From Wikipedia, the free encyclopedia
Chemical compound
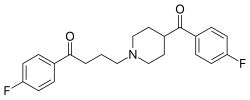
Lenperone (Elanone-V ) is a typical antipsychotic of the butyrophenone chemical class .[ 1] anti-emetic in 1974,[ 2] schizophrenia was reported in 1975.[ 1] declenperone and milenperone .
Lenperone was never approved by the FDA for use in humans in the United States,[ 3] [ 4] [ 5] [ 6]
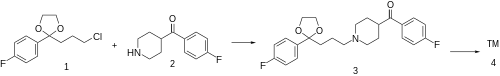
Synthesis (cmp#21):[ 7] [ 2] [ 8] [ 9] The alkylation between 2-(3-chloropropyl)-2-(4-fluorophenyl)-1,3-dioxolane [3308-94-9] (1 ) and 4-(4-fluorobenzoyl)piperidine [56346-57-7] (2 ) gives 2-(p-fluorophenyl)-2-{3-[4-(p-fluorobenzoyl)piperidino]propyl}-1,3-dioxolane, CID:20318874 (3 ). Deprotection of the ketal function completes the synthesis of lenperone (4 ).
Chemically related drugs containing the same 4-(p -fluorobenzoyl)piperidine group:
^ a b Harris M (1975). "Treatment of acute schizophrenia with a new butyrophenone-lenperone". Journal of Clinical Pharmacology . 15 (2–3): 187–190. doi :10.1002/j.1552-4604.1975.tb02355.x . PMID 1091666 . S2CID 28602974 . ^ a b FR 2227868 , Ward JW, Leonard CA, "Antiemetic compositions containing piperidine derivatives", published 1974-11-29 ^ Miller AC, Khan AM, Castro Bigalli AA, Sewell KA, King AR, Ghadermarzi S, et al. (2019). "Neuroleptanalgesia for acute abdominal pain: a systematic review" . Journal of Pain Research . 12 : 787–801. doi :10.2147/JPR.S187798 PMC 6396833 PMID 30881092 . Table 1. Marketed butyrophenones with approval status and indication ^ Booth NJ (1982). "Psychotropic Agents". In Booth NH, McDonald LE (ed.). Veterinary Pharmacology and Therapeutics (5th ed.). Ames, Iowa: Iowa State University Press. pp. 321–345. ^ Johnson SE, Zelner A, Sherding RG (April 1989). "Effect of lenperone hydrochloride on gastroesophageal sphincter pressure in healthy dogs" . Canadian Journal of Veterinary Research . 53 (2): 248–250. PMC 1255555 PMID 2565757 . ^ FDA Veterinarian . U.S. Department of Health and Human Services, Public Health Service, Food and Drug Administration, Center for Veterinary Medicine. 1988. The firm requested withdrawal of approval because the products are no longer being marketed. Effective date: July 13, 1989 ^ Duncan RL, Helsley GC, Welstead WJ, DaVanzo JP, Funderburk WH, Lunsford CD (January 1970). "Aroylpiperidines and pyrrolidines. A new class of potent central nervous system depressants". Journal of Medicinal Chemistry . 13 (1): 1–6. doi :10.1021/jm00295a001 . PMID 5460893 . ^ CN 107011133 , Yanbiao K, Jie L, issued 2017, assigned to University of Science and Technology of China USTC. ^ Liu J, Hu KF, Qu JP, Kang YB (October 2017). "Organopromoted Selectivity-Switchable Synthesis of Polyketones". Organic Letters . 19 (20): 5593–5596. doi :10.1021/acs.orglett.7b02731 . PMID 28981291 .
mAChRs Tooltip Muscarinic acetylcholine receptors
Agonists Antagonists
3-Quinuclidinyl benzilate 4-DAMP Aclidinium bromide (+formoterol )Abediterol AF-DX 250 AF-DX 384 Ambutonium bromide Anisodamine Anisodine Antihistamines (first-generation) (e.g., brompheniramine , buclizine , captodiame , chlorphenamine (chlorpheniramine) , cinnarizine , clemastine , cyproheptadine , dimenhydrinate , dimetindene , diphenhydramine , doxylamine , meclizine , mequitazine , perlapine , phenindamine , pheniramine , phenyltoloxamine , promethazine , propiomazine , triprolidine )AQ-RA 741 Atropine Atropine methonitrate Atypical antipsychotics (e.g., clozapine , fluperlapine , olanzapine (+fluoxetine ), rilapine , quetiapine , tenilapine , zotepine )Benactyzine Benzatropine (benztropine) Benzilone Benzilylcholine mustard Benzydamine Bevonium BIBN 99 Biperiden Bornaprine Camylofin CAR-226,086 CAR-301,060 CAR-302,196 CAR-302,282 CAR-302,368 CAR-302,537 CAR-302,668 Caramiphen Cimetropium bromide Clidinium bromide Cloperastine CS-27349 Cyclobenzaprine Cyclopentolate Darifenacin DAU-5884 Desfesoterodine Dexetimide DIBD Dicycloverine (dicyclomine) Dihexyverine Difemerine Diphemanil metilsulfate Ditran Drofenine EA-3167 EA-3443 EA-3580 EA-3834 Emepronium bromide Etanautine Etybenzatropine (ethybenztropine) Fenpiverinium Fentonium bromide Fesoterodine Flavoxate Glycopyrronium bromide (+beclometasone/formoterol , +indacaterol , +neostigmine )Hexahydrodifenidol Hexahydrosiladifenidol Hexbutinol Hexocyclium Himbacine HL-031,120 Homatropine Imidafenacin Ipratropium bromide (+salbutamol )Isopropamide J-104,129 Hyoscyamine Mamba toxin 3 Mamba toxin 7 Mazaticol Mebeverine Meladrazine Mepenzolate Methantheline Methoctramine Methylatropine Methylhomatropine Methylscopolamine Metixene Muscarinic toxin 7 N-Ethyl-3-piperidyl benzilate N-Methyl-3-piperidyl benzilate Nefopam Octatropine methylbromide (anisotropine methylbromide) Orphenadrine Otenzepad (AF-DX 116) Otilonium bromide Oxapium iodide Oxitropium bromide Oxybutynin Oxyphencyclimine Oxyphenonium bromide PBID PD-102,807 PD-0298029 Penthienate Pethidine pFHHSiD Phenglutarimide Phenyltoloxamine Pipenzolate bromide Piperidolate Pirenzepine Piroheptine Pizotifen Poldine Pridinol Prifinium bromide Procyclidine Profenamine (ethopropazine) Propantheline bromide Propiverine Quinidine 3-Quinuclidinyl thiochromane-4-carboxylate Revefenacin Rociverine RU-47,213 SCH-57,790 SCH-72,788 SCH-217,443 Scopolamine (hyoscine) Scopolamine butylbromide (hyoscine butylbromide) Silahexacyclium Sofpironium bromide Solifenacin SSRIs Tooltip Selective serotonin reuptake inhibitors (e.g., femoxetine , paroxetine )Telenzepine Terodiline Tetracyclic antidepressants (e.g., amoxapine , maprotiline , mianserin , mirtazapine )Tiemonium iodide Timepidium bromide Tiotropium bromide Tiquizium bromide Tofenacin Tolterodine Tricyclic antidepressants (e.g., amitriptyline (+perphenazine ), amitriptylinoxide , butriptyline , cidoxepin , clomipramine , desipramine , desmethyldesipramine , dibenzepin , dosulepin (dothiepin) , doxepin , imipramine , lofepramine , nitroxazepine , northiaden (desmethyldosulepin) , nortriptyline , protriptyline , quinupramine , trimipramine )Tridihexethyl Trihexyphenidyl Trimebutine Tripitamine (tripitramine) Tropacine Tropatepine Tropicamide Trospium chloride Typical antipsychotics (e.g., chlorpromazine , chlorprothixene , cyamemazine (cyamepromazine) , loxapine , mesoridazine , thioridazine )Umeclidinium bromide (+vilanterol )WIN-2299 Xanomeline Zamifenacin
Precursors (and prodrugs )
nAChRs Tooltip Nicotinic acetylcholine receptors
Agonists PAMs Tooltip positive allosteric modulators )
5-HIAA 6-Chloronicotine A-84,543 A-366,833 A-582,941 A-867,744 ABT-202 ABT-418 ABT-560 ABT-894 Acetylcholine Altinicline Anabasine Anatabine Anatoxin-a AR-R17779 Bephenium hydroxynaphthoate Butinoline Butyrylcholine Carbachol Choline Cotinine Cytisine Decamethonium Desformylflustrabromine Dianicline Dimethylphenylpiperazinium Epibatidine Epiboxidine Ethanol (alcohol) Ethoxysebacylcholine EVP-4473 EVP-6124 Galantamine GTS-21 Ispronicline Ivermectin JNJ-39393406 Levamisole Lobeline MEM-63,908 (RG-3487) Morantel Nicotine (tobacco )NS-1738 PHA-543,613 PHA-709,829 PNU-120,596 PNU-282,987 Pozanicline Pyrantel Rivanicline RJR-2429 Sazetidine A SB-206553 Sebacylcholine SIB-1508Y SIB-1553A SSR-180,711 Suberyldicholine Suxamethonium (succinylcholine) Suxethonium (succinyldicholine) TC-1698 TC-1734 TC-1827 TC-2216 TC-5214 TC-5619 TC-6683 Tebanicline Tribendimidine Tropisetron UB-165 Varenicline WAY-317,538 XY-4083 Antagonists NAMs Tooltip negative allosteric modulators )
Precursors (and prodrugs )
H1
Agonists Antagonists
Others: Atypical antipsychotics (e.g., aripiprazole , asenapine , brexpiprazole , brilaroxazine , clozapine , iloperidone , olanzapine , paliperidone , quetiapine , risperidone , ziprasidone , zotepine )Phenylpiperazine antidepressants (e.g., hydroxynefazodone , nefazodone , trazodone , triazoledione )Tetracyclic antidepressants (e.g., amoxapine , loxapine , maprotiline , mianserin , mirtazapine , oxaprotiline )Tricyclic antidepressants (e.g., amitriptyline , butriptyline , clomipramine , desipramine , dosulepin (dothiepin) , doxepin , imipramine , iprindole , lofepramine , nortriptyline , protriptyline , trimipramine )Typical antipsychotics (e.g., chlorpromazine , flupenthixol , fluphenazine , loxapine , perphenazine , prochlorperazine , thioridazine , thiothixene )
H2
H3
H4