Selegiline
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /səˈlɛdʒɪliːn/ sə-LEJ-i-leen ("seh-LEH-ji-leen")[1][2] |
| Trade names | Eldepryl, Jumex, Zelapar, Emsam, Anipryl, others[3] |
| Other names | L-Deprenyl; L-Deprenil; L-Deprenalin; L-Deprenaline; L-E-250; l-E-250; L-Phenylisopropyl |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a697046 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth,[4][5] buccal (ODT),[6][7] transdermal[8][9] |
| Drug class | MAO-B inhibitor; Norepinephrine releasing agent; Antidepressant |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | Oral: 4–10%[5][11][12] ODT: ~5–8× oral[13][7][14] Transdermal: 75%[9] |
| Protein binding | 85–90%[9][8][6] |
| Metabolism | Liver, other tissues (CYP2B6, CYP2C19, others)[5][21][9][22] |
| Metabolites | • Desmethylselegiline (DMS) • Levomethamphetamine (L-MA) • Levoamphetamine (L-A) |
| Onset of action | Oral: ≤1 hour[15][16] |
| Elimination half-life | Oral: • S (single): 1.2–3.5 h[5] • S (multi): 7.7–9.7 h[5][12] • DMS (single): 2.2–3.8 h[5] • DMS (multi): 9.5 h[5] • L-MA: 14–21 h[5][7] • L-A: 16–18 h[5][7] ODT: • S (single): 1.3 h[6] • S (multi): 10 h[6] Patch: • S: 20 h[12][8] |
| Duration of action | |
| Excretion | Urine (87%):[18][19][7][5][20] • L-MA: 20–63% • L-A: 9–26% • DMS: 1% • S: 0.01–0.03% Feces: 15%[18][7] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.109.269 |
| Chemical and physical data | |
| Formula | C13H17N |
| Molar mass | 187.286 g·mol−1 |
| 3D model (JSmol) | |
| Chirality | Levorotatory enantiomer |
| |
| |
| (verify) | |
Selegiline, also known as L-deprenyl and sold under the brand names Eldepryl, Zelapar, and Emsam among others, is a medication which is used in the treatment of Parkinson's disease and major depressive disorder.[4][6][8][3] It has also been studied and used off-label for a variety of other indications, but has not been formally approved for any other use.[23][24] The medication, in the form licensed for depression, has modest effectiveness for this condition that is similar to that of other antidepressants.[24][25][26] Selegiline is provided as a swallowed tablet or capsule[4][5] or an orally disintegrating tablet (ODT)[6][7] for Parkinson's disease and as a patch applied to skin for depression.[8][9]
Side effects of selegiline occurring more often than with placebo include insomnia, dry mouth, dizziness, anxiety, abnormal dreams, and application site reactions (with the patch form), among others.[24][25][27][4][8] At high doses, selegiline has the potential for dangerous food and drug interactions, such as tyramine-related hypertensive crisis (the so-called "cheese reaction") and risk of serotonin syndrome.[9][28][5] However, doses within the approved clinical range appear to have little to no risk of these interactions.[9][28][5] In addition, the ODT and transdermal patch forms of selegiline have reduced risks of such interactions compared to the conventional oral form.[7][9] Selegiline has no known misuse potential or dependence liability and is not a controlled substance except in Japan.[29][30][31][32][8][33]
Selegiline acts as a monoamine oxidase inhibitor (MAOI) and thereby increases levels of monoamine neurotransmitters in the brain.[17][11][28][5] At typical clinical doses used for Parkinson's disease, selegiline is a selective and irreversible inhibitor of monoamine oxidase B (MAO-B), increasing brain levels of dopamine.[17][11][28][5] At higher doses, it loses its specificity for MAO-B and also inhibits monoamine oxidase A (MAO-A), which increases serotonin and norepinephrine levels in the brain as well.[17][11][28][5] In addition to its MAOI activity, selegiline is a catecholaminergic activity enhancer (CAE) and enhances the impulse-mediated release of norepinephrine and dopamine in the brain.[34][35][36][37][28] This action may be mediated by TAAR1 agonism.[38][39][40] After administration, selegiline partially metabolizes into levomethamphetamine and levoamphetamine, which act as norepinephrine releasing agents (NRAs) and may contribute to its therapeutic and adverse effects as well.[41][31][42] The levels of these metabolites are much lower with the ODT and transdermal patch forms of selegiline.[7][9] Chemically, selegiline is a substituted phenethylamine and amphetamine,[43] a derivative of methamphetamine,[43] and the purified levorotatory enantiomer of deprenyl (the racemic mixture of selegiline and D-deprenyl).[44][23]
Deprenyl was discovered and studied as an antidepressant in the early 1960s by Zoltan Ecseri, József Knoll, and other colleagues at Chinoin Pharmaceutical Company in Hungary.[44][23] Subsequently, selegiline was purified from deprenyl and was studied and developed itself.[44] Selegiline was first introduced for medical use, to treat Parkinson's disease, in Hungary in 1977.[45] It was subsequently approved in the United Kingdom in 1982 and in the United States in 1989.[45][46] The ODT was approved for Parkinson's disease in the United States in 2006 and in the European Union in 2010, while the patch was introduced for depression in the United States in 2006.[45][23] Selegiline was the first selective MAO-B inhibitor to be discovered and marketed.[13][47][48] In addition to its medical use, there has been interest in selegiline as a potential anti-aging drug and nootropic.[49][50][51] However, effects of this sort are controversial and uncertain.[49][52][53][54] Generic versions of selegiline are available in the case of the conventional oral form, but not in the case of the ODT or transdermal patch forms.[55][56]
Medical uses
[edit]Parkinson's disease
[edit]In its oral and ODT forms, selegiline is used to treat symptoms of Parkinson's disease (PD).[4][6] It is most often used as an adjunct to medications such as levodopa (L-DOPA), although it has been used off-label as a monotherapy.[57][58] The rationale for adding selegiline to levodopa is to decrease the required dose of levodopa and thus reduce the motor complications of levodopa therapy.[59] Selegiline delays the point when levodopa treatment becomes necessary from about 11 months to about 18 months after diagnosis.[60] There is some evidence that selegiline acts as a neuroprotective and reduces the rate of disease progression, though this is disputed.[58][59] In addition to parkinsonism, selegiline can improve symptoms of depression in people with Parkinson's disease.[61][62] There is evidence that selegiline may be more effective than rasagiline in the treatment of Parkinson's disease.[23][38][63] This may be due to pharmacological differences between the drugs, such as the catecholaminergic activity enhancer (CAE) actions of selegiline which rasagiline lacks.[23][38][63][35]
Depression
[edit]Selegiline is used as an antidepressant in the treatment of major depressive disorder (MDD).[8][24] Both the oral selegiline and transdermal selegiline patch formulations are used in the treatment of depression.[24] However, oral selegiline is not approved for depression and is used off-label for this indication, while the transdermal patch is specifically licensed for treatment of depression.[4][8] Both standard clinical doses of oral selegiline (up to 10 mg/day) and higher doses of oral selegiline (e.g., 30 to 60 mg/day) have been used to treat depression, with the lower doses selectively inhibiting MAO-B and the higher doses producing dual inhibition of both MAO-A and MAO-B.[9][24] Unlike oral selegiline, transdermal selegiline bypasses first-pass metabolism, thereby avoiding inhibition of gastrointestinal and hepatic MAO-A and minimizing the risk of food and drug interactions, whilst still allowing for selegiline to reach the brain and inhibit MAO-B.[9]
A 2023 systematic review and meta-analysis evaluated the effectiveness and safety of selegiline in the treatment of psychiatric disorders including depression.[24] It included both randomized and non-randomized published clinical studies.[24] The meta-analysis found that selegiline was more effective than placebo in terms of reduction in depressive symptoms (SMD = −0.96, k = 10, n = 1,308), response rates for depression improvement (RR = 1.61, k = 9, n = 1,238), and response rates for improvement of depression with atypical features (RR = 2.23, k = 3, n = 136).[24] Oral selegiline was significantly more effective than the selegiline patch in terms of depressive symptom improvement (SMD = −1.49, k = 6, n = 282 vs. SMD = −0.27, k = 4, n = 1,026, respectively; p = 0.03).[24] However, this was largely due to older and less methodologically rigorous trials that were at high risk for bias.[24] Oral selegiline studies also often employed much higher doses than usual, for instance 20 to 60 mg/day.[24] The quality of evidence of selegiline for depression was rated as very low overall, very low for oral selegiline, and low to moderate for transdermal selegiline.[24] For comparison, meta-analyses of other antidepressants for depression have found a mean effect size of about 0.3 (a small effect),[26][64] which is similar to that with transdermal selegiline.[24]
In two pivotal regulatory clinical trials of 6 to 8 weeks duration, the selegiline transdermal patch decreased scores on depression rating scales (specifically the 17- and 28-item HDRS) by 9.0 to 10.9 points, whereas placebo decreased scores by 6.5 to 8.6 points, giving placebo-subtracted differences attributable to selegiline of 2.4 to 2.5 points.[8] A 2013 quantitative review of the transdermal selegiline patch for depression, which pooled the results of these two trials, found that the placebo-subtracted number needed to treat (NNT) was 11 in terms of depression response (>50% reduction in symptoms) and 9 in terms of remission of depression (score of ≤10 on the MADRS).[25] For comparison, other antidepressants, including fluoxetine, paroxetine, duloxetine, vilazodone, adjunctive aripiprazole, olanzapine/fluoxetine, and extended-release quetiapine, have NNTs ranging from 6 to 8 in terms of depression response and 7 to 14 in terms of depression remission.[25] On the basis of these results, it was concluded that transdermal selegiline has similar effectiveness to other antidepressants.[25][65] NNTs are measures of effect size and indicate how many individuals would need to be treated in order to encounter one additional outcome of interest.[25] Lower NNTs are better, and NNTs corresponding to Cohen's d effect sizes have been defined as 2.3 for a large effect (d = 0.8), 3.6 for a medium effect (d = 0.5), and 8.9 for a small effect (d = 0.2).[25] The effectiveness of transdermal selegiline for depression relative to side effects and discontinuation was considered to be favorable.[25]
While several large regulatory clinical trials of transdermal selegiline versus placebo for depression have been conducted, there is a lack of trials comparing selegiline to other antidepressants.[56][65] Although multiple doses of transdermal selegiline were assessed, a dose–response relationship for depression was never established.[56][65] Transdermal selegiline has shown similar clinical effectiveness in the treatment of atypical depression relative to typical depression and in the treatment of anxious depression relative to non-anxious depression.[56][66][65]
Transdermal selegiline does not cause sexual dysfunction and may improve certain domains of sexual function, for instance sexual interest, maintaining interest during sex, and sexual satisfaction.[67] These benefits were apparent in women but not in men.[67] The lack of sexual dysfunction with transdermal selegiline is in contrast to many other antidepressants, such as the selective serotonin reuptake inhibitors (SSRIs) and serotonin–norepinephrine reuptake inhibitors (SNRIs), which are associated with high rates of sexual dysfunction.[68]
Transdermal selegiline patches have been underutilized in the treatment of depression compared to other antidepressants.[56][65] A variety of factors contributing to this underutilization have been identified.[56] One major factor is the very high cost of transdermal selegiline, which is often not covered by insurance and frequently proves to be prohibitive.[56][65] Conversely, other widely available antidepressants are much cheaper in comparison.[56][65]
Available forms
[edit]Selegiline is available in the following three pharmaceutical forms:[55]
- Oral tablets and capsules 5 mg (brand names Eldepryl, Jumex, and generics) – indicated for Parkinson's disease[4][5][45]
- Orally disintegrating tablets (ODTs) 1.25 mg (brand name Zelapar) – indicated for Parkinson's disease[6][7]
- Transdermal patches 6, 9, and 12 mg/24 hours (brand name Emsam) – indicated for major depressive disorder[8][9][12][27][65]

The transdermal patch form is also known as the "selegiline transdermal system" or "STS" and is applied once daily.[9][12][27][65][8] They are 20, 30, or 40 cm2 in size and contain a total of 20, 30, or 40 mg selegiline per patch (so 20 mg/20 cm2, 30 mg/30 cm2, and 40 mg/40 cm2), respectively.[8][65] The selegiline transdermal patch is a matrix-type adhesive patch with a three-layer structure.[8][65] It is the only approved non-oral MAOI, having reduced dietary restrictions and side effects in comparison to oral MAOIs, and is also the only approved non-oral first-line antidepressant.[65] The selegiline patch can be useful for those who have difficulty tolerating oral medications.[65]
Contraindications
[edit]Selegiline is contraindicated with serotonergic antidepressants including selective serotonin reuptake inhibitors (SSRIs), serotonin–norepinephrine reuptake inhibitors (SNRIs), and tricyclic antidepressants (TCAs), with serotonergic opioids like meperidine, tramadol, and methadone, with other monoamine oxidase inhibitors (MAOIs) such as linezolid, phenelzine, and tranylcypromine, and with dextromethorphan, St. John's wort, cyclobenzaprine, pentazocine, propoxyphene, and carbamazepine.[6][8][4] Combination of selegiline with serotonergic agents may cause serotonin syndrome, while combination of selegiline with adrenergic or sympathomimetic agents like ephedrine or amphetamines may cause hypertensive crisis.[6][8] Long washout periods are required before starting and stopping these medications with discontinuation or initiation of selegiline.[6][8][4][65]
Consumption of tyramine-rich foods can result in hypertensive crisis with selegiline, also known as the "cheese effect" or "cheese reaction" due to the high amounts of tyramine present in some cheeses.[6][11][47][15] Examples of other foods that may have high amounts of tyramine and similar substances include yeast products, chicken liver, snails, pickled herring, red wines, some beers, canned figs, broad beans, chocolate, and cream products.[15]
The preceding drug and food contraindications are dependent on selegiline dose and route, and hence are not necessarily absolute contraindications.[4][6][5][7][9] While high oral doses of selegiline (≥20 mg/day) can cause such interactions, oral doses within the approved clinical range (≤10 mg/day) appear to have little to no risk of these interactions.[9][28][5] In addition, the ODT and transdermal forms of selegiline have reduced risks of such interactions compared to the conventional oral form.[7][9]
Selegiline is also contraindicated in children less than 12 years of age and in people with pheochromocytoma, both due to heightened risk of hypertensive crisis.[8] For all human uses and all forms, selegiline is pregnancy category C, meaning that studies in pregnant animals have shown adverse effects on the fetus but there are no adequate studies in humans.[4][8]
Side effects
[edit]Side effects of the tablet form in conjunction with levodopa include, in decreasing order of frequency, nausea, hallucinations, confusion, depression, loss of balance, insomnia, increased involuntary movements, agitation, slow or irregular heart rate, delusions, hypertension, new or increased angina pectoris, and syncope.[4] Most of the side effects are due to a high dopamine levels, and can be alleviated by reducing the dose of levodopa.[3] Selegiline can also cause cardiovascular side effects such as orthostatic hypotension, hypertension, atrial fibrillation, and other types of cardiac arrhythmias.[69]
The main side effects of the patch form for depression include application-site reactions, insomnia, dry mouth, dizziness, nervousness, and abnormal dreams.[8][27] The selegiline patch carries a black box warning about a possible increased risk of suicide, especially for young people,[8] as do all antidepressants since 2007.[70]
Side effects of selegiline that have been identified as occurring significantly more often than with placebo in meta-analyses for psychiatric disorders have included dry mouth (RR = 1.58), insomnia (RR = 1.61, NNH = 19), and application site reactions with the transdermal form (RR = 1.81, NNH = 7).[24][25] No significant diarrhea, headache, dizziness, nausea, sexual dysfunction, or weight gain were apparent in these meta-analyses.[24][25]
Selegiline, including in its oral, ODT, and patch forms, has been found to cause hypotension or orthostatic hypotension in some individuals.[4][6][8] In a clinical trial, the rate of systolic orthostatic hypotension was 21% versus 9% with placebo and the rate of diastolic orthostatic hypotension was 12% versus 4% with placebo in people with Parkinson's disease taking the ODT form of selegiline.[6] The risk of hypotension is greater at the start of treatment and in the elderly (3% vs. 0% with placebo).[6] The rate of hypotension or orthostatic hypotension with the selegiline patch was 2.2% versus 0.5% with placebo in clinical trials of people with depression.[27] Significant orthostatic blood pressure changes (≥10 mm Hg decrease) occurred in 9.8% versus 6.7% with placebo, but most of these cases were asymptomatic and heart rate was unchanged.[27][71] The rates of other orthostatic hypotension-related side effects in this population were dizziness or vertigo 4.9% versus 3.1% with placebo and fainting 0.5% versus 0.0% with placebo.[27] It is said that orthostatic hypotension is rarely seen with the selegiline transdermal patch compared to oral MAOIs.[56] Caution is advised against rapidly rising after sitting or lying, especially after prolonged periods or at the start of treatment, as this can result in fainting.[6][30][71] Falls are of particular concern in the elderly.[71] MAOIs like selegiline may lower blood pressure by increasing dopamine levels and activating dopamine receptors, by increasing levels of the false neurotransmitter octopamine, and/or by other mechanisms.[72]
Meta-analyses published in the 1990s found that the addition of selegiline to levodopa increased mortality in people with Parkinson's disease.[30] However, several subsequent meta-analyses with more trials and patients found no increase in mortality with selegiline added to levodopa.[30][73][74] If selegiline does increase mortality, it has been theorized that this may be due to cardiovascular side effects, such as its amphetamine-related sympathomimetic effects and its MAO inhibition-related hypotension.[75] Although selegiline does not seem to increase mortality, it appears to worsen cognition in people with Parkinson's disease over time.[76] Conversely, rasagiline does not seem to do so and can enhance cognition.[76]
Rarely, selegiline has been reported to induce or exacerbate impulse control disorders, pathological gambling, hypersexuality, and paraphilias in people with Parkinson's disease.[77][78][79][80][81][82][83][84] However, MAO-B inhibitors like selegiline causing impulse control disorders is uncommon, controversial, and less frequent than with dopamine receptor agonists like pramipexole.[77][78] Impulse control disorders with dopaminergic agents have been linked specifically to activation of dopamine D3 receptors in the globus pallidus.[85][86][87][88] Selegiline has also been reported to activate or worsen rapid eye movement (REM) sleep behavior disorder (RBD) in some people with Parkinson's disease.[89][90][91]
Selegiline has shown little or no misuse potential in humans or monkeys.[29][30][31][92][93][94] Likewise, it has no dependence potential in rodents.[32] This is in spite of its amphetamine active metabolites, levomethamphetamine and levoamphetamine, and is in contrast to agents like dextroamphetamine and dextromethamphetamine.[30][31][32][93][94] However, selegiline can strongly potentiate the reinforcing effects of exogenous β-phenethylamine by inhibiting its MAO-B-mediated metabolism.[31] Misuse of the combination of selegiline and β-phenethylamine has been reported.[95][96]
Overdose
[edit]Little information is available about clinically significant selegiline overdose.[4] The drug has been studied clinically at doses as high as 60 mg/day orally,[97][24] 10 mg/day as an ODT,[7] and 12 mg/24 hours as a transdermal patch.[9] In addition, deprenyl (the racemic form) has been clinically studied orally at doses as large as 100 mg/day.[17] During clinical development of oral selegiline, some individuals who were exposed to doses of 600 mg developed severe hypotension and psychomotor agitation.[4][6] Overdose may result in non-selective inhibition of both MAO-A and MAO-B and may be similar to overdose of other non-selective monoamine oxidase inhibitors (MAOIs) like phenelzine, isocarboxazid, and tranylcypromine.[4][6] Serotonin syndrome, hypertensive crisis, and/or death may occur with overdose.[4][6][8] No specific antidote to selegiline overdose is available.[8]
Interactions
[edit]Serotonin syndrome and hypertensive crisis
[edit]Both the oral and patch forms of selegiline come with strong warnings against combining it with drugs that could produce serotonin syndrome, such as selective serotonin reuptake inhibitors (SSRIs) and the cough medicine dextromethorphan.[4][8][98] Selegiline in combination with the opioid analgesic pethidine is not recommended, as it can lead to severe adverse effects.[98] Several other synthetic opioids such as tramadol and methadone, as well as various triptans, are also contraindicated due to potential for serotonin syndrome.[99][100]
All three forms of selegiline carry warnings about food restrictions to avoid hypertensive crisis that are associated with MAOIs.[4][6][8] The patch form was created in part to overcome food restrictions; clinical trials showed that it was successful.[25][8] Additionally, in post-marketing surveillance from April 2006 to October 2010, only 13 self-reports of possible hypertensive events or hypertension were made out of 29,141 exposures to the drug, and none were accompanied by objective clinical data.[25] The lowest dose of the patch method of delivery, 6 mg/24 hours, does not require any dietary restrictions.[101] Higher doses of the patch and oral formulations, whether in combination with the older non-selective MAOIs or in combination with the reversible MAO-A inhibitor (RIMA) moclobemide, require a low-tyramine diet.[98]
A study found that selegiline in transdermal patch form did not importantly modify the pharmacodynamic effects or pharmacokinetics of the sympathomimetic agents pseudoephedrine and phenylpropanolamine.[9][102] Likewise, oral selegiline at an MAO-B-selective dosage did not appear to modify the pharmacodynamic effects or pharmacokinetics of intravenous methamphetamine in another study.[103][104] Conversely, selegiline, also at MAO-B-selective doses, has been found to reduce the physiological and euphoric subjective effects of cocaine whilst not affecting its pharmacokinetics in some studies but not in others.[105][106][107][108][109][110] Cautious safe combination of MAOIs like selegiline with stimulants like lisdexamfetamine has been reported.[111][112][113] However, a hypertensive crisis with selegiline and ephedrine has also been reported.[4] The selegiline drug labels warn about combination of selegiline with indirectly-acting sympathomimetic agents, like amphetamines, ephedrine, pseudoephedrine, and phenylpropanolamine, due to the potential risk of hypertensive crisis, and recommend monitoring blood pressure with such combinations.[6][8] The combination of selegiline with certain other medications, like phenylephrine and buspirone, is also warned against for similar reasons.[8][12][114][71] In the case of phenylephrine, this drug is substantially metabolized by monoamine oxidase, including by both MAO-A and MAO-B.[115][116] Selegiline can interact with exogenous dopamine, which is metabolized by MAO-A and MAO-B, and result in hypertensive crisis as well.[117][118]
Besides norepinephrine releasing agents, selective norepinephrine reuptake inhibitors (NRIs) may be safe in combination with MAOIs like selegiline.[119][120][121] Potent NRIs, such as reboxetine, desipramine, protriptyline, and nortriptyline, can reduce or block the pressor effects of tyramine, including in those taking MAOIs.[119][120][121] This is by inhibiting the norepinephrine transporter (NET) and preventing entry of tyramine into presynaptic noradrenergic neurons where tyramine induces the release of norepinephrine.[119][120][121] As a result, NRIs may reduce the risk of tyramine-related hypertensive crisis in people taking MAOIs.[119][120][121] Norepinephrine–dopamine reuptake inhibitors (NDRIs), like methylphenidate and bupropion, are also considered to be safe in combination with MAOIs.[122] However, initiation at low doses and slow upward dose titration is advisable in the case of both NRIs and NDRIs due to possible potentiation of their effects and side effects by MAOIs.[122]
Cytochrome P450 inhibitors and inducers
[edit]The cytochrome P450 enzymes involved in the metabolism of selegiline have not been fully elucidated.[5][21] CYP2D6 and CYP2C19 metabolizer phenotypes did not significantly affect the pharmacokinetics of selegiline, suggesting that these enzymes are minimally involved in its metabolism and that inhibitors and inducers of these enzymes would not importantly affect its pharmacokinetics.[21][43][123][124] However, although most pharmacokinetic variables were unaffected, overall exposure to selegiline's metabolite levomethamphetamine was 46% higher in CYP2D6 poor metabolizers compared to extensive metabolizers and exposure to its metabolite desmethylselegiline was 68% higher in CYP2C19 poor metabolizers compared to extensive metabolizers.[43][123][124] As with the cases of CYP2D6 and CYP2C19, the strong CYP3A4 and CYP3A5 inhibitor itraconazole has minimal impact on the pharmacokinetics of selegiline, suggesting lack of major involvement of this enzyme as well.[21][125][6] On the other hand, the anticonvulsant carbamazepine, which is known to act as a strong inducer of CYP3A enzymes,[126] has paradoxically been found to increase exposure to selegiline and its metabolites levomethamphetamine and levoamphetamine by approximately 2-fold (with selegiline used as the transdermal patch form).[8][9] One enzyme thought to be majorly involved in the metabolism of selegiline based on in-vitro studies is CYP2B6.[5][21][9][22] However, there are no clinical studies of different CYP2B6 metabolizer phenotypes or of CYP2B6 inhibitors or inducers on the pharmacokinetics of selegiline.[47] In addition to CYP2B6, CYP2A6 may be involved in the metabolism of selegiline to a lesser extent.[47][127]
Birth control pills containing the synthetic estrogen ethinylestradiol and a progestin like gestodene or levonorgestrel have been found to increase peak levels and overall exposure to oral selegiline by 10- to 20-fold.[21][128][129] High levels of selegiline can lead to loss of MAO-B selectivity and inhibition of MAO-A as well.[21][129] This increases susceptibility to side effects and interactions of non-selective monoamine oxidase inhibitors (MAOIs), such as tyramine-induced hypertensive crisis and serotonin toxicity when combined with serotonergic medications.[21][129] However, this study had a small sample size of four individuals as well as other methodological limitations.[21][129] The precise mechanism underlying the interaction is unknown, but is likely related to cytochrome P450 inhibition and consequent inhibition of selegiline first-pass metabolism by ethinylestradiol.[21] In contrast to birth control pills containing ethinylestradiol, menopausal hormone therapy with estradiol and levonorgestrel did not modify peak levels of selegiline and only modestly increased overall exposure (+59%).[21][128][130] Hence, menopausal hormone therapy does not pose the same risk of interaction as ethinylestradiol-containing birth control pills when taken together with selegiline.[128][130]
Overall exposure to selegiline with oral selegiline has been found to be 23-fold lower in people taking anticonvulsants known to strongly activate drug-metabolizing enzymes.[131] The anticonvulsants included phenobarbital, phenytoin, carbamazepine, and amobarbital.[131] In a previous study however, carbamazepine specifically did not reduce selegiline exposure.[8][9] Phenobarbital and certain other anticonvulsants are known to strongly induce CYP2B6, one of the major enzymes believed to be involved in selegiline metabolism.[131] As such, it was concluded that strong CYP2B6 induction was most likely responsible for the dramatically reduced exposure to selegiline observed in the study.[131]
Selegiline inhibition of cytochrome P450 enzymes
[edit]Selegiline has been reported to inhibit several cytochrome P450 enzymes, including CYP2D6, CYP3A4/5, CYP2C19, CYP2B6, and CYP2A6.[8][132] It is a mechanism-based inhibitor (suicide inhibitor) of CYP2B6 and has been said to "potently" or "strongly" inhibit this enzyme in vitro.[133][132][134][135] It may inhibit the metabolism of bupropion, a major CYP2B6 substrate, into its active metabolite hydroxybupropion.[133][132][134] However, a study predicted that inhibition of CYP2B6 by selegiline would non-significantly affect exposure to bupropion.[135] Selegiline has not been listed or described as a clinically significant CYP2B6 inhibitor by the Food and Drug Administration (FDA) as of 2023.[126][8] One small study observing three patients found that selegiline was safe and well-tolerated in combination with bupropion.[134][136] In addition to CYP2B6 and other cytochrome P450 enzymes, selegiline is a potent mechanism-based inhibitor of CYP2A6 and may increase exposure to nicotine (a major CYP2A6 substrate).[137][138] By inhibiting cytochrome P450 enzymes like CYP2B6 and CYP1A2, selegiline may inhibit its own metabolism and thereby interact with itself.[138][139]
Other interactions
[edit]Dopamine antagonists like antipsychotics or metoclopramide, which block dopamine receptors and thereby antagonize the dopaminergic effects of selegiline, could potentially reduce the effectiveness of the medication.[6] Dopamine-depleting agents like reserpine and tetrabenazine, by reducing dopamine levels, can also oppose the effectiveness of dopaminergic medications like selegiline.[140]
Pharmacology
[edit]Pharmacodynamics
[edit]Selegiline has multiple known mechanisms of action in terms of its pharmacodynamic activity.[17][11][28][5] It is most notably an irreversible monoamine oxidase (MAO) inhibitor (MAOI).[17][11][28][5] More specifically, it is a selective inhibitor of monoamine oxidase B (MAO-B) at lower doses (≤10 mg/day) but additionally inhibits monoamine oxidase A (MAO-A) at higher doses (≥20 mg/day).[17][11][28][5] MAO-B inhibition is thought to result in increased levels of dopamine and β-phenethylamine, whereas MAO-A inhibition results in increased levels of serotonin, norepinephrine, and dopamine.[17][11][28][5] Selegiline is also a catecholaminergic activity enhancer (CAE) and enhances the action potential-evoked release of norepinephrine and dopamine.[34][35][36][37][28] The CAE activity of selegiline may be mediated by TAAR1 agonism.[38][39][40] Both the MAOI activity and CAE activity of selegiline may be involved in its therapeutic effects in the treatment of Parkinson's disease and depression.[35][23][38][63][141][37] According to József Knoll and other researchers, selegiline might have dopaminergic neuroprotective effects, might be able to modestly slow the rate of aging-related dopaminergic neurodegeneration, and might thereby have a disease-modifying effect in Parkinson's disease and antiaging effects generally.[23][142][35][143][144][145] However, these theoretical effects of selegiline have not been clearly demonstrated in humans as of present and remain to be substantiated.[54][23][146][147][148] Through its active metabolites levomethamphetamine (L-MA) and levoamphetamine (L-A), selegiline acts as a weak norepinephrine and/or dopamine releasing agent (NDRA).[41][31][42] The clinical significance of this action is unclear, but it may be relevant to the effects and side effects of selegiline, especially at higher doses.[41][31][42] Its active metabolite desmethylselegiline (DMS) also has MAOI and CAE activity and likely contributes to its effects as well.[41][17][149][150][151] Levels of selegiline's metabolites are much lower with the ODT and transdermal patch forms of selegiline than with the oral form and this may result in differences in its effects and side effects.[7][9]
Pharmacokinetics
[edit]
Selegiline is available in forms for use by multiple different routes of administration and its pharmacokinetics vary by route.[55][7][9][12] The bioavailability of the oral form of selegiline is 4 to 10%,[5][11][12] of the ODT is 5 to 8 times that of the oral form,[13][7][14] and of the transdermal patch is 75%.[9] The time to peak levels of selegiline with oral administration is about 0.5 to 1.5 hours.[5] The plasma protein binding of selegiline is 85 to 90%.[9][8][6] It is metabolized extensively in the liver by the cytochrome P450 enzyme CYP2B6 among other enzymes.[5][21][9][22] Metabolites of selegiline include desmethylselegiline (DMS), levomethamphetamine (L-MA), and levoamphetamine (L-A).[17][28][5][152] The oral form of selegiline is subject to strong first-pass metabolism and levels of the metabolites of selegiline are much lower with the ODT and transdermal patch forms than with the oral form.[7][9] The elimination half-lives of selegiline and its metabolites range from 1.2 to 10 hours for selegiline, 2.2 to 9.5 hours for DMS, 14 to 21 hours for levomethamphetamine, and 16 to 18 hours for levoamphetamine.[5][12][7][6][8] Selegiline and its metabolites are eliminated mainly in urine (87% in urine and 15% in feces via oral administration), with its metabolites accounting for virtually all of the eliminated material in the case of the oral form.[18][19][7][5][20] Hepatic impairment and renal impairment have been found to dramatically increase exposure to selegiline.[153][154][131][4][6]
Chemistry
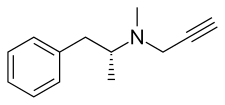
[edit]Selegiline is a substituted phenethylamine and amphetamine derivative.[43] It is also known as (R)-(–)-N,α-dimethyl-N-(2-propynyl)phenethylamine, (R)-(–)-N-methyl-N-2-propynylamphetamine, or N-propargyl-L-methamphetamine.[155][156][157][7] Selegiline (L-deprenyl) is the enantiopure levorotatory enantiomer of the racemic mixture deprenyl, whereas D-deprenyl is the dextrorotatory enantiomer.[44][23] Selegiline is a derivative of levomethamphetamine (L-methamphetamine), the levorotatory enantiomer of the psychostimulant and sympathomimetic agent methamphetamine (N-methylamphetamine), with a propargyl group attached to the nitrogen atom of the molecule.[65]
Selegiline is a small-molecule compound, with the molecular formula C13H17N and a low molecular weight of 187.281 g/mol.[155][156][157][4][65] It has high lipophilicity, with an experimental log P of 2.7 and predicted log P values of 2.9 to 3.1.[155][156][157][65] Pharmaceutically, selegiline is used almost always as the hydrochloride salt, though the free base form has also been used.[4][158] At room temperature, selegiline hydrochloride is a white to near white crystalline powder.[4] Selegiline hydrochloride is freely soluble in water, chloroform, and methanol.[4]
Analogues
[edit]Selegiline is a close analogue of methamphetamine and amphetamine, and in fact produces their levorotatory forms, levomethamphetamine and levoamphetamine, as metabolites.[41][31] Selegiline is structurally similar to the antihypertensive agent pargyline (N-methyl-N-propargylbenzylamine), an earlier non-selective MAOI of the phenylalkylamine group.[159][36] Besides selegiline and pargyline, another clinically used MAOI of the phenylalkylamine and amphetamine families is the antidepressant tranylcypromine (trans-2-phenylcyclopropylamine).[47] Tranylcypromine can be conceptualized as a cyclized amphetamine and has amphetamine-like actions at high doses similarly to selegiline.[47][160][161] Another notable analogue of selegiline is 4-fluoroselegiline, a variation of selegiline in which one of the hydrogen atoms of the phenyl ring has been replaced with a fluorine atom.[162] A large number of other analogues of selegiline derived via structural modification have been synthesized and characterized.[163][162][164][165]
Rasagiline ((R)-N-propargyl-1-aminoindan) is an analogue of selegiline in which the amphetamine base structure has been replaced with a 1-aminoindan structure and the N-methyl group has been removed.[41] Like selegiline, it is also a selective MAO-B inhibitor and used to treat Parkinson's disease.[41] In contrast to selegiline however, rasagiline lacks the amphetamine metabolites and activity of selegiline.[41] A further derivative of rasagiline, ladostigil ([N-propargyl-(3R)-aminoindan-5-yl]-N-propylcarbamate), a dual MAO-B inhibitor and acetylcholinesterase inhibitor, was developed for treatment of Alzheimer's disease and other conditions but was ultimately never introduced for medical use.[166]
Synthesis
[edit]Selegiline can be synthesized by the alkylation of levomethamphetamine using propargyl bromide.[47][167][168][169][170]

History
[edit]Following the discovery in 1952 that the tuberculosis drug iproniazid elevated the mood of people taking it, and the subsequent discovery that the effect was likely due to inhibition of monoamine oxidase (MAO) and elevation of monoamine neurotransmitters in the brain, many people and companies started trying to discover monoamine oxidase inhibitors (MAOIs) to use as antidepressants.[11][171] Deprenyl, the racemic form of selegiline, was synthesized and discovered by Zoltan Ecseri at the Chinoin Pharmaceutical Company (part of Sanofi since 1993) in Budapest, Hungary.[11][172] Chinoin received a patent on the drug in 1962 and the compound was first published in the scientific literature in English in 1965.[11][173] Chinoin researchers had been studying substituted amphetamines since 1960, and decided to try synthesizing amphetamines that acted as MAOIs.[15] It had been known that methamphetamine was a reversible inhibitor of MAO.[15] Deprenyl, also known as N-propargyl-N-methylamphetamine,[36] is closely related to and inspired by pargyline (N-propargyl-N-methylbenzylamine), another MAOI that had been synthesized earlier.[11][15][174] Deprenyl was initially referred to by the chemical name phenylisopropylmethylpropinylamine and the developmental code name E-250.[11][15][173] Work on the biology and effects of E-250 in animals and humans was conducted by a group led by József Knoll at Semmelweis University, which was also in Budapest.[11]
Deprenyl is a racemic compound (a mixture of two isomers called enantiomers).[11][15] The racemic form has mild amphetamine-like psychostimulant effects that are diminished compared to those of amphetamine but are still present.[15] The levorotatory enantiomer has further reduced stimulant effects, and further work, published in 1967, determined that the levorotatory enantiomer was a more potent MAOI than the dextrorotatory enantiomer.[11][15][175][176] As a result, subsequent work was done with the single enantiomer L-deprenyl.[11][15][175][176] In 1968, it was discovered by J. P. Johnston that monoamine oxidase exists in multiple forms.[11][15][177] In 1971, Knoll showed that selegiline highly selectively inhibits the B-isoform of monoamine oxidase (MAO-B) and proposed that it is unlikely to cause the infamous "cheese effect" (hypertensive crisis resulting from consuming foods containing tyramine) that occurs with non-selective MAOIs.[11][15][178] The lack of potentiation of tyramine effect by deprenyl had previously been reported in 1966 and 1968 studies, but could not be mechanistically explained until after the existence of multiple forms of MAO was discovered.[11][15][179] Selegiline was the first selective MAO-B inhibitor to be discovered[13] and hence is described as prototypical of these agents.[47][48]
Deprenyl and selegiline were initially studied as antidepressants for treatment of depression.[50][173] Deprenyl was first found to be effective for depression from 1965 to 1967,[50][180][181] while selegiline was first found to be effective for depression in 1971 and this was further corroborated in 1980.[50][182][183] A 1984 study that combined selegiline with phenylalanine reported remarkably high effectiveness in the treatment of depression similar to that with electroconvulsive therapy (ECT).[50][184] However, selegiline in its original oral form was never further developed or approved for the treatment of depression.[50]
A few years after the discovery that selegiline was a selective MAO-B inhibitor, two Parkinson's disease researchers based in Vienna, Peter Riederer and Walther Birkmayer, realized that selegiline could be useful in Parkinson's disease. One of their colleagues, Moussa B. H. Youdim, visited Knoll in Budapest and took selegiline from him to Vienna. In 1975, Birkmayer's group published the first paper on the effect of selegiline in Parkinson's disease.[175][185]
Speculation, by József Knoll, that selegiline could be useful as an anti-aging and pro-sexual agent, began in the 1980s.[15][186][187][188][189] The New York Times reported that selegiline was being used non-medically as a "smart drug" by 1992.[190]
Selegiline was first introduced for clinical use in Hungary in 1977.[45] It was approved in the oral pill form under the brand name Jumex to treat Parkinson's disease.[45] The drug was then introduced in the United Kingdom in 1982.[45] In 1987, Somerset Pharmaceuticals in New Jersey, which had acquired the rights to develop selegiline in the United States, filed a New Drug Application (NDA) with the Food and Drug Administration (FDA) to market the drug for Parkinson's disease in this country.[46] While the NDA was under review, Somerset was acquired in a joint venture by two generic drug companies, Mylan and Bolan Pharmaceuticals.[46] Selegiline was approved for Parkinson's disease by the FDA in 1989.[46]
It had been known since the mid-1960s that high doses of deprenyl had psychostimulant effects.[17][11][173][181] Selegiline was first shown to metabolize into levomethamphetamine and levoamphetamine in humans in 1978.[31][191] The involvement of these metabolites in the effects and side effects of selegiline has remained controversial and unresolved in the decades afterwards.[31][41] In any case, concerns about these metabolites have contributed to the development of newer MAO-B inhibitors like rasagiline and safinamide that lack such metabolites.[41][192]
The catecholaminergic activity enhancer (CAE) effects of selegiline became well-characterized and distinctly named in 1994.[193][35][28][150][23][37][194][195][196] These effects had been observed much earlier, dating back to the 1960s and 1970s, but were not properly distinguished from the other actions of selegiline, like MAO-B inhibition, until the 1990s.[35][28][37][193] More potent, selective, and/or expansive monoaminergic activity enhancers (MAEs), like phenylpropylaminopentane (PPAP) and benzofuranylpropylaminopentane (BPAP), were derived from selegiline and other compounds and were first described in 1988 and 1999, respectively.[36][39][197][50][198] These drugs had been proposed for potential treatment of psychiatric disorders like depression as well as for Parkinson's disease and Alzheimer's disease, but were never developed or marketed.[141][37][39][142][50]
In the 1990s, J. Alexander Bodkin at McLean Hospital, an affiliate of Harvard Medical School, began a collaboration with Somerset to develop delivery of selegiline via a transdermal patch in order to avoid the well known dietary restrictions of MAOIs.[189][199][200] Somerset obtained FDA approval to market the patch for depression in 2006.[201] Similarly, the orally disintegrating tablet (ODT) form of selegiline, marketed under the brand name Zelapar, was approved for Parkinson's disease in the United States in 2006 and in the European Union in 2010.[45]
Binding to and agonism of the trace amine-associated receptors (TAARs) as the mechanism responsible for the MAE effects of selegiline and related MAEs like PPAP and BPAP was first suggested in the early 2000s following the discovery of the TAARs.[39][142][40] Activation of the TAAR1 as the mechanism of the MAE effects was first clearly substantiated in 2022.[202][38]
Society and culture
[edit]Names
[edit]Selegiline is the generic name of the drug and its INN, BAN, and DCF, while selegiline hydrochloride is the USAN.[203][204][158] The word "selegiline" is pronounced /səˈlɛdʒɪliːn/ (sə-LEJ-i-leen) or as "seh-LEH-ji-leen".[1][2] Selegiline is also known as L-deprenyl, L-deprenil, L-deprenalin, L-deprenaline, L-phenylisopropylmethylpropinylamine, and L-E-250.[23][203][204][158][173] It should not be confused with the racemic form, deprenyl (E-250), or with the dextrorotatory enantiomer, D-deprenyl, which are distinct substances.[203][44][23]
Major brand names of selegiline include Eldepryl, Jumex, and Movergan (oral tablet and/or capsule), Zelapar (orally disintegrating tablet or ODT), and Emsam (transdermal patch).[3][158][152] Selegiline has been marketed under more than 70 brand names worldwide.[205][3] The brand name "Emsam" was derived from the names of two children, Emily and Samuel, of one of the executives at Somerset Pharmaceuticals, the developer of Emsam.[65][206]
Generic forms
[edit]Generic forms of oral selegiline are available in the United States.[55] However, generic forms of the orally disintegrating tablet and the transdermal patch are not available in this country.[55][56] The latter formulations of selegiline are very expensive, and this can be prohibitive to their use.[56][207] There has been poor insurance coverage of the transdermal patch form for depression, with insurance companies often requiring patients to first fail to respond to one or two other antidepressants and to be responsible for larger copayments.[56] It is expected that generics of the transdermal patch will become available at some point in the future.[56]
Availability
[edit]Conventional oral selegiline (brand names Eldepryl, Jumex) is widely marketed throughout the world, including in over 70 countries.[3][158][23][205] Conversely, the selegiline transdermal patch (brand name Emsam) is only marketed in the United States, while the selegiline orally disintegrating tablet (brand name Zelapar) is marketed in the United States, the United Kingdom, and the European Union.[3][45][23]
Notable users
[edit]József Knoll, one of the developers of selegiline, began taking a low 1 mg daily dose of selegiline on January 1, 1989 at the age of 64.[144]: 92 [175] He reported in 2012 that this had continued for 22 years uninterrupted.[144]: 92 Knoll stated that he had become so fascinated with the possible longevity-promoting effects of selegiline that he had decided to start taking it as a self-experiment.[144]: 92 [175] Knoll later died in 2018 at the age of 93.[208]
David Pearce, a British transhumanist philosopher, wrote his self-published book-length internet manifesto The Hedonistic Imperative[209] six weeks after starting to take selegiline.[210]
Sam Bankman-Fried, the founder and former CEO of the FTX cryptocurrency exchange, is known to have used selegiline for depression in the form of the Emsam patch for at least 5 to 10 years.[211][212] He is also known to have simultaneously taken Adderall for treatment of attention deficit hyperactivity disorder (ADHD)[211][212] and to have possessed non-pharmaceutical adrafinil, a prodrug of modafinil.[213]
Fictional representations
[edit]In Gregg Hurwitz's novel Out of the Dark, selegiline (Emsam) and tyramine-containing food were used to assassinate the president of the United States.[214]
Internet vendors
[edit]Selegiline in non-pharmaceutical form is sold on the Internet without a prescription by online vendors for uses such as purported cognitive enhancement (i.e., as a so-called "smart drug" or nootropic) and anti-aging effects.[215][150][216] It is widely available for such purposes, for instance under informal brand names like Dep-Pro, Selepryl, and Cyprenil, which are oral liquid solutions of selegiline at a concentration of 1 mg per drop.[150][216][144]: 86
Presence in ecstasy
[edit]In his 1993 book E for Ecstasy examining the uses of the street drug ecstasy in the United Kingdom, the writer, activist, and ecstasy advocate Nicholas Saunders highlighted test results showing that certain consignments of the drug also contained selegiline.[217] Consignments of ecstasy known as "Strawberry" contained what Saunders described as a "potentially dangerous combination of ketamine, ephedrine and selegiline," as did a consignment of "Sitting Duck" Ecstasy tablets.[218]
Doping in sport
[edit]Selegline is on the World Anti-Doping Agency (WADA)'s list of prohibited substances.[219] It is classified as a "stimulant" in this list, along with various amphetamines, methylphenidate, adrenergic sympathomimetics, modafinil, and other agents.[219] A review of the pharmacology of WADA prohibited substances noted that although selegiline is classified as a stimulant in the WADA prohibited substances list and stimulants can enhance physical performance, selegiline was seemingly included in the list not because of any short-term stimulant effects of its own, but rather because it metabolizes into small amounts of levomethamphetamine and levoamphetamine and can produce false positives for amphetamines on drug tests.[219] In any case, levomethamphetamine and levoamphetamine are catecholamine releasing agents and can produce sympathomimetic and psychostimulant effects with sufficiently high exposure.[220][221][222] Such actions may have performance-enhancing effects.[219]
Regulatory status
[edit]Selegiline is a prescription drug.[4][8][6] It is not specifically a controlled substance in the United States and hence is not an illegal drug.[8] However, deprenyl and selegiline are controlled substances in Japan.[223][33] They are classified as "Stimulants", alongside a variety of other amphetamines, under Article 2 of Japan's Narcotics and Psychotropics Control Law.[33] Selegiline is known to metabolize into small amounts of levoamphetamine and levomethamphetamine but is thought to have little to no misuse potential or dependence liability.[29][30][31][32][92][8]
Non-medical use
[edit]Anti-aging and longevity
[edit]József Knoll and his team are credited with having developed selegiline. Although selegiline's development as a potential treatment for Parkinson's disease, Alzheimer's disease, and depression was headed by other teams, Knoll remained at the forefront of research into the potential longevity enhancing effects of selegiline up until his death in 2018.[208][224][225] Knoll published his 2012 book How Selegiline ((–)-Deprenyl) Slows Brain Aging wherein he claims that:[144]: 90
"In humans, maintenance from sexual maturity on (–)-deprenyl (1mg daily) is, for the time being, the most promising prophylactic treatment to fight against the age related decay of behavioral performances, prolonging life, and preventing or delaying the onset of age-related neurodegenerative diseases such as Parkinson's and Alzheimer's".
The mechanism of selegiline's longevity-promoting effect has been researched by several groups, including Knoll and his associates at Semmelweis University, Budapest.[23] The drug has been determined to be a catecholaminergic activity enhancer when present in minuscule concentrations far below those at which monoamine oxidase inhibitory activity can be observed, thereby potentiating the release of catecholamine neurotransmitters in response to stimuli. Knoll maintains that micro-doses of selegiline act as a synthetic analogue to a known or unknown trace amine in order to preserve the brain catecholaminergic system, which he perceives as integral to the organism's ability to function in an adaptive, goal-directed and motivated manner during advancing physical age:[144]: 70, 43
"[...] enhancer regulation in the catecholaminergic brain stem neurons play[s] a key role in controlling the uphill period of life and the transition from adolescence to adulthood. The results of our longevity studies support the hypothesis that quality and duration of life rests upon the inborn efficiency of the catecholaminergic brain machinery, i.e. a high performing, long-living individual has a more active, more slowly deteriorating catecholaminergic system than its low performing, shorter living peer. Thus, a better brain engine allows for a better performance and a longer lifespan."
"Since the catecholaminergic and serotonergic neurons in the brain stem are of key importance in ensuring that the mammalian organism works as a purposeful, motivated, goal-directed entity, it is hard to overestimate the significance of finding safe and efficient means to slow the decay of these systems with passing time. The conclusion that the maintenance on (–)-deprenyl that keeps the catecholaminergic neurons on a higher activity level is a safe and efficient anti-aging therapy follows from the discovery of the enhancer regulation in the catecholaminergic neurons of the brain stem. From the finding that this regulation starts working on a high activity level after weaning and the enhanced activity subsists during the uphill period of life, until sexual hormones dampen the enhancer regulation in the catecholaminergic and serotonergic neurons in the brain stem, and this event signifies the transition from developmental longevity into postdevelopmental longevity, the downhill period of life."
Despite findings by Knoll that selegiline can prolong lifespan in rodents by 35% however, other studies have had conflicting findings and have even found increased mortality with selegiline in rodents.[54] In humans with Parkinson's disease, selegiline has been associated with cardiovascular and psychiatric complications and has not been found to reduce mortality in long-term studies.[54] As such, the claimed anti-aging and longevity benefits of selegiline have yet to be substantiated in humans and are controversial and uncertain.[54][53]
Nootropic or "smart drug"
[edit]Selegiline is considered by some to be a nootropic, otherwise known as a cognitive enhancer or "smart drug", both at clinical and sub-clinical dosages, and has been used off-label and non-medically to improve cognitive performance.[49][226] It is one of the most popular such agents.[49] Selegiline has been found to have neuroprotective activity against certain neurotoxins and to increase the production of several brain growth factors, such as nerve growth factor (NGF), brain-derived neurotrophic factor (BDNF), and glial cell line-derived neurotrophic factor (GDNF).[23] The drug has also been found in animal models to improve learning ability and to help preserve it during ischemia and aging.[227][228][229][230] Despite claims that selegiline and other claimed nootropics have cogintive-enhancing effects however, these effects are controversial and their benefits versus risks are uncertain.[49]
Research
[edit]Depression
[edit]Selegiline has been clinically studied in combination with oral L-phenylalanine or β-phenethylamine in the treatment of depression and was reported to be effective.[39][231][184][232][233] L-Phenylalanine is known to be metabolized into β-phenethylamine, selegiline is known to strongly inhibit the metabolism of β-phenethylamine, and β-phenethylamine has been implicated in having psychostimulant-like mood-lifting effects.[39][17][231]
Social anxiety
[edit]A small clinical study found that oral selegiline (10 mg/day) reduced symptoms of social anxiety disorder.[12][24][234] The effectiveness was modest, with a reduction in social anxiety scores from baseline of 32% over 6 weeks of treatment.[12][24][234] It was seemingly less effective than certain other agents used in the treatment of social anxiety, such as the non-selective MAOI phenelzine (45% symptom reduction) and the benzodiazepine clonazepam (51% symptom reduction), though it was similar to the SSRI sertraline (32% symptom decrease).[234]
ADHD
[edit]Selegiline has been limitedly studied in the treatment of attention deficit hyperactivity disorder (ADHD) in children, adolescents, and adults.[24][235][236][237] In a small randomized trial of selegiline for treatment of ADHD in children, there were improvements in attention, hyperactivity, and learning/memory performance but not in impulsivity.[238] A small clinical randomized trial compared selegiline to methylphenidate, a first line treatment for ADHD, and reported equivalent efficacy as assessed by parent and teacher ratings.[239] In another small randomized controlled trial of selegiline for the treatment of adult ADHD, a high dose of the medication for 6 weeks was not significantly more effective than placebo in improving symptoms.[236][240][241] Selegiline in its transdermal patch form (brand name Emsam) has also been assessed in the treatment of ADHD in children and adolescents in a small open-label pilot study sponsored by the manufacturer in 2003.[12][242] However, there was a high rate of discontinuation and development was not further pursued.[12][242]
Motivational disorders
[edit]Selegiline has been found to produce pro-motivational effects and to reverse motivational deficits in rodents.[243][244][245][246] In case reports and small clinical studies, selegiline has been reported to improve disorders of diminished motivation like apathy and abulia due to conditions such as traumatic brain injury.[243][247][248][249][250][251] In accordance with the preceding findings, selegiline, along with other dopaminergic and activating agents, may be useful in the treatment of disorders of diminished motivation, including apathy, abulia, and akinetic mutism.[244][252][248]
Addiction
[edit]Selegiline has been evaluated for smoking cessation both as a monotherapy and in combination with nicotine replacement therapy in five clinical studies.[253][254][24] However, it is limitedly or not effective for this use.[253][254][24] It was also evaluated for treatment of cocaine dependence in one study, but was similarly not effective.[255] Studies are mixed on whether selegiline, at MAO-B-selective doses, reduces the effects of cocaine in humans.[105][106][107][108][109][110] Selegiline, also at an MAO-B-selective dosage, did not modify or potentiate the pharmacological effects of intravenous methamphetamine in a small clinical study.[103][104]
Sexual dysfunction
[edit]Selegiline has been assessed for treatment of sexual dysfunction induced by antipsychotics in people with schizophrenia, but was not effective in a single small clinical study.[256][257] It also did not improve sexual function in men with depression, but did improve several domains of sexual function in women with depression.[67]
Psychosis
[edit]Selegiline has been studied as an adjunct to antipsychotics in the treatment of schizophrenia in four clinical studies.[24][258] However, it failed to significantly reduce positive or negative symptoms of schizophrenia in meta-analyses of these studies.[24][258]
Excessive sleepiness
[edit]Selegiline has been evaluated for the treatment of narcolepsy in three small clinical studies.[259][260][261] It was found to be effective in these studies.[259][260] A dosage of 10 mg/day had no effect on symptoms, but 20 to 30 mg/day improved alertness, mood, and somewhat reduced cataplexy, clinical effects that have been described as comparable to the same dosages of amphetamine.[260] Animal research indicates that the beneficial effects of high doses of selegiline in narcolepsy are likely due to conversion into its active metabolites, levoamphetamine and levomethamphetamine.[260][261] Selegiline has also been evaluated for treatment of hypersomnia (excessive sleeping or sleepiness) in people with myotonic dystrophy, but was not effective in a single small clinical study.[262][259]
Periodic limb movement disorder
[edit]Selegiline has been studied in the treatment of periodic limb movement disorder (PLMD) in a single small open-label clinical study.[263][264][265] It was reported to be effective as assessed by polysomnography, reducing periodic limb movements during sleep by about 60%.[263][265] Selegiline has not been studied for the related condition restless legs syndrome (RLS) as of 2023.[263][264] The drug has not been studied well enough in PLMD or RLS to be widely used in their treatment.[263]
Tardive dyskinedia
[edit]Selegiline was studied in the treatment of antipsychotic-induced tardive dyskinesia in one small clinical study, but was ineffective.[266]
Dementia and stroke
[edit]Selegiline has also been used off-label as a palliative treatment for dementia in Alzheimer's disease.[58] However, its clinical effectiveness is limited or lacking for this use.[267][268][269][270] It was also ineffective in the treatment of Lewy body dementia.[271] Selegiline has been used to support motor rehabilitation in stroke recovery, but evidence for this use is inadequate and no recommendation can be made for or against it.[272]
Disorders of consciousness
[edit]Selegiline has been studied in patients with disorders of consciousness, such as minimally conscious state, persistent vegetative state, and persistent coma, in a small open-label clinical study.[273][274] It was found to be effective in enhancing arousal and promoting recovery of consciousness in some of these individuals.[273][274]
Neurotoxicity
[edit]Selegiline has been reported to protect against the damage caused by the potent dopaminergic and/or noradrenergic neurotoxins 6-hydroxydopamine (6-OHDA), N-(2-chloroethyl)-N-ethyl-2-bromobenzylamine (DSP-4), and 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) in animals.[275][17][276][277][278][279] Conversely, selegiline is ineffective in protecting against the serotonergic and noradrenergic neurotoxin 5,7-dihydroxytryptamine (5,7-DHT).[17][280]
Selegiline has also been reported to protect against methylenedioxymethamphetamine (MDMA)-induced serotonergic neurotoxicity in rodents.[281][282][283][284][285] The serotonergic neurotoxicity of MDMA appears to be dependent on release of dopamine and its subsequent metabolism by MAO-B within serotonergic neurons into hydroxyl radicals, which is blocked by MAO-B inhibition.[281][282] Likewise, selegiline prevented the serotonergic neurotoxicity of a combination of methylenedioxyaminoindane (MDAI) and dextroamphetamine.[286][287]
Conversely, selegiline failed to reduce the serotonergic neurotoxicity caused by fenfluramine and either did not affect or potentiated the serotonergic neurotoxicity caused by para-chloroamphetamine (PCA).[277][288][289][290] In addition, findings are mixed and conflicting on whether selegiline prevents amphetamine- and methamphetamine-induced dopaminergic neurotoxicity in rodents.[291][292][293][294]
Although MAO-B-selective doses of selegiline protect against MDMA-induced serotonergic neurotoxicity in rodents, combination of amphetamines like MDMA with MAOIs, including selegiline, can produce serious complications, including serotonin syndrome, hypertensive crisis, and death.[295][296]
Other formulations
[edit]The original oral formulation of selegiline was developed for the treatment of depression.[50] However, it ended up being developed and approved for the treatment of Parkinson's disease instead.[50][45][4] In any case, oral selegiline has been widely used off-label to treat depression.[24] The transdermal patch form of selegiline was developed and approved specifically for the treatment of depression.[297][12][9][8] It was also under development for the treatment of Alzheimer's disease, attention deficit hyperactivity disorder (ADHD), cognition disorders, and Parkinson's disease, but development for these indications was discontinued.[297] The ODT form of selegiline was developed and licensed exclusively for the treatment of Parkinson's disease.[298][7][6]
Veterinary use
[edit]In veterinary medicine, selegiline is sold under the brand name Anipryl and is manufactured by Zoetis.[299] It is available in the form of 2, 5, 10, 15, and 30 mg oral tablets for use in animals.[299] Selegiline is used in dogs to treat canine cognitive dysfunction (CCD) and, at higher doses, to treat pituitary-dependent hyperadrenocorticism (PDH).[300][301]
CCD is a form of dementia that mimics Alzheimer's disease in humans.[302] Geriatric dogs treated with selegiline show improvements in sleeping pattern, reduced urinary incontinence, and increased activity level, with most showing improvements by one month of treatment.[303][304] Though it is labeled for use in dogs only, selegiline has been used off-label for geriatric cats with cognitive dysfunction.[305]
PDH is a hormonal disorder and is analogous to pituitary-dependent Cushing's syndrome in humans.[299] Selegiline's effectiveness in treating PDH has been disputed.[300] Theoretically, it works by increasing dopamine levels, which downregulates the secretion of adrenocorticotropic hormone (ACTH) from the brain, eventually leading to reduced levels of cortisol.[305] Some claim that selegiline is only effective at treating PDH caused by lesions in the anterior pituitary (which comprise most canine cases).[306] The greatest sign of improvement is lessening of PDH-related abdominal distention.[303]
Side effects in dogs are uncommon, but they include vomiting, diarrhea, diminished hearing, salivation, decreased weight, and behavioral changes such as hyperactivity, listlessness, disorientation, and repetitive motions.[301][306]
Selegiline has been limitedly studied in large animals like horses and its dosage in these animals has not been established.[306] In preliminary research, a dose of selegiline of 30 mg orally or intravenously in horses had no observable effects on behavior or locomotor activity.[306]
The doses of selegiline used in animals are described as extremely high relative to those used in humans (which are ~0.1 mg/kg body weight).[152]
References
[edit]- ^ a b "Drug Treatments for Parkinson's" (PDF). Retrieved July 5, 2024.
Selegiline (seh-LEH-ji-leen)
- ^ a b Acosta WR (2020). Pharmacology for Health Professionals. Jones & Bartlett Learning. p. 66. ISBN 978-1-284-24083-2. Retrieved July 5, 2024.
sell-eh'-geh-leen
- ^ a b c d e f g "Selegiline". Drugs.com. Archived from the original on July 3, 2024. Retrieved February 7, 2016.
- ^ a b c d e f g h i j k l m n o p q r s t u v w x y z aa "ELDEPRYL® (Selegiline Hydrochloride) Tablets, USP Label" (PDF). Food and Drug Administration. January 2008. Retrieved July 3, 2024.
- ^ a b c d e f g h i j k l m n o p q r s t u v w x y z aa ab ac ad ae Mahmood I (August 1997). "Clinical pharmacokinetics and pharmacodynamics of selegiline. An update". Clin Pharmacokinet. 33 (2): 91–102. doi:10.2165/00003088-199733020-00002. PMID 9260033.
- ^ a b c d e f g h i j k l m n o p q r s t u v w x y z aa ab ac "ZELAPAR® (Selegiline Hydrochloride) Orally Disintegrating Tablets" (PDF). Food and Drug Administration. July 2021. Retrieved July 3, 2024.
- ^ a b c d e f g h i j k l m n o p q r s t u Poston KL, Waters C (October 2007). "Zydis selegiline in the management of Parkinson's disease". Expert Opin Pharmacother. 8 (15): 2615–2624. doi:10.1517/14656566.8.15.2615. PMID 17931095.
- ^ a b c d e f g h i j k l m n o p q r s t u v w x y z aa ab ac ad ae af ag ah ai aj ak al am "EMSAM® (Selegiline Transdermal System) Label" (PDF). Food and Drug Administration. July 2017. Retrieved July 2, 2024.
- ^ a b c d e f g h i j k l m n o p q r s t u v w x y z aa ab Lee KC, Chen JJ (November 2007). "Transdermal selegiline for the treatment of major depressive disorder". Neuropsychiatric Disease and Treatment. 3 (5): 527–537. doi:10.2147/ndt.s12160200 (inactive November 1, 2024). PMC 2656289. PMID 19300583.
{{cite journal}}: CS1 maint: DOI inactive as of November 2024 (link) - ^ Anvisa (March 31, 2023). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published April 4, 2023). Archived from the original on August 3, 2023. Retrieved August 16, 2023.
- ^ a b c d e f g h i j k l m n o p q r s t u v w Magyar K (2011). "The Pharmacology of Selegiline". In Youdim M, Riederer P (eds.). Monoamine Oxidases and Their Inhibitors. International Review of Neurobiology. Vol. 100. Academic Press. pp. 65–84. doi:10.1016/B978-0-12-386467-3.00004-2. ISBN 978-0-12-386467-3. PMID 21971003.
- ^ a b c d e f g h i j k l m n Pae CU, Lim HK, Han C, Neena A, Lee C, Patkar AA (August 2007). "Selegiline transdermal system: current awareness and promise". Prog Neuropsychopharmacol Biol Psychiatry. 31 (6): 1153–1163. doi:10.1016/j.pnpbp.2007.04.020. PMID 17614182.
- ^ a b c d Löhle M, Storch A (November 2008). "Orally disintegrating selegiline for the treatment of Parkinson's disease". Expert Opin Pharmacother. 9 (16): 2881–2891. doi:10.1517/14656566.9.16.2881. PMID 18937619.
- ^ a b Clarke A, Brewer F, Johnson ES, Mallard N, Hartig F, Taylor S, et al. (November 2003). "A new formulation of selegiline: improved bioavailability and selectivity for MAO-B inhibition". Journal of Neural Transmission. 110 (11): 1241–1255. doi:10.1007/s00702-003-0036-4. PMID 14628189. S2CID 711419.
- ^ a b c d e f g h i j k l m n o p Knoll J (1983). "Deprenyl (selegiline): the history of its development and pharmacological action". Acta Neurol Scand Suppl. 95: 57–80. doi:10.1111/j.1600-0404.1983.tb01517.x. PMID 6428148.
- ^ Knoll J (1986). "Role of B-Type Monoamine Oxidase Inhibition in the Treatment of Parkinson's Disease". Movement Disorders. Boston, MA: Springer US. pp. 53–81. doi:10.1007/978-1-4684-5038-5_3. ISBN 978-1-4684-5040-8.
- ^ a b c d e f g h i j k l m n o Heinonen EH, Lammintausta R (1991). "A review of the pharmacology of selegiline". Acta Neurologica Scandinavica. Supplementum. 136: 44–59. doi:10.1111/j.1600-0404.1991.tb05020.x. PMID 1686954.
- ^ a b c Heinonen EH, Anttila MI, Lammintausta RA (December 1994). "Pharmacokinetic aspects of l-deprenyl (selegiline) and its metabolites". Clin Pharmacol Ther. 56 (6 Pt 2): 742–749. doi:10.1038/clpt.1994.204. PMID 7995016.
- ^ a b Heinonen EH, Myllylä V, Sotaniemi K, Lamintausta R, Salonen JS, Anttila M, et al. (November 1989). "Pharmacokinetics and metabolism of selegiline". Acta Neurologica Scandinavica. Supplementum. 126: 93–99. doi:10.1111/j.1600-0404.1989.tb01788.x. PMID 2515726. S2CID 221440315.
- ^ a b Chrisp P, Mammen GJ, Sorkin EM (May 1991). "Selegiline: A Review of its Pharmacology, Symptomatic Benefits and Protective Potential in Parkinson's Disease". Drugs Aging. 1 (3): 228–248. doi:10.2165/00002512-199101030-00006. PMID 1794016.
- ^ a b c d e f g h i j k l Rodrigues AD (June 2022). "Drug Interactions Involving 17α-Ethinylestradiol: Considerations Beyond Cytochrome P450 3A Induction and Inhibition". Clin Pharmacol Ther. 111 (6): 1212–1221. doi:10.1002/cpt.2383. PMID 34342002.
- ^ a b c Hidestrand M, Oscarson M, Salonen JS, Nyman L, Pelkonen O, Turpeinen M, et al. (November 2001). "CYP2B6 and CYP2C19 as the major enzymes responsible for the metabolism of selegiline, a drug used in the treatment of Parkinson's disease, as revealed from experiments with recombinant enzymes". Drug Metab Dispos. 29 (11): 1480–1484. PMID 11602525.
- ^ a b c d e f g h i j k l m n o p q Miklya I (November 2016). "The significance of selegiline/(-)-deprenyl after 50 years in research and therapy (1965-2015)". Molecular Psychiatry. 21 (11): 1499–1503. doi:10.1038/mp.2016.127. PMID 27480491.
- ^ a b c d e f g h i j k l m n o p q r s t u v w x y Rossano F, Caiazza C, Sobrino A, Solini N, Vellucci A, Zotti N, et al. (July 2023). "Efficacy and safety of selegiline across different psychiatric disorders: A systematic review and meta-analysis of oral and transdermal formulations". Eur Neuropsychopharmacol. 72: 60–78. doi:10.1016/j.euroneuro.2023.03.012. hdl:2066/293870. PMID 37087864.
- ^ a b c d e f g h i j k l Citrome L, Goldberg JF, Portland KB (November 2013). "Placing transdermal selegiline for major depressive disorder into clinical context: number needed to treat, number needed to harm, and likelihood to be helped or harmed". Journal of Affective Disorders. 151 (2): 409–417. doi:10.1016/j.jad.2013.06.027. PMID 23890583.
- ^ a b Cipriani A, Furukawa TA, Salanti G, Chaimani A, Atkinson LZ, Ogawa Y, et al. (April 2018). "Comparative efficacy and acceptability of 21 antidepressant drugs for the acute treatment of adults with major depressive disorder: a systematic review and network meta-analysis". Lancet. 391 (10128): 1357–1366. doi:10.1016/S0140-6736(17)32802-7. PMC 5889788. PMID 29477251.
- ^ a b c d e f g Robinson DS, Amsterdam JD (January 2008). "The selegiline transdermal system in major depressive disorder: a systematic review of safety and tolerability". J Affect Disord. 105 (1–3): 15–23. doi:10.1016/j.jad.2007.04.024. PMID 17568687.
- ^ a b c d e f g h i j k l m n o Gerlach M, Youdim MB, Riederer P (December 1996). "Pharmacology of selegiline". Neurology. 47 (6 Suppl 3): S137–S145. doi:10.1212/wnl.47.6_suppl_3.137s. PMID 8959982.
- ^ a b c Finberg JP, Rabey JM (2016). "Inhibitors of MAO-A and MAO-B in Psychiatry and Neurology". Front Pharmacol. 7: 340. doi:10.3389/fphar.2016.00340. PMC 5067815. PMID 27803666.
- ^ a b c d e f g Fabbrini G, Abbruzzese G, Marconi S, Zappia M (2012). "Selegiline: a reappraisal of its role in Parkinson disease". Clin Neuropharmacol. 35 (3): 134–140. doi:10.1097/WNF.0b013e318255838b. PMID 22592509.
- ^ a b c d e f g h i j k Yasar S, Goldberg JP, Goldberg SR (January 1, 1996). "Are metabolites of l-deprenyl (Selegiline) useful or harmful? Indications from preclinical research". Deprenyl — Past and Future. Journal of Neural Transmission. Supplementum. Vol. 48. pp. 61–73. doi:10.1007/978-3-7091-7494-4_6. ISBN 978-3-211-82891-5. PMID 8988462.
- ^ a b c d Nickel B, Szelenyi I, Schulze G (December 1994). "Evaluation of physical dependence liability of l-deprenyl (selegiline) in animals". Clin Pharmacol Ther. 56 (6 Pt 2): 757–767. doi:10.1038/clpt.1994.206. PMID 7995018.
- ^ a b c "KEGG DRUG: Narcotics and Psychotropics in Japan". KEGG. April 26, 2024. Retrieved July 10, 2024.
- ^ a b Knoll J (1997). "Istoriia deprenil--pervogo selektivnogo ingibitora monoaminoksidazy tipa B" [History of deprenyl--the first selective inhibitor of monoamine oxidase type B]. Voprosy Meditsinskoi Khimii. 43 (6): 482–493. PMID 9503565.
- ^ a b c d e f g Knoll J (February 1998). "(-)Deprenyl (selegiline), a catecholaminergic activity enhancer (CAE) substance acting in the brain". Pharmacol Toxicol. 82 (2): 57–66. doi:10.1111/j.1600-0773.1998.tb01399.x. PMID 9498233.
- ^ a b c d e Miklya I (March 13, 2014). "The History of Selegiline/(-)-Deprenyl the First Selective Inhibitor of B-Type Monoamine Oxidase and The First Synthetic Catecholaminergic Activity Enhancer Substance". International Network for the History of Neuropsychopharmacology. Archived from the original on February 7, 2016. Retrieved January 7, 2016.
- ^ a b c d e f Gaszner P, Miklya I (January 2006). "Major depression and the synthetic enhancer substances, (-)-deprenyl and R-(-)-1-(benzofuran-2-yl)-2-propylaminopentane". Prog Neuropsychopharmacol Biol Psychiatry. 30 (1): 5–14. doi:10.1016/j.pnpbp.2005.06.004. PMID 16023777.
- ^ a b c d e f Harsing LG, Timar J, Miklya I (August 2023). "Striking Neurochemical and Behavioral Differences in the Mode of Action of Selegiline and Rasagiline". Int J Mol Sci. 24 (17): 13334. doi:10.3390/ijms241713334. PMC 10487936. PMID 37686140.
- ^ a b c d e f g Shimazu S, Miklya I (May 2004). "Pharmacological studies with endogenous enhancer substances: β-phenylethylamine, tryptamine, and their synthetic derivatives". Prog Neuropsychopharmacol Biol Psychiatry. 28 (3): 421–427. doi:10.1016/j.pnpbp.2003.11.016. PMID 15093948.
- ^ a b c Berry MD (January 2007). "The potential of trace amines and their receptors for treating neurological and psychiatric diseases". Rev Recent Clin Trials. 2 (1): 3–19. doi:10.2174/157488707779318107. PMID 18473983.
- ^ a b c d e f g h i j Gerlach M, Reichmann H, Riederer P (2012). "A critical review of evidence for preclinical differences between rasagiline and selegiline". Basal Ganglia. 2 (4): S9–S15. doi:10.1016/j.baga.2012.04.032.
- ^ a b c Rothman RB, Baumann MH (October 2003). "Monoamine transporters and psychostimulant drugs". Eur J Pharmacol. 479 (1–3): 23–40. doi:10.1016/j.ejphar.2003.08.054. PMID 14612135.
- ^ a b c d e Kraemer T, Maurer HH (April 2002). "Toxicokinetics of amphetamines: metabolism and toxicokinetic data of designer drugs, amphetamine, methamphetamine, and their N-alkyl derivatives". Ther Drug Monit. 24 (2): 277–289. doi:10.1097/00007691-200204000-00009. PMID 11897973.
- ^ a b c d e Parnham MJ (1993). "The History of l-Deprenyl". Inhibitors of Monoamine Oxidase B: Pharmacology and Clinical Use in Neurodegenerative Disorders. Milestones in Drug Therapy. Basel: Birkhäuser Basel. pp. 237–251. doi:10.1007/978-3-0348-6348-3_12. ISBN 978-3-0348-6349-0.
- ^ a b c d e f g h i j Tábi T, Vécsei L, Youdim MB, Riederer P, Szökő É (May 2020). "Selegiline: a molecule with innovative potential". J Neural Transm (Vienna). 127 (5): 831–842. doi:10.1007/s00702-019-02082-0. PMC 7242272. PMID 31562557.
- ^ a b c d Seaman J, Landry JT (2011). Mylan: 50 Years of Unconventional Success: Making Quality Medicine Affordable and Accessible. University Press of New England. p. 50. ISBN 978-1-61168-269-4.
- ^ a b c d e f g h Hoffman GR, Olson MG, Schoffstall AM, Estévez RF, Van den Eynde V, Gillman PK, et al. (December 2023). "Classics in Chemical Neuroscience: Selegiline, Isocarboxazid, Phenelzine, and Tranylcypromine". ACS Chem Neurosci. 14 (23): 4064–4075. doi:10.1021/acschemneuro.3c00591. PMID 37966854.
- ^ a b Golbe LI (October 1988). "Deprenyl as symptomatic therapy in Parkinson's disease". Clin Neuropharmacol. 11 (5): 387–400. doi:10.1097/00002826-198810000-00001. PMID 3146432.
- ^ a b c d e Schifano F, Catalani V, Sharif S, Napoletano F, Corkery JM, Arillotta D, et al. (April 2022). "Benefits and Harms of 'Smart Drugs' (Nootropics) in Healthy Individuals". Drugs. 82 (6): 633–647. doi:10.1007/s40265-022-01701-7. PMID 35366192.
- ^ a b c d e f g h i j Knoll J (2001). "Antiaging compounds: (-)deprenyl (selegeline) and (-)1-(benzofuran-2-yl)-2-propylaminopentane, [(-)BPAP], a selective highly potent enhancer of the impulse propagation mediated release of catecholamine and serotonin in the brain". CNS Drug Rev. 7 (3): 317–345. doi:10.1111/j.1527-3458.2001.tb00202.x. PMC 6494119. PMID 11607046.
- ^ Schneider LS, Tariot PN, Goldstein B (December 1994). "Therapy with l-deprenyl (selegiline) and relation to abuse liability". Clin Pharmacol Ther. 56 (6 Pt 2): 750–756. doi:10.1038/clpt.1994.205. PMID 7995017.
- ^ Blazer DG, Yaffe K, Liverman CT (July 21, 2015). Risk and Protective Factors and Interventions: General Cognitive Aging Interventions and Next Steps. National Academies Press (US). Retrieved July 5, 2024.
- ^ a b Brown RP, Gerbarg PL (2008). Muskin PR (ed.). "Integrative Psychopharmacology: A Practical Approach to Herbs and Nutrients in Psychiatry". Review of Psychiatry. Complementary and Alternative Medicine and Psychiatry. 19 (1). American Psychiatric Publishing: 1–66 (39). ISBN 978-1-58562-827-8. Retrieved July 5, 2024.
- ^ a b c d e Finberg JP (April 2019). "Inhibitors of MAO-B and COMT: their effects on brain dopamine levels and uses in Parkinson's disease". Journal of Neural Transmission. 126 (4): 433–448. doi:10.1007/s00702-018-1952-7. PMID 30386930.
- ^ a b c d e "Drugs@FDA: FDA-Approved Drugs". accessdata.fda.gov. Retrieved July 1, 2024.
- ^ a b c d e f g h i j k l m Asnis GM, Henderson MA (2014). "EMSAM (deprenyl patch): how a promising antidepressant was underutilized". Neuropsychiatr Dis Treat. 10: 1911–1923. doi:10.2147/NDT.S59107. PMC 4200016. PMID 25336957.
- ^ Riederer P, Lachenmayer L, Laux G (August 2004). "Clinical applications of MAO-inhibitors". Current Medicinal Chemistry. 11 (15): 2033–2043. doi:10.2174/0929867043364775 (inactive November 2, 2024). PMID 15279566.
{{cite journal}}: CS1 maint: DOI inactive as of November 2024 (link) - ^ a b c "Selegiline Hydrochloride Monograph for Professionals". Drugs.com. Retrieved February 23, 2018.
- ^ a b Ives NJ, Stowe RL, Marro J, Counsell C, Macleod A, Clarke CE, et al. (September 2004). "Monoamine oxidase type B inhibitors in early Parkinson's disease: meta-analysis of 17 randomised trials involving 3525 patients". BMJ. 329 (7466): 593. doi:10.1136/bmj.38184.606169.AE. PMC 516655. PMID 15310558.
- ^ Riederer P, Lachenmayer L (November 2003). "Selegiline's neuroprotective capacity revisited". Journal of Neural Transmission. 110 (11): 1273–1278. doi:10.1007/s00702-003-0083-x. PMID 14628191. S2CID 20232921.
- ^ Frisina PG, Tenenbaum HR, Borod JC, Foldi NS (May 2008). "The effects of antidepressants in Parkinson's disease: a meta-analysis". Int J Neurosci. 118 (5): 667–682. doi:10.1080/00207450701239418. PMID 18446583.
- ^ Tsuboi T, Satake Y, Hiraga K, Yokoi K, Hattori M, Suzuki M, et al. (June 2022). "Effects of MAO-B inhibitors on non-motor symptoms and quality of life in Parkinson's disease: A systematic review". npj Parkinsons Dis. 8 (1): 75. doi:10.1038/s41531-022-00339-2. PMC 9192747. PMID 35697709.
- ^ a b c Binde CD, Tvete IF, Gåsemyr J, Natvig B, Klemp M (September 2018). "A multiple treatment comparison meta-analysis of monoamine oxidase type B inhibitors for Parkinson's disease". Br J Clin Pharmacol. 84 (9): 1917–1927. doi:10.1111/bcp.13651. PMC 6089809. PMID 29847694.
- ^ Hengartner MP, Jakobsen JC, Sørensen A, Plöderl M (2020). "Efficacy of new-generation antidepressants assessed with the Montgomery-Asberg Depression Rating Scale, the gold standard clinician rating scale: A meta-analysis of randomised placebo-controlled trials". PLOS ONE. 15 (2): e0229381. Bibcode:2020PLoSO..1529381H. doi:10.1371/journal.pone.0229381. PMC 7043778. PMID 32101579.
- ^ a b c d e f g h i j k l m n o p q r Cristancho MA, Thase ME (2016). "Critical appraisal of selegiline transdermal system for major depressive disorder". Expert Opinion on Drug Delivery. 13 (5): 659–665. doi:10.1517/17425247.2016.1140145. PMID 26837935.
- ^ Pae CU, Patkar AA, Jang S, Portland KB, Jung S, Nelson JC (August 2014). "Efficacy and safety of selegiline transdermal system (STS) for the atypical subtype of major depressive disorder: pooled analysis of 5 short-term, placebo-controlled trials". CNS Spectr. 19 (4): 324–329. doi:10.1017/S1092852913000655. PMID 24168807.
- ^ a b c Clayton AH, Campbell BJ, Favit A, Yang Y, Moonsammy G, Piontek CM, et al. (December 2007). "Symptoms of sexual dysfunction in patients treated for major depressive disorder: a meta-analysis comparing selegiline transdermal system and placebo using a patient-rated scale". J Clin Psychiatry. 68 (12): 1860–1866. doi:10.4088/jcp.v68n1205. PMID 18162016.
- ^ Winter J, Curtis K, Hu B, Clayton AH (July 2022). "Sexual dysfunction with major depressive disorder and antidepressant treatments: impact, assessment, and management". Expert Opin Drug Saf. 21 (7): 913–930. doi:10.1080/14740338.2022.2049753. PMID 35255754.
- ^ Alborghetti M, Nicoletti F (2019). "Different Generations of Type-B Monoamine Oxidase Inhibitors in Parkinson's Disease: From Bench to Bedside". Curr Neuropharmacol. 17 (9): 861–873. doi:10.2174/1570159X16666180830100754. PMC 7052841. PMID 30160213.
- ^ Friedman RA, Leon AC (June 2007). "Expanding the black box - depression, antidepressants, and the risk of suicide". The New England Journal of Medicine. 356 (23): 2343–2346. doi:10.1056/NEJMp078015. PMID 17485726.
- ^ a b c d Patkar AA, Pae CU, Masand PS (May 2006). "Transdermal selegiline: the new generation of monoamine oxidase inhibitors". CNS Spectr. 11 (5): 363–375. doi:10.1017/s1092852900014498. PMID 16641841.
- ^ Finberg JP, Gillman K (2011). Selective inhibitors of monoamine oxidase type B and the "cheese effect". International Review of Neurobiology. Vol. 100. pp. 169–190. doi:10.1016/B978-0-12-386467-3.00009-1. ISBN 978-0-12-386467-3. PMID 21971008.
- ^ Olanow CW, Myllylä VV, Sotaniemi KA, Larsen JP, Pålhagen S, Przuntek H, et al. (September 1998). "Effect of selegiline on mortality in patients with Parkinson's disease: a meta-analysis". Neurology. 51 (3): 825–830. doi:10.1212/wnl.51.3.825. PMID 9748034.
- ^ Aaltonen H, Kilkku O, Heinonen E, Mäki-Ikola O (December 1998). "Effect of adding selegiline to levodopa in early, mild Parkinson's disease. Evidence is insufficient to show that combined treatment increases mortality". BMJ. 317 (7172): 1586–1587. doi:10.1136/bmj.317.7172.1586. PMC 1114394. PMID 9890764.
- ^ Abassi ZA, Binah O, Youdim MB (October 2004). "Cardiovascular activity of rasagiline, a selective and potent inhibitor of mitochondrial monoamine oxidase B: comparison with selegiline". Br J Pharmacol. 143 (3): 371–378. doi:10.1038/sj.bjp.0705962. PMC 1575354. PMID 15339864.
- ^ a b Roy MA, Doiron M, Talon-Croteau J, Dupré N, Simard M (July 2018). "Effects of Antiparkinson Medication on Cognition in Parkinson's Disease: A Systematic Review". Can J Neurol Sci. 45 (4): 375–404. doi:10.1017/cjn.2018.21. PMID 29747716.
- ^ a b Vitale C, Amboni M, Erro R, Picillo M, Pellecchia MT, Barone P, et al. (June 2019). "Parkinson's disease management and impulse control disorders: current state and future perspectives". Expert Rev Neurother. 19 (6): 495–508. doi:10.1080/14737175.2019.1620603. PMID 31148487.
- ^ a b Djamshidian A, Cardoso F, Grosset D, Bowden-Jones H, Lees AJ (September 2011). "Pathological gambling in Parkinson's disease--a review of the literature". Mov Disord. 26 (11): 1976–1984. doi:10.1002/mds.23821. PMID 21661054.
- ^ Drapier D, Drapier S, Sauleau P, Derkinderen P, Damier P, Allain H, et al. (November 2006). "Pathological gambling secondary to dopaminergic therapy in Parkinson's disease". Psychiatry Res. 144 (2–3): 241–244. doi:10.1016/j.psychres.2006.04.017. PMID 17011634.
- ^ Solla P, Bortolato M, Cannas A, Mulas CS, Marrosu F (April 2015). "Paraphilias and paraphilic disorders in Parkinson's disease: A systematic review of the literature". Mov Disord. 30 (5): 604–613. doi:10.1002/mds.26157. PMC 4428164. PMID 25759330.
- ^ Hirao K, Kaneko Y, Hirose D, Fukasawa R, Shimizu S, Kanetaka H, et al. (September 2019). "Patient with Parkinson's disease presenting with impulse control disorders following treatment with selegiline". Int Psychogeriatr. 31 (9): 1375–1376. doi:10.1017/S1041610218001862. PMID 30520410.
- ^ Uitti RJ, Tanner CM, Rajput AH, Goetz CG, Klawans HL, Thiessen B (October 1989). "Hypersexuality with antiparkinsonian therapy". Clin Neuropharmacol. 12 (5): 375–383. doi:10.1097/00002826-198910000-00002. PMID 2575449.
- ^ Riley DE (2002). "Reversible transvestic fetishism in a man with Parkinson's disease treated with selegiline". Clin Neuropharmacol. 25 (4): 234–237. doi:10.1097/00002826-200207000-00008. PMID 12151912.
- ^ Shapiro MA, Chang YL, Munson SK, Okun MS, Fernandez HH (September 2006). "Hypersexuality and paraphilia induced by selegiline in Parkinson's disease: report of 2 cases". Parkinsonism Relat Disord. 12 (6): 392–395. doi:10.1016/j.parkreldis.2006.01.010. PMID 16730214.
- ^ Seeman P (April 2015). "Parkinson's disease treatment may cause impulse-control disorder via dopamine D3 receptors". Synapse. 69 (4): 183–189. doi:10.1002/syn.21805. PMID 25645960.
- ^ Garcia-Ruiz PJ (2018). "Impulse Control Disorders and Dopamine-Related Creativity: Pathogenesis and Mechanism, Short Review, and Hypothesis". Front Neurol. 9: 1041. doi:10.3389/fneur.2018.01041. PMC 6291460. PMID 30574117.
- ^ Williams BD, Lee K, Ewah SO, Neelam K (2024). "Aripiprazole and Other Third-Generation Antipsychotics as a Risk Factor for Impulse Control Disorders: A Systematic Review and Meta-Analysis". J Clin Psychopharmacol. 44 (1): 39–48. doi:10.1097/JCP.0000000000001773. PMID 38011021.
- ^ Kubota H, Zhou X, Zhang X, Watanabe H, Nagai T (August 2024). "Pramipexole Hyperactivates the External Globus Pallidus and Impairs Decision-Making in a Mouse Model of Parkinson's Disease". Int J Mol Sci. 25 (16): 8849. doi:10.3390/ijms25168849. PMC 11354263. PMID 39201535.
- ^ Howell M, Avidan AY, Foldvary-Schaefer N, Malkani RG, During EH, Roland JP, et al. (April 2023). "Management of REM sleep behavior disorder: an American Academy of Sleep Medicine clinical practice guideline". J Clin Sleep Med. 19 (4): 759–768. doi:10.5664/jcsm.10424. PMC 10071384. PMID 36515157.
- ^ Hoque R, Chesson AL (February 2010). "Pharmacologically induced/exacerbated restless legs syndrome, periodic limb movements of sleep, and REM behavior disorder/REM sleep without atonia: literature review, qualitative scoring, and comparative analysis". J Clin Sleep Med. 6 (1): 79–83. doi:10.5664/jcsm.27716. PMC 2823282. PMID 20191944.
- ^ Louden MB, Morehead MA, Schmidt HS (1995). "Activation by selegiline (Eldepryle) of REM sleep behavior disorder in parkinsonism". W V Med J. 91 (3): 101. PMID 7747490.
- ^ a b Goldberg SR, Yasar S, Bergman J, Youdim MB (December 1994). "Introduction: examination of clinical and preclinical pharmacologic data relating to abuse liability of l-deprenyl (selegiline)". Clin Pharmacol Ther. 56 (6 Pt 2): 721–724. doi:10.1038/clpt.1994.201. PMID 7995013.
- ^ a b Winger GD, Yasar S, Negus SS, Goldberg SR (December 1994). "Intravenous self-administration studies with l-deprenyl (selegiline) in monkeys". Clin Pharmacol Ther. 56 (6 Pt 2): 774–780. doi:10.1038/clpt.1994.208. hdl:2027.42/110034. PMID 7995020.
- ^ a b Yasar S, Gaál J, Panlilio LV, Justinova Z, Molnár SV, Redhi GH, et al. (January 2006). "A comparison of drug-seeking behavior maintained by D-amphetamine, L-deprenyl (selegiline), and D-deprenyl under a second-order schedule in squirrel monkeys". Psychopharmacology (Berl). 183 (4): 413–421. doi:10.1007/s00213-005-0200-7. PMC 1360227. PMID 16292593.
- ^ McKean AJ, Leung JG, Dare FY, Sola CL, Schak KM (2015). "The Perils of Illegitimate Online Pharmacies: Substance-Induced Panic Attacks and Mood Instability Associated With Selegiline and Phenylethylamine". Psychosomatics. 56 (5): 583–587. doi:10.1016/j.psym.2015.05.003. PMID 26198572.
- ^ Monteith S, Glenn T, Bauer R, Conell J, Bauer M (March 2016). "Availability of prescription drugs for bipolar disorder at online pharmacies". J Affect Disord. 193: 59–65. doi:10.1016/j.jad.2015.12.043. PMID 26766033.
- ^ Kuhn W, Müller T (1996). "The clinical potential of Deprenyl in neurologic and psychiatric disorders". Deprenyl — Past and Future. Vol. 48. pp. 85–93. doi:10.1007/978-3-7091-7494-4_8. ISBN 978-3-211-82891-5. PMID 8988464.
{{cite book}}:|journal=ignored (help) - ^ a b c Heinonen EH, Myllylä V (July 1998). "Safety of selegiline (deprenyl) in the treatment of Parkinson's disease". Drug Safety. 19 (1): 11–22. doi:10.2165/00002018-199819010-00002. PMID 9673855. S2CID 9632549.
- ^ Csoti I, Storch A, Müller W, Jost WH (December 1, 2012). "Drug interactions with selegiline versus rasagiline". Basal Ganglia. Monoamine oxidase B Inhibitors. 2 (4, Supplement): S27–S31. doi:10.1016/j.baga.2012.06.003. ISSN 2210-5336.
- ^ Gillman PK (October 2005). "Monoamine oxidase inhibitors, opioid analgesics and serotonin toxicity". British Journal of Anaesthesia. 95 (4): 434–441. doi:10.1093/bja/aei210. PMID 16051647.
- ^ Jessen L, Kovalick LJ, Azzaro AJ (April 2008). "The selegiline transdermal system (emsam): a therapeutic option for the treatment of major depressive disorder". P & T. 33 (4): 212–246. PMC 2730099. PMID 19750165.
- ^ Azzaro AJ, VanDenBerg CM, Ziemniak J, Kemper EM, Blob LF, Campbell BJ (August 2007). "Evaluation of the potential for pharmacodynamic and pharmacokinetic drug interactions between selegiline transdermal system and two sympathomimetic agents (pseudoephedrine and phenylpropanolamine) in healthy volunteers". J Clin Pharmacol. 47 (8): 978–90. doi:10.1177/0091270007302950. PMID 17554106.
- ^ a b Elkashef A, Vocci F, Hanson G, White J, Wickes W, Tiihonen J (2008). "Pharmacotherapy of methamphetamine addiction: an update". Subst Abus. 29 (3): 31–49. doi:10.1080/08897070802218554. PMC 2597382. PMID 19042205.
- ^ a b Newton TF, De La Garza R, Fong T, Chiang N, Holmes TH, Bloch DA, et al. (December 2005). "A comprehensive assessment of the safety of intravenous methamphetamine administration during treatment with selegiline". Pharmacol Biochem Behav. 82 (4): 704–711. doi:10.1016/j.pbb.2005.11.012. PMID 16413604.
- ^ a b Finberg JP (August 2014). "Update on the pharmacology of selective inhibitors of MAO-A and MAO-B: focus on modulation of CNS monoamine neurotransmitter release". Pharmacol Ther. 143 (2): 133–152. doi:10.1016/j.pharmthera.2014.02.010. PMID 24607445.
- ^ a b Houtsmuller EJ, Notes LD, Newton T, van Sluis N, Chiang N, Elkashef A, et al. (February 2004). "Transdermal selegiline and intravenous cocaine: safety and interactions". Psychopharmacology (Berl). 172 (1): 31–40. doi:10.1007/s00213-003-1616-6. PMID 14605792.
- ^ a b Bartzokis G, Beckson M, Newton T, Mandelkern M, Mintz J, Foster JA, et al. (June 1999). "Selegiline effects on cocaine-induced changes in medial temporal lobe metabolism and subjective ratings of euphoria". Neuropsychopharmacology. 20 (6): 582–590. doi:10.1016/S0893-133X(98)00092-X. PMID 10327427.
- ^ a b Haberny KA, Walsh SL, Ginn DH, Wilkins JN, Garner JE, Setoda D, et al. (July 1995). "Absence of acute cocaine interactions with the MAO-B inhibitor selegiline". Drug Alcohol Depend. 39 (1): 55–62. doi:10.1016/0376-8716(95)01137-n. PMID 7587975.
- ^ a b Harris DS, Everhart T, Jacob P, Lin E, Mendelson JE, Jones RT (August 2009). "A phase 1 trial of pharmacologic interactions between transdermal selegiline and a 4-hour cocaine infusion". BMC Clin Pharmacol. 9: 13. doi:10.1186/1472-6904-9-13. PMC 2731040. PMID 19646280.
- ^ a b Newton TF, Kalechstein A, Beckson M, Bartzokis G, Bridge TP, Ling W (October 1999). "Effects of selegiline pretreatment on response to experimental cocaine administration". Psychiatry Res. 87 (2–3): 101–106. doi:10.1016/s0165-1781(99)00058-x. PMID 10579543.
- ^ Feinberg SS (November 2004). "Combining stimulants with monoamine oxidase inhibitors: a review of uses and one possible additional indication". J Clin Psychiatry. 65 (11): 1520–1524. doi:10.4088/jcp.v65n1113. PMID 15554766.
- ^ Thomas SJ, Shin M, McInnis MG, Bostwick JR (April 2015). "Combination therapy with monoamine oxidase inhibitors and other antidepressants or stimulants: strategies for the management of treatment-resistant depression". Pharmacotherapy. 35 (4): 433–449. doi:10.1002/phar.1576. hdl:2027.42/111275. PMID 25884531.
- ^ Israel JA (2015). "Combining Stimulants and Monoamine Oxidase Inhibitors: A Reexamination of the Literature and a Report of a New Treatment Combination". Prim Care Companion CNS Disord. 17 (6). doi:10.4088/PCC.15br01836. PMC 4805402. PMID 27057401.
- ^ Culpepper L, Kovalick LJ (2008). "A review of the literature on the selegiline transdermal system: an effective and well-tolerated monoamine oxidase inhibitor for the treatment of depression". Prim Care Companion J Clin Psychiatry. 10 (1): 25–30. doi:10.4088/pcc.v10n0105. PMC 2249821. PMID 18311418.
- ^ Eccles R (January 2007). "Substitution of phenylephrine for pseudoephedrine as a nasal decongeststant. An illogical way to control methamphetamine abuse". Br J Clin Pharmacol. 63 (1): 10–14. doi:10.1111/j.1365-2125.2006.02833.x. PMC 2000711. PMID 17116124.
- ^ Richards E, Lopez MJ, Maani CV (2023). "Phenylephrine". StatPearls. Treasure Island, Florida: StatPearls Publishing. PMID 30521222. Retrieved April 27, 2023.
- ^ Schachter M (2002). "Drugs affecting autonomic functions or the extrapyramidal system". Side Effects of Drugs Annual. Vol. 25. Elsevier. pp. 166–174. doi:10.1016/s0378-6080(02)80020-4. ISBN 978-0-444-50674-0.
- ^ Rose LM, Ohlinger MJ, Mauro VF (September 2000). "A hypertensive reaction induced by concurrent use of selegiline and dopamine". Ann Pharmacother. 34 (9): 1020–1024. doi:10.1345/aph.19221. PMID 10981248.
- ^ a b c d Gillman PK (February 2011). "Advances pertaining to the pharmacology and interactions of irreversible nonselective monoamine oxidase inhibitors". J Clin Psychopharmacol. 31 (1): 66–74. doi:10.1097/JCP.0b013e31820469ea. PMID 21192146.
- ^ a b c d Gillman PK (November 2018). "A reassessment of the safety profile of monoamine oxidase inhibitors: elucidating tired old tyramine myths". J Neural Transm (Vienna). 125 (11): 1707–1717. doi:10.1007/s00702-018-1932-y. PMID 30255284.
- ^ a b c d Van den Eynde V, Godet L, Redhead C, Horwitz A, Barnett B (2023). "Monoamine Oxidase Inhibitors and Clinically Relevant Drug Interactions: A Guide for Preventing Serotonin Toxicity and Hypertensive Reactions". Psychiatric Annals. 53 (8): 353–358. doi:10.3928/00485713-20230713-02. ISSN 0048-5713.
- ^ a b Van den Eynde V, Abdelmoemin WR, Abraham MM, Amsterdam JD, Anderson IM, Andrade C, et al. (July 2022). "The prescriber's guide to classic MAO inhibitors (phenelzine, tranylcypromine, isocarboxazid) for treatment-resistant depression". CNS Spectr. 28 (4): 427–440. doi:10.1017/S1092852922000906. hdl:2292/61637. PMID 35837681.
- ^ a b Scheinin H, Anttila M, Dahl ML, Karnani H, Nyman L, Taavitsainen P, et al. (October 1998). "CYP2D6 polymorphism is not crucial for the disposition of selegiline". Clin Pharmacol Ther. 64 (4): 402–411. doi:10.1016/S0009-9236(98)90071-6. PMID 9797797.
- ^ a b Laine K, Anttila M, Nyman L, Wahlberg A, Bertilsson L (May 2001). "CYP2C19 polymorphism is not important for the in vivo metabolism of selegiline". Eur J Clin Pharmacol. 57 (2): 137–142. doi:10.1007/s002280100289. PMID 11417445.
- ^ Kivistö KT, Wang JS, Backman JT, Nyman L, Taavitsainen P, Anttila M, et al. (April 2001). "Selegiline pharmacokinetics are unaffected by the CYP3A4 inhibitor itraconazole". Eur J Clin Pharmacol. 57 (1): 37–42. doi:10.1007/s002280100278. PMID 11372588.
- ^ a b "Table of Substrates, Inhibitors and Inducers". U.S. Food and Drug Administration. June 5, 2023. Retrieved July 5, 2024.
- ^ Naoi M, Maruyama W, Shamoto-Nagai M (September 2022). "Neuroprotective Function of Rasagiline and Selegiline, Inhibitors of Type B Monoamine Oxidase, and Role of Monoamine Oxidases in Synucleinopathies". Int J Mol Sci. 23 (19): 11059. doi:10.3390/ijms231911059. PMC 9570229. PMID 36232361.
- ^ a b c Klietz M, Greten S, Wegner F, Höglinger GU (June 2019). "Safety and Tolerability of Pharmacotherapies for Parkinson's Disease in Geriatric Patients". Drugs & Aging. 36 (6): 511–530. doi:10.1007/s40266-019-00654-z. PMID 30937878.
- ^ a b c d Laine K, Anttila M, Helminen A, Karnani H, Huupponen R (March 1999). "Dose linearity study of selegiline pharmacokinetics after oral administration: evidence for strong drug interaction with female sex steroids". Br J Clin Pharmacol. 47 (3): 249–254. doi:10.1046/j.1365-2125.1999.00891.x. PMC 2014223. PMID 10215747.
- ^ a b Palovaara S, Anttila M, Nyman L, Laine K (July 2002). "Effect of concomitant hormone replacement therapy containing estradiol and levonorgestrel on the pharmacokinetics of selegiline". European Journal of Clinical Pharmacology. 58 (4): 259–263. doi:10.1007/s00228-002-0469-y. PMID 12136372.
- ^ a b c d e Anttila M, Sotaniemi EA, Pelkonen O, Rautio A (January 2005). "Marked effect of liver and kidney function on the pharmacokinetics of selegiline". Clin Pharmacol Ther. 77 (1): 54–62. doi:10.1016/j.clpt.2004.09.004. PMID 15637531.
- ^ a b c Zanger UM, Klein K (2013). "Pharmacogenetics of cytochrome P450 2B6 (CYP2B6): advances on polymorphisms, mechanisms, and clinical relevance". Front Genet. 4: 24. doi:10.3389/fgene.2013.00024. PMC 3588594. PMID 23467454.
- ^ a b Hedrich WD, Hassan HE, Wang H (September 2016). "Insights into CYP2B6-mediated drug-drug interactions". Acta Pharm Sin B. 6 (5): 413–425. doi:10.1016/j.apsb.2016.07.016. PMC 5045548. PMID 27709010.
- ^ a b c Sridar C, Kenaan C, Hollenberg PF (December 2012). "Inhibition of bupropion metabolism by selegiline: mechanism-based inactivation of human CYP2B6 and characterization of glutathione and peptide adducts". Drug Metab Dispos. 40 (12): 2256–2266. doi:10.1124/dmd.112.046979. PMC 3500550. PMID 22936314.
- ^ a b Nirogi R, Palacharla RC, Mohammed AR, Manoharan A, Ponnamaneni RK, Bhyrapuneni G (March 2015). "Evaluation of metabolism dependent inhibition of CYP2B6 mediated bupropion hydroxylation in human liver microsomes by monoamine oxidase inhibitors and prediction of potential as perpetrators of drug interaction". Chem Biol Interact. 230: 9–20. Bibcode:2015CBI...230....9N. doi:10.1016/j.cbi.2015.01.028. PMID 25656918.
- ^ Ritter JL, Alexander B (March 1997). "Retrospective study of selegiline-antidepressant drug interactions and a review of the literature". Ann Clin Psychiatry. 9 (1): 7–13. doi:10.1023/a:1026222106851. PMID 9167831.
- ^ Tanner JA, Tyndale RF (December 2017). "Variation in CYP2A6 Activity and Personalized Medicine". J Pers Med. 7 (4): 18. doi:10.3390/jpm7040018. PMC 5748630. PMID 29194389.
- ^ a b Siu EC, Tyndale RF (March 2008). "Selegiline is a mechanism-based inactivator of CYP2A6 inhibiting nicotine metabolism in humans and mice". J Pharmacol Exp Ther. 324 (3): 992–9. doi:10.1124/jpet.107.133900. PMID 18065502.
- ^ Laine K, Anttila M, Huupponen R, Mäki-Ikola O, Heinonen E (2000). "Multiple-dose pharmacokinetics of selegiline and desmethylselegiline suggest saturable tissue binding". Clin Neuropharmacol. 23 (1): 22–27. doi:10.1097/00002826-200001000-00005. PMID 10682227.
- ^ Pfeiffer RF (May 1996). "Antiparkinsonian agents. Drug interactions of clinical significance". Drug Saf. 14 (5): 343–354. doi:10.2165/00002018-199614050-00006. PMID 8800629.
- ^ a b Gaszner P, Miklya I (December 2004). "The use of the synthetic enhancer substances (-)-deprenyl and (-)-BPAP in major depression". Neuropsychopharmacol Hung. 6 (4): 210–220. PMID 15825677.
- ^ a b c Knoll J (August 2003). "Enhancer regulation/endogenous and synthetic enhancer compounds: a neurochemical concept of the innate and acquired drives". Neurochem Res. 28 (8): 1275–1297. doi:10.1023/a:1024224311289. PMID 12834268.
- ^ Knoll J (1995). "Rationale for (-)deprenyl (selegiline) medication in Parkinson's disease and in prevention of age-related nigral changes". Biomed Pharmacother. 49 (4): 187–195. doi:10.1016/0753-3322(96)82619-9. PMID 7669938.
- ^ a b c d e f g Knoll J (2012). How Selegiline ((-)-Deprenyl) Slows Brain Aging. Bentham Science Publishers. pp. 16, 43, 70, 86, 90, 92. ISBN 978-1-60805-470-1. Retrieved July 4, 2024.
- ^ Knoll J (August 1994). "Memories of my 45 years in research". Pharmacol Toxicol. 75 (2): 65–72. doi:10.1111/j.1600-0773.1994.tb00326.x. PMID 7971740.
- ^ Goldstein DS, Kopin IJ, Sharabi Y (December 2014). "Catecholamine autotoxicity. Implications for pharmacology and therapeutics of Parkinson disease and related disorders". Pharmacol Ther. 144 (3): 268–82. doi:10.1016/j.pharmthera.2014.06.006. PMC 4591072. PMID 24945828.
- ^ Goldstein DS (June 2021). "The Catecholaldehyde Hypothesis for the Pathogenesis of Catecholaminergic Neurodegeneration: What We Know and What We Do Not Know". Int J Mol Sci. 22 (11): 5999. doi:10.3390/ijms22115999. PMC 8199574. PMID 34206133.
- ^ Goldstein DS (February 2020). "The catecholaldehyde hypothesis: where MAO fits in". J Neural Transm (Vienna). 127 (2): 169–177. doi:10.1007/s00702-019-02106-9. PMC 10680281. PMID 31807952.
- ^ Heinonen EH, Anttila MI, Karnani HL, Nyman LM, Vuorinen JA, Pyykkö KA, et al. (July 1997). "Desmethylselegiline, a metabolite of selegiline, is an irreversible inhibitor of monoamine oxidase type B in humans". J Clin Pharmacol. 37 (7): 602–609. doi:10.1002/j.1552-4604.1997.tb04342.x. PMID 9243353.
- ^ a b c d Miklya I (June 2014). "Essential difference between the pharmacological spectrum of (-)-deprenyl and rasagiline". Pharmacol Rep. 66 (3): 453–458. doi:10.1016/j.pharep.2013.11.003. PMID 24905523.
- ^ Miklya I (March 2008). "(-)-deprenil, az N-metilprogargilamin-1-aminoindan (J-508) és a J-508 dezmetil analógjának (rasagilin) összehasonlító farmakológiai analízise" [A comparison of the pharmacology of (-)-deprenyl to N-methylpropargylamine-1-aminoindane (J-508) and rasagiline, the desmethyl-analogue of J-508] (PDF). Neuropsychopharmacol Hung (in Hungarian). 10 (1): 15–22. PMID 18771016.
- ^ a b c Kalász H, Magyar K, Szőke É, Adeghate E, Adem A, Hasan MY, et al. (2014). "Metabolism of selegiline [(-)-deprenyl)]". Curr Med Chem. 21 (13): 1522–1530. doi:10.2174/0929867321666131218094352. PMID 24350849.
- ^ Velenosi TJ, Urquhart BL (August 2014). "Pharmacokinetic considerations in chronic kidney disease and patients requiring dialysis". Expert Opin Drug Metab Toxicol. 10 (8): 1131–1143. doi:10.1517/17425255.2014.931371. PMID 24961255.
- ^ Baghdady NT, Banik S, Swartz SA, McIntyre RS (April 2009). "Psychotropic drugs and renal failure: translating the evidence for clinical practice". Adv Ther. 26 (4): 404–424. doi:10.1007/s12325-009-0021-x. PMID 19444657.
- ^ a b c "Selegiline". PubChem. Retrieved July 18, 2024.
- ^ a b c "Selegiline: Uses, Interactions, Mechanism of Action". DrugBank Online. June 5, 1989. Retrieved July 18, 2024.
- ^ a b c "Selegiline". ChemSpider. July 21, 2022. Retrieved July 18, 2024.
- ^ a b c d e Schweizerischer Apotheker-Verein (2000). Index Nominum 2000: International Drug Directory. Medpharm Scientific Publishers. p. 939. ISBN 978-3-88763-075-1. Retrieved July 4, 2024.
- ^ Fowler CJ, Oreland L, Callingham BA (June 1981). "The acetylenic monoamine oxidase inhibitors clorgyline, deprenyl, pargyline and J-508: their properties and applications". J Pharm Pharmacol. 33 (6): 341–347. doi:10.1111/j.2042-7158.1981.tb13800.x. PMID 6115003.
- ^ Ulrich S, Ricken R, Adli M (August 2017). "Tranylcypromine in mind (Part I): Review of pharmacology". Eur Neuropsychopharmacol. 27 (8): 697–713. doi:10.1016/j.euroneuro.2017.05.007. PMID 28655495.
- ^ Ricken R, Ulrich S, Schlattmann P, Adli M (August 2017). "Tranylcypromine in mind (Part II): Review of clinical pharmacology and meta-analysis of controlled studies in depression". Eur Neuropsychopharmacol. 27 (8): 714–731. doi:10.1016/j.euroneuro.2017.04.003. PMID 28579071.
- ^ a b Magyar K (1994). "Behaviour of (-)-deprenyl and its analogues". Amine Oxidases: Function and Dysfunction. Vol. 41. pp. 167–175. doi:10.1007/978-3-7091-9324-2_23. ISBN 978-3-211-82521-1. PMID 7931223.
{{cite book}}:|journal=ignored (help) - ^ Knoll J, Ecsery Z, Magyar K, Sátory E (1978). "Novel (-)deprenyl-derived selective inhibitors of B-type monoamine oxidase. The relation of structure to their action". Biochem Pharmacol. 27 (13): 1739–1747. doi:10.1016/0006-2952(78)90550-6. PMID 708454.
- ^ Magyar K, Ecseri Z, Bernáth G, Satory E, Knoll J (1980). "Structure-activity relationship of selective inhibitors of MAO-B". Monoamine Oxidases and Their Selective Inhibition. Pergamon. pp. 11–21.
- ^ Knoll J (1980). "Selective inhibitors of MAO-B with different pharmacological profiles". Monoamine Oxidases and Their Selective Inhibition. Pergamon. pp. 23–36.
- ^ Weinreb O, Amit T, Bar-Am O, Youdim MB (April 2012). "Ladostigil: a novel multimodal neuroprotective drug with cholinesterase and brain-selective monoamine oxidase inhibitory activities for Alzheimer's disease treatment". Curr Drug Targets. 13 (4): 483–494. doi:10.2174/138945012799499794. PMID 22280345.
- ^ DE 1568277, Ecsery Z, Kosa I, Knoll J, Somfai E, "Verfahren zur Herstellung von neuen,optisch aktiven Phenylisopylamin-Derivaten [Process for the preparation of new, optically active phenylisopylamine derivatives]", published 1970-04-30, assigned to Chinoin Gyógyszer-és Vegyészeti Termékek Gyára RT
- ^ J. Hermann Nee Voeroes, Z. Ecsery, G. Sabo, L. Arvai, L. Nagi, O. Orban, E. Sanfai, U.S. patent 4,564,706 (1986)
- ^ EP 344675, Hájicek J, Hrbata J, Pihera P, Brunová B, Ferenc M, Krepelka J, Kvapil L, Pospisil J, "Method for the production of selegiline hydrochloride", published 989-12-06, assigned to SPOFA Spojené Podniky Pro Zdravotnickou Vyrobu
- ^ Fowler JS (July 1977). "2-Methyl-3-butyn-2-ol as an acetylene precursor in the Mannich reaction. A new synthesis of suicide inactivators of monoamine oxidase". The Journal of Organic Chemistry. 42 (15): 2637–2639. doi:10.1021/jo00435a026. PMID 874623.
- ^ Zeller EA, Barsky J (November 1952). "In vivo inhibition of liver and brain monoamine oxidase by 1-Isonicotinyl-2-isopropyl hydrazine". Proc Soc Exp Biol Med. 81 (2): 459–461. doi:10.3181/00379727-81-19910. PMID 13027339.
- ^ "Sanofi Extends Holding in Chinoin". The Pharma Letter. September 19, 1993.
- ^ a b c d e Knoll J, Ecseri Z, Kelemen K, Nievel J, Knoll B (May 1965). "Phenylisopropylmethylpropinylamine (E-250), a new spectrum psychic energizer". Archives Internationales de Pharmacodynamie et de Therapie. 155 (1): 154–164. PMID 4378644.
- ^ Bryant JM, Torosdag S, Schvartz N, Fletcher L, Fertig H, Schwartz MS, et al. (October 1961). "Antihypertensive properties of pargyline hydrochloride. New non-hydrazine monoamine oxidase inhibitor compared with sulphonamide diuretics". JAMA. 178: 406–409. doi:10.1001/jama.1961.73040430005010. PMID 13874134.
- ^ a b c d e Healy D (2000). "The Psychopharmacology of Life and Death. Interview with Joseph Knoll.". The Psychopharmacologists, Vol. III: Interviews. London: Arnold. pp. 81–110. doi:10.4324/9781003058892-3. ISBN 978-0-340-76110-6.
- ^ a b Magyar K, Vizi ES, Ecseri Z, Knoll J (1967). "Comparative pharmacological analysis of the optical isomers of phenyl-isopropyl-methyl-propinylamine (E-250)". Acta Physiologica Academiae Scientiarum Hungaricae. 32 (4): 377–387. PMID 5595908.
- ^ Johnston JP (July 1968). "Some observations upon a new inhibitor of monoamine oxidase in brain tissue". Biochem Pharmacol. 17 (7): 1285–1297. doi:10.1016/0006-2952(68)90066-x. PMID 5659776.
- ^ Knoll J, Magyar K (1972). "Some puzzling pharmacological effects of monoamine oxidase inhibitors". Adv Biochem Psychopharmacol. 5: 393–408. PMID 5066229.
- ^ Knoll J, Vizi ES, Somogyi G (1968). "Phenylisopropylmethylpropynylamine (E-250), a Monoaminooxidase Inhibitor Antagonising Effects of Tyramine". Arzneimittel-Forschung. 18 (1): 109–112.
- ^ Varga E (1965). "Vorläufiger Bericht über die Wirkung des Präparates E-250 (phenyl-isopropyl-methyl-propinylamine-chlorhydrat)". III Conferentia Hungarica pro Therapia et Investigatione in Pharmacologia. Budapest: Publishing House of the Hungarian Academy of Sciences. pp. 197–201.
- ^ a b Varga E, Tringer L (1967). "Clinical trial of a new type promptly acting psychoenergetic agent (phenyl-isopropyl-methylpropinyl-HCl, "E-250")". Acta Med Acad Sci Hung. 23 (3): 289–295. PMID 6056555.
- ^ Tringer L, Haits G, Varga E (1971). "The effect of (-) E-250,(-) L-phenyl-isopropylmethylpropinyl-amine HCl, in depression". V. Conferentia Hungarica pro Therapia et Investigatione in Pharmacologia. pp. 111–114.
- ^ Mann J, Gershon S (March 1980). "L-deprenyl, a selective monoamine oxidase type-B inhibitor in endogenous depression". Life Sci. 26 (11): 877–882. doi:10.1016/0024-3205(80)90350-1. PMID 6768943.
- ^ a b Birkmayer W, Riederer P, Linauer W, Knoll J (1984). "L-deprenyl plus L-phenylalanine in the treatment of depression". J Neural Transm. 59 (1): 81–87. doi:10.1007/BF01249880. PMID 6425455.
- ^ Birkmayer W, Riederer P, Youdim MB, Linauer W (1975). "The potentiation of the anti akinetic effect after L-dopa treatment by an inhibitor of MAO-B, Deprenil". Journal of Neural Transmission. 36 (3–4): 303–326. doi:10.1007/BF01253131. PMID 1172524. S2CID 38179089. Archived from the original on February 12, 2013.
- ^ Knoll J (May 1985). "The facilitation of dopaminergic activity in the aged brain by (-)deprenyl. A proposal for a strategy to improve the quality of life in senescence". Mech Ageing Dev. 30 (2): 109–122. doi:10.1016/0047-6374(85)90001-6. PMID 3927074.
- ^ Birkmayer W, Knoll J, Riederer P, Youdim MB, Hars V, Marton J (1985). "Increased life expectancy resulting from addition of L-deprenyl to Madopar treatment in Parkinson's disease: a longterm study". J Neural Transm. 64 (2): 113–127. doi:10.1007/BF01245973. PMID 3935752.
- ^ Knoll J (December 1988). "The striatal dopamine dependency of life span in male rats. Longevity study with (-)deprenyl". Mech Ageing Dev. 46 (1–3): 237–262. doi:10.1016/0047-6374(88)90128-5. PMID 3147347.
- ^ a b Cromie WJ (November 7, 2002). "Bodkin is Patching up Depression". Harvard University Gazette. Retrieved September 8, 2007.
- ^ Bishop K (June 10, 1992). "'Smart Drugs': Elixir or Snake Oil?". The New York Times. Retrieved August 16, 2024.
And in the case of selegiline (also known as Deprenyl or l-deprenyl), which is prescribed for the treatment of Parkinson's disease and which smart-drug users have ordered by mail from Switzerland, the drug itself produces amphetamine as a byproduct in the brain. [...]
- ^ Reynolds GP, Elsworth JD, Blau K, Sandler M, Lees AJ, Stern GM (December 1978). "Deprenyl is metabolized to methamphetamine and amphetamine in man". Br J Clin Pharmacol. 6 (6): 542–544. doi:10.1111/j.1365-2125.1978.tb00883.x. PMC 1429688. PMID 728327.
- ^ Dezsi L, Vecsei L (2017). "Monoamine Oxidase B Inhibitors in Parkinson's Disease". CNS Neurol Disord Drug Targets. 16 (4): 425–439. doi:10.2174/1871527316666170124165222. PMID 28124620.
- ^ a b Knoll J (2005). "Enhancer Regulation: A Neurochemical Approach to the Innate and Acquired Drives". The Brain and Its Self: A Neurochemical Concept of the Innate and Acquired Drives. Berlin/Heidelberg: Springer-Verlag. pp. 25–94. doi:10.1007/3-540-27434-0_4. ISBN 978-3-540-23969-7.
- ^ Knoll J, Miklya I (1994). "Multiple, small dose administration of (-)deprenyl enhances catecholaminergic activity and diminishes serotoninergic activity in the brain and these effects are unrelated to MAO-B inhibition". Arch Int Pharmacodyn Ther. 328 (1): 1–15. PMID 7893186.
- ^ Knoll J, Miklya I, Knoll B, Markó R, Kelemen K (1996). "(-)Deprenyl and (-)1-phenyl-2-propylaminopentane, [(-)PPAP], act primarily as potent stimulants of action potential-transmitter release coupling in the catecholaminergic neurons". Life Sci. 58 (10): 817–827. doi:10.1016/0024-3205(96)00014-8. PMID 8602114.
- ^ Knoll J, Knoll B, Török Z, Timár J, Yasar S (1992). "The pharmacology of 1-phenyl-2-propylamino-pentane (PPAP), a deprenyl-derived new spectrum psychostimulant". Arch Int Pharmacodyn Ther. 316: 5–29. PMID 1356324.
- ^ Knoll J, Yoneda F, Knoll B, Ohde H, Miklya I (December 1999). "(-)1-(Benzofuran-2-yl)-2-propylaminopentane, [(-)BPAP], a selective enhancer of the impulse propagation mediated release of catecholamines and serotonin in the brain". Br J Pharmacol. 128 (8): 1723–1732. doi:10.1038/sj.bjp.0702995. PMC 1571822. PMID 10588928.
- ^ Knoll B, Timar J, Knoll J (1988). "Amphetamine-derived potent psychostimulants devoid of biogenic amine releasing properties". Pharmacological Research Communications. 20: 119–120. doi:10.1016/S0031-6989(88)80670-2.
- ^ Frampton JE, Plosker GL (2007). "Selegiline transdermal system: in the treatment of major depressive disorder". Drugs. 67 (2): 257–265, discussion 266–267. doi:10.2165/00003495-200767020-00006. PMID 17284087. S2CID 42425086.
- ^ Duffy M (December 3, 2002). "Patch Raises New Hope For Beating Depression". The New York Times. ISSN 0362-4331.
- ^ Cascade EF, Kalali AH, Preskorn SH (June 2007). "Emsam: the first year". Psychiatry. 4 (6): 19–21. PMC 2921248. PMID 20711332.
- ^ Harsing LG, Knoll J, Miklya I (August 2022). "Enhancer Regulation of Dopaminergic Neurochemical Transmission in the Striatum". Int J Mol Sci. 23 (15): 8543. doi:10.3390/ijms23158543. PMC 9369307. PMID 35955676.
- ^ a b c Elks J (1990). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer US. p. 441. ISBN 978-1-4757-2085-3. Retrieved July 4, 2024.
- ^ a b Morton IK, Hall JM (1999). Concise Dictionary of Pharmacological Agents: Properties and Synonyms. Springer Netherlands. p. 254. ISBN 978-94-011-4439-1. Retrieved July 4, 2024.
- ^ a b Knoll J, Zelena D, Timar J, Baghy K, Mervai Z, Miklya I (January 2020). "Synthetic enhancer compounds, besides acting on biogenic amine system, influence the glutamate transmission and stress response". Behav Brain Res. 378: 112290. doi:10.1016/j.bbr.2019.112290. PMID 31610214.
- ^ Feinberg S (November 1, 2006). "EMSAM: A User-Friendly MAOI?". CARLAT PUBLISHING. Retrieved July 4, 2024.
- ^ Dorsey ER, Thompson JP, Dayoub EJ, George B, Saubermann LA, Holloway RG (July 2009). "Selegiline shortage: Causes and costs of a generic drug shortage". Neurology. 73 (3): 213–217. doi:10.1212/WNL.0b013e3181ae7b04. PMC 2715573. PMID 19620609.
- ^ a b Ferdinandy P, Yoneda F, Muraoka S, Fürst S, Gyires K, Miklya I (February 2020). "Geroprotection in the future. In memoriam of Joseph Knoll: The selegiline story continues". European Journal of Pharmacology. 868: 172793. doi:10.1016/j.ejphar.2019.172793. PMID 31743738. S2CID 208185366.
- ^ Pearce D (1995). The Hedonistic Imperative. OCLC 44325836.
- ^ "Sam Barker and David Pearce on Art, Paradise Engineering, and Existential Hope (With Guest Mix) | The FLI Podcast". Future of Life Institute (audio, transcript). June 24, 2020.
- ^ a b Murphy HT (December 14, 2022). "Sam Bankman-Fried Confirmed He Wears an Emsam Patch. What's an Emsam Patch?". Slate Magazine. Retrieved July 2, 2024.
- ^ a b Sigalos M (August 14, 2023). "Sam Bankman-Fried wins approval to receive Adderall for ADHD while in jail". CNBC. Retrieved July 2, 2024.
- ^ Alexander S (November 16, 2022). "The Psychopharmacology Of The FTX Crash". Astral Codex Ten. Retrieved July 4, 2024.
- ^ Hurwitz G (2019). Out of the dark. Penguin Books. p. 431. ISBN 9780718185480.
- ^ Lidsky T, Schneider J (2010). Brain Candy: Boost Your Brain Power with Vitamins, Supplements, Drugs, and Other Substance. Touchstone. pp. 89–93. ISBN 978-0-7432-1843-6. Retrieved July 5, 2024.
- ^ a b Miklya I (2011). "The Knoll-Concept to Decrease the Prevalence of Parkinson's Disease". In Finkelstein D (ed.). Towards New Therapies for Parkinson's Disease. pp. 77–100. ISBN 978-953-307-463-4. Retrieved July 29, 2024.
- ^ Saunders N, Heron L (1993). E for Ecstasy. London: N. Saunders. ISBN 978-0-9501628-8-1. OCLC 29388575.[page needed]
- ^ Saunders N. "Test results of 30 samples of Ecstasy bought in British clubs between 11/94 and 7/95".
- ^ a b c d Docherty JR (June 2008). "Pharmacology of stimulants prohibited by the World Anti-Doping Agency (WADA)". Br J Pharmacol. 154 (3): 606–622. doi:10.1038/bjp.2008.124. PMC 2439527. PMID 18500382.
- ^ Heal DJ, Smith SL, Gosden J, Nutt DJ (June 2013). "Amphetamine, past and present--a pharmacological and clinical perspective". J Psychopharmacol. 27 (6): 479–496. doi:10.1177/0269881113482532. PMC 3666194. PMID 23539642.
- ^ Barkholtz HM, Hadzima R, Miles A (July 2023). "Pharmacology of R-(-)-Methamphetamine in Humans: A Systematic Review of the Literature". ACS Pharmacol Transl Sci. 6 (7): 914–924. doi:10.1021/acsptsci.3c00019. PMC 10353062. PMID 37470013.
- ^ Smith RC, Davis JM (June 1977). "Comparative effects of d-amphetamine, l-amphetamine, and methylphenidate on mood in man". Psychopharmacology (Berl). 53 (1): 1–12. doi:10.1007/BF00426687. PMID 407607.
- ^ Zhou SF, Liu JP, Lai XS (2009). "Substrate specificity, inhibitors and regulation of human cytochrome P450 2D6 and implications in drug development". Curr Med Chem. 16 (21): 2661–805. doi:10.2174/092986709788681985. PMID 19601803.
- ^ Knoll J, Miklya I (December 2016). "Longevity study with low doses of selegiline/(-)-deprenyl and (2R)-1-(1-benzofuran-2-yl)-N-propylpentane-2-amine (BPAP)". Life Sciences. 167: 32–38. doi:10.1016/j.lfs.2016.10.023. PMID 27777099.
- ^ Furst S (2018). "In memoriam Joseph Knoll (1925-2018) | Hungarian Society for Experimental and Clinical Pharmacology". Retrieved April 10, 2023.
- ^ Froestl W, Muhs A, Pfeifer A (2014). "Cognitive enhancers (nootropics). Part 2: drugs interacting with enzymes. Update 2014". J Alzheimers Dis. 42 (1): 1–68. doi:10.3233/JAD-140402. PMID 24903780.
- ^ Carageorgiou H, Sideris AC, Messari I, Liakou CI, Tsakiris S (August 2008). "The effects of rivastigmine plus selegiline on brain acetylcholinesterase, (Na, K)-, Mg-ATPase activities, antioxidant status, and learning performance of aged rats". Neuropsychiatric Disease and Treatment. 4 (4): 687–699. doi:10.2147/ndt.s3272. PMC 2536534. PMID 19043511.
- ^ Stoll S, Hafner U, Pohl O, Müller WE (1994). "Age-related memory decline and longevity under treatment with selegiline". Life Sciences. 55 (25–26): 2155–2163. doi:10.1016/0024-3205(94)00396-3. PMID 7997074.
- ^ Puurunen K, Jolkkonen J, Sirviö J, Haapalinna A, Sivenius J (February 2001). "Selegiline combined with enriched-environment housing attenuates spatial learning deficits following focal cerebral ischemia in rats". Experimental Neurology. 167 (2): 348–355. doi:10.1006/exnr.2000.7563. PMID 11161623. S2CID 22769187.
- ^ Knoll J (May 1992). "The pharmacological profile of (-)deprenyl (selegiline) and its relevance for humans: a personal view". Pharmacology & Toxicology. 70 (5 Pt 1): 317–321. doi:10.1111/j.1600-0773.1992.tb00480.x. PMID 1608919.
- ^ a b Janssen PA, Leysen JE, Megens AA, Awouters FH (September 1999). "Does phenylethylamine act as an endogenous amphetamine in some patients?". Int J Neuropsychopharmacol. 2 (3): 229–240. doi:10.1017/S1461145799001522. PMID 11281991.
- ^ Sabelli HC (March 1991). "Rapid treatment of depression with selegiline-phenylalanine combination". J Clin Psychiatry. 52 (3): 137. PMID 1900832.
- ^ Sabelli H, Fink P, Fawcett J, Tom C (1996). "Sustained antidepressant effect of PEA replacement". J Neuropsychiatry Clin Neurosci. 8 (2): 168–71. doi:10.1176/jnp.8.2.168. PMID 9081552.
- ^ a b c Simpson HB, Schneier FR, Marshall RD, Campeas RB, Vermes D, Silvestre J, et al. (1998). "Low dose selegiline (L-Deprenyl) in social phobia". Depress Anxiety. 7 (3): 126–129. doi:10.1002/(SICI)1520-6394(1998)7:3<126::AID-DA5>3.0.CO;2-9. PMID 9656093.
- ^ Padilha SC, Virtuoso S, Tonin FS, Borba HH, Pontarolo R (October 2018). "Efficacy and safety of drugs for attention deficit hyperactivity disorder in children and adolescents: a network meta-analysis". European Child & Adolescent Psychiatry. 27 (10): 1335–1345. doi:10.1007/s00787-018-1125-0. PMID 29460165. S2CID 3402756.
- ^ a b Buoli M, Serati M, Cahn W (2016). "Alternative pharmacological strategies for adult ADHD treatment: a systematic review". Expert Review of Neurotherapeutics. 16 (2): 131–144. doi:10.1586/14737175.2016.1135735. PMID 26693882. S2CID 33004517.
- ^ Bloch MH, Panza KE, Landeros-Weisenberger A, Leckman JF (September 2009). "Meta-analysis: treatment of attention-deficit/hyperactivity disorder in children with comorbid tic disorders". J Am Acad Child Adolesc Psychiatry. 48 (9): 884–893. doi:10.1097/CHI.0b013e3181b26e9f. PMC 3943246. PMID 19625978.
- ^ Rubinstein S, Malone MA, Roberts W, Logan WJ (August 2006). "Placebo-controlled study examining effects of selegiline in children with attention-deficit/hyperactivity disorder". Journal of Child and Adolescent Psychopharmacology. 16 (4): 404–415. doi:10.1089/cap.2006.16.404. PMID 16958566.
- ^ Akhondzadeh S, Tavakolian R, Davari-Ashtiani R, Arabgol F, Amini H (August 2003). "Selegiline in the treatment of attention deficit hyperactivity disorder in children: a double blind and randomized trial". Progress in Neuro-Psychopharmacology & Biological Psychiatry. 27 (5): 841–845. doi:10.1016/S0278-5846(03)00117-9. PMID 12921918. S2CID 23234928.
- ^ Wilens TE, Spencer TJ, Biederman J (March 2002). "A review of the pharmacotherapy of adults with attention-deficit/hyperactivity disorder". Journal of Attention Disorders. 5 (4): 189–202. doi:10.1177/108705470100500401. PMID 11967475. S2CID 37417459.
- ^ Tcheremissine OV, Salazar JO (June 2008). "Pharmacotherapy of adult attention deficit/hyperactivity disorder: review of evidence-based practices and future directions". Expert Opinion on Pharmacotherapy. 9 (8): 1299–1310. doi:10.1517/14656566.9.8.1299. PMID 18473705. S2CID 73193888.
- ^ a b Mechcatie E (July 2003). "Transdermal MAO inhibitor patch effective for ADHD". Clinical Psychiatry News.
- ^ a b Hailwood JM (September 27, 2018). Novel approaches towards pharmacological enhancement of motivation (Thesis). University of Cambridge. pp. 13–14. doi:10.17863/CAM.40216.
- ^ a b Callaghan CK, Rouine J, O'Mara SM (2018). "Potential roles for opioid receptors in motivation and major depressive disorder". The Opioid System as the Interface between the Brain's Cognitive and Motivational Systems. Progress in Brain Research. Vol. 239. pp. 89–119. doi:10.1016/bs.pbr.2018.07.009. ISBN 978-0-444-64167-0. PMID 30314570.
- ^ Yohn SE, Reynolds S, Tripodi G, Correa M, Salamone JD (April 2018). "The monoamine-oxidase B inhibitor deprenyl increases selection of high-effort activity in rats tested on a progressive ratio/chow feeding choice procedure: Implications for treating motivational dysfunctions". Behav Brain Res. 342: 27–34. doi:10.1016/j.bbr.2017.12.039. PMID 29292157.
- ^ Contreras-Mora H, Rowland MA, Yohn SE, Correa M, Salamone JD (March 2018). "Partial reversal of the effort-related motivational effects of tetrabenazine with the MAO-B inhibitor deprenyl (selegiline): Implications for treating motivational dysfunctions". Pharmacol Biochem Behav. 166: 13–20. doi:10.1016/j.pbb.2018.01.001. PMID 29309800.
- ^ van Dalen JW, Moll van Charante EP, Nederkoorn PJ, van Gool WA, Richard E (March 2013). "Poststroke apathy". Stroke. 44 (3): 851–860. doi:10.1161/STROKEAHA.112.674614. PMID 23362076.
- ^ a b Al-Adawi SH (1998). The neuropsychopharmacology of motivation: an examination of reward and frontal-subcortical mechanisms and functions (Thesis). Retrieved July 5, 2024.
- ^ Marin RS, Fogel BS, Hawkins J, Duffy J, Krupp B (1995). "Apathy: a treatable syndrome". J Neuropsychiatry Clin Neurosci. 7 (1): 23–30. doi:10.1176/jnp.7.1.23. PMID 7711487.
- ^ Newburn G, Newburn D (February 2005). "Selegiline in the management of apathy following traumatic brain injury". Brain Inj. 19 (2): 149–154. doi:10.1080/02699050410001719989. PMID 15841758.
- ^ Moutaouakil F, El Otmani H, Fadel H, Slassi I (December 2009). "Apathie sévère après un traumatisme crânien: évolution favorable sous Sélégiline" [Severe apathy following head injury: improvement with Selegiline treatment]. Neurochirurgie (in French). 55 (6): 551–554. doi:10.1016/j.neuchi.2008.10.009. PMID 19084243.
- ^ Marin RS, Wilkosz PA (2005). "Disorders of diminished motivation". J Head Trauma Rehabil. 20 (4): 377–88. doi:10.1097/00001199-200507000-00009. PMID 16030444.
- ^ a b Deng X, Shang X, Guo K, Zhou L, Wang Y, Wu Y, et al. (August 2023). "Efficacy and safety of antidepressants for smoking cessation: A systematic review and network meta-analysis". Addict Biol. 28 (8): e13303. doi:10.1111/adb.13303. PMID 37500482.
- ^ a b Hajizadeh A, Howes S, Theodoulou A, Klemperer E, Hartmann-Boyce J, Livingstone-Banks J, et al. (May 2023). "Antidepressants for smoking cessation". Cochrane Database Syst Rev. 2023 (5): CD000031. doi:10.1002/14651858.CD000031.pub6. PMC 10207863. PMID 37230961.
- ^ Castells X, Casas M, Pérez-Mañá C, Roncero C, Vidal X, Capellà D (February 2010). Castells X (ed.). "Efficacy of psychostimulant drugs for cocaine dependence". Cochrane Database Syst Rev (2): CD007380. doi:10.1002/14651858.CD007380.pub3. PMID 20166094.
- ^ Costa AM, Lima MS, Mari Jde J (September 2006). "A systematic review on clinical management of antipsychotic-induced sexual dysfunction in schizophrenia". Sao Paulo Med J. 124 (5): 291–297. doi:10.1590/s1516-31802006000500012. PMC 11068300. PMID 17262163.
- ^ Schmidt HM, Hagen M, Kriston L, Soares-Weiser K, Maayan N, Berner MM (November 2012). "Management of sexual dysfunction due to antipsychotic drug therapy". Cochrane Database Syst Rev. 11 (11): CD003546. doi:10.1002/14651858.CD003546.pub3. PMC 7003677. PMID 23152218.
- ^ a b Murphy BP, Chung YC, Park TW, McGorry PD (December 2006). "Pharmacological treatment of primary negative symptoms in schizophrenia: a systematic review". Schizophr Res. 88 (1–3): 5–25. doi:10.1016/j.schres.2006.07.002. PMID 16930948.
- ^ a b c Maski K, Trotti LM, Kotagal S, Robert Auger R, Swick TJ, Rowley JA, et al. (September 2021). "Treatment of central disorders of hypersomnolence: an American Academy of Sleep Medicine systematic review, meta-analysis, and GRADE assessment". J Clin Sleep Med. 17 (9): 1895–1945. doi:10.5664/jcsm.9326. PMC 8636345. PMID 34743790.
- ^ a b c d Nishino S, Kotorii N (2016). "Modes of Action of Drugs Related to Narcolepsy: Pharmacology of Wake-Promoting Compounds and Anticataplectics". Narcolepsy: A Clinical Guide (2 ed.). Cham: Springer International Publishing. pp. 307–329. doi:10.1007/978-3-319-23739-8_22. ISBN 978-3-319-23738-1.
- ^ a b Nishino S, Arrigoni J, Kanbayashi T, Dement WC, Mignot E (1996). "Comparative effects of MAO-A and MAO-B selective inhibitors on canine cataplexy". Sleep Res. 25: 315.
- ^ Annane D, Moore DH, Barnes PR, Miller RG (July 2006). "Psychostimulants for hypersomnia (excessive daytime sleepiness) in myotonic dystrophy". Cochrane Database Syst Rev. 2006 (3): CD003218. doi:10.1002/14651858.CD003218.pub2. PMC 9006877. PMID 16855999.
- ^ a b c d Yeh PG, Spruyt K, DelRosso LM, Walters AS (2023). "A Narrative Review of the Lesser Known Medications for Treatment of Restless Legs Syndrome and Pathogenetic Implications for Their Use". Tremor Other Hyperkinet Mov (N Y). 13: 7. doi:10.5334/tohm.739. PMC 9983500. PMID 36873914.
- ^ a b Aurora RN, Kristo DA, Bista SR, Rowley JA, Zak RS, Casey KR, et al. (August 2012). "The treatment of restless legs syndrome and periodic limb movement disorder in adults--an update for 2012: practice parameters with an evidence-based systematic review and meta-analyses: an American Academy of Sleep Medicine Clinical Practice Guideline". Sleep. 35 (8): 1039–1062. doi:10.5665/sleep.1988. PMC 3397811. PMID 22851801.
- ^ a b Grewal M, Hawa R, Shapiro C (March 2002). "Treatment of periodic limb movements in sleep with selegiline HCl". Mov Disord. 17 (2): 398–401. doi:10.1002/mds.10082. PMID 11921131.
- ^ Soares-Weiser K, Rathbone J, Ogawa Y, Shinohara K, Bergman H (March 2018). "Miscellaneous treatments for antipsychotic-induced tardive dyskinesia". Cochrane Database Syst Rev. 2018 (3): CD000208. doi:10.1002/14651858.CD000208.pub2. PMC 6494382. PMID 29552749.
- ^ Wilcock GK, Birks J, Whitehead A, Evans SJ (February 2002). "The effect of selegiline in the treatment of people with Alzheimer's disease: a meta-analysis of published trials". Int J Geriatr Psychiatry. 17 (2): 175–183. doi:10.1002/gps.545. PMID 11813282.
- ^ Birks J, Flicker L (2003). "Selegiline for Alzheimer's disease". Cochrane Database Syst Rev (1): CD000442. doi:10.1002/14651858.CD000442. PMID 12535396.
- ^ Sanders O, Rajagopal L (June 2020). "Phosphodiesterase Inhibitors for Alzheimer's Disease: A Systematic Review of Clinical Trials and Epidemiology with a Mechanistic Rationale". J Alzheimers Dis Rep. 4 (1): 185–215. doi:10.3233/ADR-200191. PMC 7369141. PMID 32715279.
- ^ Laver K, Dyer S, Whitehead C, Clemson L, Crotty M (April 2016). "Interventions to delay functional decline in people with dementia: a systematic review of systematic reviews". BMJ Open. 6 (4): e010767. doi:10.1136/bmjopen-2015-010767. PMC 4854009. PMID 27121704.
- ^ Stinton C, McKeith I, Taylor JP, Lafortune L, Mioshi E, Mak E, et al. (August 2015). "Pharmacological Management of Lewy Body Dementia: A Systematic Review and Meta-Analysis". Am J Psychiatry. 172 (8): 731–742. doi:10.1176/appi.ajp.2015.14121582. PMID 26085043.
- ^ Beghi E, Binder H, Birle C, Bornstein N, Diserens K, Groppa S, et al. (September 2021). "European Academy of Neurology and European Federation of Neurorehabilitation Societies guideline on pharmacological support in early motor rehabilitation after acute ischaemic stroke". Eur J Neurol. 28 (9): 2831–2845. doi:10.1111/ene.14936. PMID 34152062.
- ^ a b Szymkowicz E, Alnagger N, Seyfzadehdarabad F, Cardone P, Whyte J, Gosseries O (2023). "Pharmacological Treatments". Coma and Disorders of Consciousness (3 ed.). Cham: Springer International Publishing. pp. 115–146. doi:10.1007/978-3-031-50563-8_7. ISBN 978-3-031-50562-1.
- ^ a b Masotta O, Trojano L, Loreto V, Moretta P, Estraneo A (November 2018). "Selegiline in Patients With Disorder of Consciousness: An Open Pilot Study". Can J Neurol Sci. 45 (6): 688–691. doi:10.1017/cjn.2018.315. PMID 30430963.
- ^ Knoll J (1989). "The pharmacology of selegiline ((-)deprenyl). New aspects". Acta Neurologica Scandinavica. Supplementum. 126: 83–91. doi:10.1111/j.1600-0404.1989.tb01787.x. PMID 2515725.
- ^ Ebadi M, Sharma S, Shavali S, El Refaey H (February 2002). "Neuroprotective actions of selegiline". Journal of Neuroscience Research. 67 (3): 285–289. doi:10.1002/jnr.10148. PMID 11813232.
- ^ a b Bentué-Ferrer D, Ménard G, Allain H (1996). "Monoamine Oxidase B Inhibitors: Current Status and Future Potential". CNS Drugs. 6 (3): 217–236. doi:10.2165/00023210-199606030-00005. ISSN 1172-7047.
- ^ Knoll J (1978). "The possible mechanisms of action of (-)deprenyl in Parkinson's disease". Journal of Neural Transmission. 43 (3–4): 177–198. doi:10.1007/BF01246955. PMID 745011.
- ^ Cohen G, Pasik P, Cohen B, Leist A, Mytilineou C, Yahr MD (October 1984). "Pargyline and deprenyl prevent the neurotoxicity of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) in monkeys". European Journal of Pharmacology. 106 (1): 209–210. doi:10.1016/0014-2999(84)90700-3. PMID 6442232.
- ^ Finnegan KT, DeLanney LE, Irwin I, Ricaurte GA, Langston JW (September 1989). "The amine-depleting effects of 5,7-dihydroxytryptamine (5,7-DHT) in C57BL/6 mice do not increase with age". Brain Research. 496 (1–2): 251–256. doi:10.1016/0006-8993(89)91072-x. PMID 2804634.
- ^ a b Moratalla R, Khairnar A, Simola N, Granado N, García-Montes JR, Porceddu PF, et al. (August 2017). "Amphetamine-related drugs neurotoxicity in humans and in experimental animals: Main mechanisms". Prog Neurobiol. 155: 149–170. doi:10.1016/j.pneurobio.2015.09.011. hdl:10261/156486. PMID 26455459.
- ^ a b Puerta E, Aguirre N (July 5, 2011). "Methylenedioxymethamphetamine (MDMA, 'Ecstasy'): Neurodegeneration versus Neuromodulation". Pharmaceuticals. 4 (7): 992–1018. doi:10.3390/ph4070992. ISSN 1424-8247. PMC 4058674.
- ^ Sprague JE, Nichols DE (April 1995). "Inhibition of MAO-B protects against MDMA-induced neurotoxicity in the striatum". Psychopharmacology. 118 (3): 357–359. doi:10.1007/BF02245967. PMID 7542394.
- ^ Sprague JE, Nichols DE (May 1995). "The monoamine oxidase-B inhibitor L-deprenyl protects against 3,4-methylenedioxymethamphetamine-induced lipid peroxidation and long-term serotonergic deficits". J Pharmacol Exp Ther. 273 (2): 667–673. PMID 7538579.
- ^ Alves E, Summavielle T, Alves CJ, Gomes-da-Silva J, Barata JC, Fernandes E, et al. (September 2007). "Monoamine oxidase-B mediates ecstasy-induced neurotoxic effects to adolescent rat brain mitochondria". J Neurosci. 27 (38): 10203–10210. doi:10.1523/JNEUROSCI.2645-07.2007. PMC 6672671. PMID 17881526.
- ^ Corkery JM, Elliott S, Schifano F, Corazza O, Ghodse AH (July 2013). "MDAI (5,6-methylenedioxy-2-aminoindane; 6,7-dihydro-5H-cyclopenta[f][1,3]benzodioxol-6-amine; 'sparkle'; 'mindy') toxicity: a brief overview and update". Hum Psychopharmacol. 28 (4): 345–355. doi:10.1002/hup.2298. PMID 23881883.
- ^ Johnson MP, Huang XM, Nichols DE (December 1991). "Serotonin neurotoxicity in rats after combined treatment with a dopaminergic agent followed by a nonneurotoxic 3,4-methylenedioxymethamphetamine (MDMA) analogue". Pharmacol Biochem Behav. 40 (4): 915–922. doi:10.1016/0091-3057(91)90106-c. PMID 1726189.
- ^ Halladay AK, Kirschner E, Hesse K, Fisher H, Wagner GC (November 2001). "Role of monoamine oxidase inhibition and monoamine depletion in fenfluramine-induced neurotoxicity and serotonin release". Pharmacol Toxicol. 89 (5): 237–248. doi:10.1034/j.1600-0773.2001.d01-154.x (inactive November 17, 2024). PMID 11881977.
{{cite journal}}: CS1 maint: DOI inactive as of November 2024 (link) - ^ Benmansour S, Brunswick DJ (July 1994). "The MAO-B inhibitor deprenyl, but not the MAO-A inhibitor clorgyline, potentiates the neurotoxicity of p-chloroamphetamine". Brain Res. 650 (2): 305–312. doi:10.1016/0006-8993(94)91796-5. PMID 7953696.
- ^ Sprague JE, Johnson MP, Schmidt CJ, Nichols DE (October 1996). "Studies on the mechanism of p-chloroamphetamine neurotoxicity". Biochem Pharmacol. 52 (8): 1271–1277. doi:10.1016/0006-2952(96)00482-0. PMID 8937435.
- ^ Goldberg SR, Yasar S (1997). "Methamphetamine Administration and Associated Neurotoxicity: Effects of Selegiline (l-Deprenyl)". Neurochemistry: Cellular, Molecular, and Clinical Aspects. Boston, MA: Springer US. pp. 327–330. doi:10.1007/978-1-4615-5405-9_55. ISBN 978-1-4613-7468-8.
- ^ Wan FJ, Shiah IS, Lin HC, Huang SY, Tung CS (June 2000). "Nomifensine attenuates d-amphetamine-induced dopamine terminal neurotoxicity in the striatum of rats". Chin J Physiol. 43 (2): 69–74. PMID 10994696.
- ^ Grasing K, Azevedo R, Karuppan S, Ghosh S (January 2001). "Biphasic effects of selegiline on striatal dopamine: lack of effect on methamphetamine-induced dopamine depletion". Neurochem Res. 26 (1): 65–74. doi:10.1023/a:1007632700126. PMID 11358284.
- ^ Davidson C, Chen Q, Zhang X, Xiong X, Lazarus C, Lee TH, et al. (November 2007). "Deprenyl treatment attenuates long-term pre- and post-synaptic changes evoked by chronic methamphetamine". Eur J Pharmacol. 573 (1–3): 100–110. doi:10.1016/j.ejphar.2007.06.046. PMID 17651730.
- ^ Carvalho M, Carmo H, Costa VM, Capela JP, Pontes H, Remião F, et al. (August 2012). "Toxicity of amphetamines: an update". Arch Toxicol. 86 (8): 1167–231. Bibcode:2012ArTox..86.1167C. doi:10.1007/s00204-012-0815-5. PMID 22392347.
- ^ Edinoff AN, Swinford CR, Odisho AS, Burroughs CR, Stark CW, Raslan WA, et al. (2022). "Clinically Relevant Drug Interactions with Monoamine Oxidase Inhibitors". Health Psychol Res. 10 (4): 39576. doi:10.52965/001c.39576. PMC 9680847. PMID 36425231.
- ^ a b "Selegiline transdermal (Emsam) - Somerset Pharmaceuticals". AdisInsight. November 5, 2023. Retrieved July 15, 2024.
- ^ "Selegiline orally disintegrating tablet (Efupi; FPF 1100 NW) - FP Pharmaceutical". AdisInsight. June 3, 2016. Retrieved July 15, 2024.
- ^ a b c Bruyette DS, Ruehl WW, Entriken T, Griffin D, Darling L (March 1997). "Management of canine pituitary-dependent hyperadrenocorticism with l-deprenyl (Anipryl)". Vet Clin North Am Small Anim Pract. 27 (2): 273–286. doi:10.1016/s0195-5616(97)50031-3. PMID 9076907.
- ^ a b Braddock JA, Church DB, Robertson ID (2004). "Selegiline Treatment of Canine Pituitary-Dependent Hyperadrenocorticism" (PDF). Australian Veterinary Journal. Archived from the original (PDF) on November 29, 2010. Retrieved April 8, 2011. (PDF)
- ^ a b Eghianruwa K (2014). Essential Drug Data for Rational Therapy in Veterinary Practice. AuthorHouse. pp. 127–128. ISBN 978-1-4918-0010-2.
- ^ Prpar Mihevc S, Majdič G (2019). "Canine Cognitive Dysfunction and Alzheimer's Disease - Two Facets of the Same Disease?". Front Neurosci. 13: 604. doi:10.3389/fnins.2019.00604. PMC 6582309. PMID 31249505.
- ^ a b "Anipryl Tablets for Animal Use". Drugs.com. Retrieved August 31, 2017.
- ^ Lundgren B. "Canine Cognitive Dysfunction". Veterinary Partner. Retrieved April 8, 2011.
- ^ a b Riviere JE, Papich MG (2013). Veterinary Pharmacology and Therapeutics. John Wiley & Sons. p. 530. ISBN 978-1-118-68590-7.
- ^ a b c d Papich MG (2015). Saunders Handbook of Veterinary Drugs: Small and Large Animal. Elsevier Health Sciences. p. 722. ISBN 978-0-323-24485-5.
External links
[edit]- Aldehyde dehydrogenase inhibitors
- Anti-aging substances
- Antidepressants
- Antiparkinsonian agents
- Aphrodisiacs
- Attention deficit hyperactivity disorder management
- Drugs with unknown mechanisms of action
- Enantiopure drugs
- Methamphetamines
- Monoamine oxidase inhibitors
- Monoaminergic activity enhancers
- Neuroprotective agents
- Nootropics
- Norepinephrine-dopamine releasing agents
- Phenethylamines
- Pro-motivational agents
- Prodrugs
- Propargyl compounds
- Selegiline
- Sigma receptor ligands
- Stimulants
- Substituted amphetamines
- TAAR1 agonists
- Wakefulness-promoting agents
- World Anti-Doping Agency prohibited substances
