A-234 (nerve agent)
![Chemical structure of A-234 according to Mirzayanov[1]](http://upload.wikimedia.org/wikipedia/commons/thumb/c/c5/A-234_Mirzayanov.svg/220px-A-234_Mirzayanov.svg.png)
| |
| Names | |
|---|---|
| IUPAC name
ethyl N-[(1E)-1-(diethylamino)ethylidene]-phosphoramidofluoridate
| |
| Other names
N-2-diethylaminomethylacetamido-ethoxyphosphonofluoridate
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C8H18FN2O2P | |
| Molar mass | 224.216 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
A-234 is an organophosphate nerve agent. It was developed in the Soviet Union under the FOLIANT program and is one of the group of compounds referred to as Novichok agents that were revealed by Vil Mirzayanov.[2][3][4][5] In March 2018 the Russian ambassador to the UK, Alexander Yakovenko, claimed to have been informed by British authorities that A-234 had been identified as the agent used in the poisoning of Sergei and Yulia Skripal.[6] Vladimir Uglev, one of the inventors of the Novichok series of compounds, said he was "99 percent sure that it was A-234" in relation to the 2018 Amesbury poisonings, noting its unusually high persistence in the environment.[7][8]
According to a classified report by the United States Army National Ground Intelligence Center,[9] the agent designated as A-232 and its ethyl analog A-234, developed under the FOLIANT program, were "as toxic as VX, as resistant to treatment as soman, and more difficult to detect and easier to manufacture than VX".
No certain data on toxicity exist but it is estimated that the median lethal concentration of A-234 is 7 mg/m3. This means that half of 70 kg men under slight physical activity – breathing 15 litres of air per minute – would die within two minutes of exposure. This equates to median lethal dose of 0.2 mg via respiration.[10]
Regulated chemical under the Chemical Weapons Convention
[edit]A-234 has recently (entry into force 7 June 2020) been added to Schedule 1 of the Annex on Chemicals of the Chemical Weapons Convention and it has been explicitly named as an example compound for schedule 1.A.14.[11][12] For chemicals listed in Schedule 1, the most stringent declaration and verification measures are in place combined with far-reaching limits and bans on production and use. It is notable to say that Annex 1 does not explicitly relate this structure to the name A-234, instead adding this particular structure to the prohibited compounds section.
Alternative structure
[edit]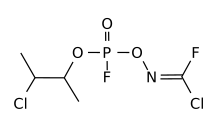
| |
| Names | |
|---|---|
| IUPAC name
([(2-chloro-1-methylpropoxy)fluorohydroxyphosphinyl]oxy)carbonimidic chloride fluoride[13]
| |
| Identifiers | |
3D model (JSmol)
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C5H8Cl2F2NO3P | |
| Molar mass | 269.99 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
An alternative structure for A-234 and the related Novichok agents had previously been proposed by Western chemical weapons experts such as Steven Hoenig and D. Hank Ellison.[14][15][16] These structures were supported by Soviet literature of the time and were tested as acetylcholinesterase inhibitors,[17][18][19] however Mirzayanov explained that a number of weaker agents developed as part of the Foliant program were published in the open literature as organophosphate pesticides, in order to disguise the secret nerve agent program as legitimate pesticide research.
However, the presence of a carbonimidic chloride fluoride group and a dimethylchloroethyl group suggest that this molecule is a member of the C-series of organophosphate agents (C01), with additional mustard and (chlorofluoro-)phosgene oxime radicals, added to produce skin and lung toxicity as well as blistering effects.[20]
See also
[edit]- C01-A035
- C01-A039
- A-208
- A-233 (VR analogue)
- A-230
- A-235
- A-242
- A-243
- A-255
- A-256 (cycloheptoxy seleno-VX)
- VP
References
[edit]- ^ Chai, Peter R.; Hayes, Bryan D.; Erickson, Timothy B.; Boyer, Edward W. (2018). "Novichok agents: A historical, current, and toxicological perspective". Toxicology Communications. 2 (1): 45–48. doi:10.1080/24734306.2018.1475151. PMC 6039123. PMID 30003185.
- ^ Mirzayanov, Vil S. (2008). State Secrets: An Insider's Chronicle of the Russian Chemical Weapons Program. Outskirts Press. ISBN 978-1-4327-2566-2.
- ^ Vásárhelyi, Györgyi; Földi, László (2007). "History of Russia's chemical weapons" (PDF). AARMS. 6 (1): 135–146. Archived from the original (PDF) on 2018-03-14.
- ^ Halámek, E.; Kobliha, Z. (2011). "Potenciální Bojové Chemické Látky". Chemické Listy. 105 (5): 323–333.
- ^ Peplow, Mark (19 March 2018). "Nerve agent attack on spy used 'Novichok' poison". Chemical & Engineering News. 96 (12): 3. Retrieved 16 March 2018.
- ^ "Russian spy: What are Novichok agents and what do they do?". BBC News. 19 March 2018.
- ^ Carroll, Oliver (6 July 2018). "Novichok inventor on Amesbury poisoning: 'I completely understand panic of those living in Salisbury'". The Independent. Archived from the original on 2018-07-05. Retrieved 26 May 2020.
- ^ Harvey, Steven P.; McMahon, Leslie R.; Berg, Frederic J. (2020). "Hydrolysis and enzymatic degradation of Novichok nerve agents". Heliyon. 6: e03153. Bibcode:2020Heliy...603153H. doi:10.1016/j.heliyon.2019.e03153. PMC 7002793. PMID 32042950.
- ^ Gertz, Bill (4 February 1997). "Russia dodges chemical arms ban". The Washington Times. Retrieved 26 May 2020.
- ^ Franca, Tanos C. C.; Kitagawa, Daniel A. S.; Cavalcante, Samir F. de A.; da Silva, Jorge A. V.; Nepovimova, Eugenie; Kuca, Kamil (January 2019). "Novichoks: The Dangerous Fourth Generation of Chemical Weapons". International Journal of Molecular Sciences. 20 (5): 1222. doi:10.3390/ijms20051222. PMC 6429166. PMID 30862059.
- ^ "S/1821/2019/Rev.1 Note by the Technical Secretariat GUIDANCE FOR STATES PARTIES ON ARTICLE VI DECLARATION OBLIGATIONS AND INSPECTIONS FOLLOWING ENTRY INTO FORCE OF CHANGES TO SCHEDULE 1 OF THE ANNEX ON CHEMICALS TO THE CHEMICAL WEAPONS CONVENTION" (PDF). 14 January 2020.
- ^ "Schedule 1". OPCW. Retrieved 2020-07-26.
- ^ Chalaris, Michail; Koufou, Antonios (2023-05-31). "Antoine Equation Coefficients for Novichok Agents (A230, A232, and A234) via Molecular Dynamics Simulations". Physchem. 3 (2): 244–258. doi:10.3390/physchem3020017. ISSN 2673-7167.
- ^ Hoenig, Steven L. (2007), Compendium of Chemical Warfare Agents, Springer, ISBN 978-0-387-34626-7
- ^ Ellison, D. Hank (2008), Handbook of Chemical and Biological Warfare Agents (Second ed.), CRC Press, ISBN 978-0-849-31434-6
- ^ Jeong, Keunhong; Choi, Junwon (2019). "Theoretical study on the toxicity of 'Novichok' agent candidates". Royal Society Open Science. 6 (8). Bibcode:2019RSOS....690414J. doi:10.1098/rsos.190414. PMC 6731729. PMID 31598242.
- ^ Kruglyak, Yu. L.; Malekin, S. I.; Martynov, I. V. (1972). "Phosphorylated oximes. XII. Reactions of 2-halophospholanes with dichlorofluoronitrosomethane". Zhurnal Obshchei Khimii. 42 (4): 811–814.
- ^ Raevskii, O. A.; Chapysheva, N. V.; Ivanov, A. N.; Sokolov, V. B.; Martynov, I. V. (1987). "Effect of Alkyl Substituents in Phosphorylated Oximes". Zhurnal Obshchei Khimii. 57 (12): 2720–2723.
- ^ Raevskii, O. A.; Grigor'ev, V. Yu.; Solov'ev, V. P.; Ivanov, A. N.; Sokolov, V. B.; Martynov, I. V. (1987). "Electron-Donor Functions of Ethyl Methylchloroformimino Methylphosphonate". Zhurnal Obshchei Khimii. 57 (9): 2073–2078.
- ^ Hank Ellison, D. (9 December 2022). Handbook of Chemical and Biological Warfare Agents, Volume 1: Military Chemical and Toxic Industrial Agents. CRC Press. ISBN 9781000484403.
