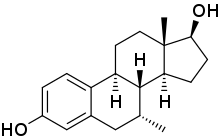
(7R ,8R ,9S ,13S ,14S ,17S )-7,13-Dimethyl-6,7,8,9,11,12,14,15,16,17-decahydrocyclopenta[a]phenanthrene-3,17-diol
CAS Number PubChem CID UNII Formula C 19 H 26 O 2 Molar mass −1 3D model (JSmol )
[H][C@@]12CC[C@H](O)[C@@]1(C)CC[C@]1([H])C3=CC=C(O)C=C3C[C@@H](C)[C@@]21[H]
InChI=1/C19H26O2/c1-11-9-12-10-13(20)3-4-14(12)15-7-8-19(2)16(18(11)15)5-6-17(19)21/h3-4,10-11,15-18,20-21H,5-9H2,1-2H3/t11-,15-,16+,17+,18-,19+/s2
Key:DXWWYJWUFULMAP-BQXDHOISNA-N
7α-Methylestradiol (7α-Me-E2 ), also known as 7α-methylestra-1,3,5(10)-triene-3,17β-diol , is a synthetic estrogen and an active metabolite of the androgen /anabolic steroids trestolone/Methandienone .[ 1] [ 2] [ 3] estrogenic activity of trestolone.[ 2] [ 3] affinity for the estrogen receptor than estradiol .[ 1]
Relative affinities (%) of 7α-methylestradiol and related steroids[ 1] [ 4] [ 5] [ 6]
Compound
PR Tooltip Progesterone receptor AR Tooltip Androgen receptor ER Tooltip Estrogen receptor GR Tooltip Glucocorticoid receptor MR Tooltip Mineralocorticoid receptor SHBG Tooltip Sex hormone-binding globulin CBG Tooltip Corticosteroid binding globulin
Estradiol 2.6
7.9
100
0.6
0.13
8.7
<0.1
7α-Methylestradiol
1–3
15–25
101
<1
<1
?
?
Trestolone 50–75
100–125
?
<1
?
?
?
Values are percentages (%). Reference ligands (100%) were progesterone for the PR Tooltip progesterone receptor , testosterone for the AR Tooltip androgen receptor , E2 ER Tooltip estrogen receptor , DEXA Tooltip dexamethasone for the GR Tooltip glucocorticoid receptor , aldosterone for the MR Tooltip mineralocorticoid receptor , DHT Tooltip dihydrotestosterone for SHBG Tooltip sex hormone-binding globulin , and cortisol for CBG Tooltip Corticosteroid-binding globulin .
^ a b c Raynaud JP, Ojasoo T, Bouton MM, Philibert D (1979). "Receptor Binding as a Tool in the Development of New Bioactive Steroids" . Drug Design . pp. 169–214. doi :10.1016/B978-0-12-060308-4.50010-X . ISBN 9780120603084 ^ a b García-Becerra R, Ordaz-Rosado D, Noé G, Chávez B, Cooney AJ, Larrea F (February 2012). "Comparison of 7α-methyl-19-nortestosterone effectiveness alone or combined with progestins on androgen receptor mediated-transactivation" . Reproduction . 143 (2): 211–219. doi :10.1530/REP-11-0171 PMID 22065861 . ^ a b Attardi BJ, Pham TC, Radler LC, Burgenson J, Hild SA, Reel JR (June 2008). "Dimethandrolone (7alpha,11beta-dimethyl-19-nortestosterone) and 11beta-methyl-19-nortestosterone are not converted to aromatic A-ring products in the presence of recombinant human aromatase" . The Journal of Steroid Biochemistry and Molecular Biology . 110 (3–5): 214–222. doi :10.1016/j.jsbmb.2007.11.009 . PMC 2575079 PMID 18555683 . ^ Ojasoo T, Delettré J, Mornon JP, Turpin-VanDycke C, Raynaud JP (1987). "Towards the mapping of the progesterone and androgen receptors". Journal of Steroid Biochemistry . 27 (1–3): 255–269. doi :10.1016/0022-4731(87)90317-7 . PMID 3695484 . ^ Ojasoo T, Raynaud JP (November 1978). "Unique steroid congeners for receptor studies" . Cancer Research . 38 (11 Pt 2): 4186–4198. PMID 359134 . ^ Raynaud JP, Bouton MM, Moguilewsky M, Ojasoo T, Philibert D, Beck G, et al. (January 1980). "Steroid hormone receptors and pharmacology". Journal of Steroid Biochemistry . 12 : 143–157. doi :10.1016/0022-4731(80)90264-2 . PMID 7421203 .
ER Tooltip Estrogen receptor
Agonists
Steroidal: 2-Hydroxyestradiol 2-Hydroxyestrone 3-Methyl-19-methyleneandrosta-3,5-dien-17β-ol 3α-Androstanediol 3α,5α-Dihydrolevonorgestrel 3β,5α-Dihydrolevonorgestrel 3α-Hydroxytibolone 3β-Hydroxytibolone 3β-Androstanediol 4-Androstenediol 4-Androstenedione 4-Fluoroestradiol 4-Hydroxyestradiol 4-Hydroxyestrone 4-Methoxyestradiol 4-Methoxyestrone 5-Androstenediol 7-Oxo-DHEA 7α-Hydroxy-DHEA 7α-Methylestradiol 7β-Hydroxyepiandrosterone 8,9-Dehydroestradiol 8,9-Dehydroestrone 8β-VE2 10β,17β-Dihydroxyestra-1,4-dien-3-one (DHED) 11β-Chloromethylestradiol 11β-Methoxyestradiol 15α-Hydroxyestradiol 16-Ketoestradiol 16-Ketoestrone 16α-Fluoroestradiol 16α-Hydroxy-DHEA 16α-Hydroxyestrone 16α-Iodoestradiol 16α-LE2 16β-Hydroxyestrone 16β,17α-Epiestriol (16β-hydroxy-17α-estradiol) 17α-Estradiol (alfatradiol )17α-Dihydroequilenin 17α-Dihydroequilin 17α-Epiestriol (16α-hydroxy-17α-estradiol) 17α-Ethynyl-3α-androstanediol 17α-Ethynyl-3β-androstanediol 17β-Dihydroequilenin 17β-Dihydroequilin 17β-Methyl-17α-dihydroequilenin Abiraterone Abiraterone acetate Alestramustine Almestrone Anabolic steroids (e.g., testosterone and esters , methyltestosterone , metandienone (methandrostenolone) , nandrolone and esters , many others; via estrogenic metabolites)Atrimustine Bolandiol Bolandiol dipropionate Butolame Clomestrone Cloxestradiol
Conjugated estriol Conjugated estrogens Cyclodiol Cyclotriol DHEA DHEA-S ent -EstradiolEpiestriol (16β-epiestriol, 16β-hydroxy-17β-estradiol) Epimestrol Equilenin Equilin ERA-63 (ORG-37663) Esterified estrogens Estetrol Estradiol
Estramustine Estramustine phosphate Estrapronicate Estrazinol Estriol
Estrofurate Estrogenic substances Estromustine Estrone
Etamestrol (eptamestrol) Ethinylandrostenediol
Ethinylestradiol
Ethinylestriol Ethylestradiol Etynodiol Etynodiol diacetate Hexolame Hippulin Hydroxyestrone diacetate Lynestrenol Lynestrenol phenylpropionate Mestranol Methylestradiol Moxestrol Mytatrienediol Nilestriol Norethisterone Noretynodrel Orestrate Pentolame Prodiame Prolame Promestriene RU-16117 Quinestradol Quinestrol Tibolone Xenoestrogens: Anise -related (e.g., anethole , anol , dianethole , dianol , photoanethole )Chalconoids (e.g., isoliquiritigenin , phloretin , phlorizin (phloridzin) , wedelolactone )Coumestans (e.g., coumestrol , psoralidin )Flavonoids (incl. 7,8-DHF , 8-prenylnaringenin , apigenin , baicalein , baicalin , biochanin A , calycosin , catechin , daidzein , daidzin , ECG , EGCG , epicatechin , equol , formononetin , glabrene , glabridin , genistein , genistin , glycitein , kaempferol , liquiritigenin , mirificin , myricetin , naringenin , penduletin , pinocembrin , prunetin , puerarin , quercetin , tectoridin , tectorigenin )Lavender oil Lignans (e.g., enterodiol , enterolactone , nyasol (cis -hinokiresinol) )Metalloestrogens (e.g., cadmium )Pesticides (e.g., alternariol , dieldrin , endosulfan , fenarimol , HPTE , methiocarb , methoxychlor , triclocarban , triclosan )Phytosteroids (e.g., digitoxin (digitalis ), diosgenin , guggulsterone )Phytosterols (e.g., β-sitosterol , campesterol , stigmasterol )Resorcylic acid lactones (e.g., zearalanone , α-zearalenol , β-zearalenol , zearalenone , zeranol (α-zearalanol) , taleranol (teranol, β-zearalanol) )Steroid -like (e.g., deoxymiroestrol , miroestrol )Stilbenoids (e.g., resveratrol , rhaponticin )Synthetic xenoestrogens (e.g., alkylphenols , bisphenols (e.g., BPA , BPF , BPS ), DDT , parabens , PBBs , PHBA , phthalates , PCBs )Others (e.g., agnuside , rotundifuran ) MixedSERMs Tooltip Selective estrogen receptor modulators ) Antagonists
Coregulator-binding modulators: ERX-11
GPER Tooltip G protein-coupled estrogen receptor
Agonists Antagonists Unknown