PF-04455242
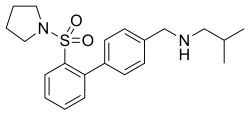 | |
| Clinical data | |
|---|---|
| Other names | PF-4455242; PF04455242; PF4455242 |
| Routes of administration | Oral[1] |
| Drug class | κ-Opioid receptor antagonist |
| ATC code |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C21H28N2O2S |
| Molar mass | 372.53 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
PF-04455242 is an experimental κ-opioid receptor (KOR) antagonist which was under development by Pfizer for the treatment of bipolar depression but was never marketed.[1][2][3] Its development was discontinued in early clinical trials.[2][3] It is taken by mouth.[1]
Pharmacology
[edit]The drug is a selective KOR antagonist and shows approximately 10- to 20-fold higher affinity for the KOR (Ki = 1–3 nM) over the μ-opioid receptor (MOR) (Ki = 10–64 nM) and has negligible affinity for the δ-opioid receptor (DOR) (Ki > 4,000 nM).[2][3] It is a "short-acting" or non-inactivating antagonist of the KOR (as opposed to irreversible antagonists like JDTic).[4] Although originally characterized as a KOR neutral antagonist however, subsequent research revealed in 2020 that PF-04455242 is actually only a moderately efficacious partial antagonist of the KOR (Imax ≈ 50%).[5] In any case, the drug reversed the analgesic and prolactin-elevating effects of the KOR agonist spiradoline in animals, showed efficacy in animal models predictive of antidepressant activity, and reversed stress-induced reinstatement of cocaine-seeking behavior.[2][3] However, PF-04455242 also showed a variety of other weak off-target activities.[2][3]
Clinical studies
[edit]PF-04455242 reached phase 1 clinical trials for bipolar depression prior to the discontinuation of its development in 2010.[1][3] Its development was discontinued upon unfavorable toxicological findings in animals that had been exposed to the drug for 3 months.[2][3] Along with JDTic, which was also discontinued due to toxicity findings early in clinical trials, PF-04455242 was one of the first KOR antagonists to be developed for potential treatment of psychiatric disorders.[2] It was in phase 1 trials by 2009[1] and was first described in the scientific literature by 2010.[6][7][8]
See also
[edit]References
[edit]- ^ a b c d e "PF 4455242". AdisInsight. Springer Nature Switzerland AG. 29 September 2010. Retrieved 28 September 2024.
- ^ a b c d e f g Li W, Sun H, Chen H, Yang X, Xiao L, Liu R, et al. (2016). "Major Depressive Disorder and Kappa Opioid Receptor Antagonists". Translational Perioperative and Pain Medicine. 1 (2): 4–16. PMC 4871611. PMID 27213169.
- ^ a b c d e f g Urbano M, Guerrero M, Rosen H, Roberts E (May 2014). "Antagonists of the kappa opioid receptor". Bioorganic & Medicinal Chemistry Letters. 24 (9): 2021–2032. doi:10.1016/j.bmcl.2014.03.040. PMID 24690494.
- ^ Melief EJ, Miyatake M, Carroll FI, Béguin C, Carlezon WA, Cohen BM, et al. (November 2011). "Duration of action of a broad range of selective κ-opioid receptor antagonists is positively correlated with c-Jun N-terminal kinase-1 activation". Molecular Pharmacology. 80 (5): 920–929. doi:10.1124/mol.111.074195. PMC 3198912. PMID 21832171.
- ^ Margolis EB, Wallace TL, Van Orden LJ, Martin WJ (2020). "Differential effects of novel kappa opioid receptor antagonists on dopamine neurons using acute brain slice electrophysiology". PLOS ONE. 15 (12): e0232864. doi:10.1371/journal.pone.0232864. PMC 7771853. PMID 33373369.
Although PF-04455242 is reported to be a full antagonist in heterologous systems [36], we found that it only partially blocked the U-69,593 responses in the electrophysiology assay (Fig 1B). We observed a maximal blockade asymptoting towards 45% of the baseline U-69,593 responses by 100 nM PF-04455242. The concentration of PF-04455242 that produced half of the maximum effect for this antagonist is 6.7 ± 15.1 nM. These data indicate that PF-04455242 is a partial antagonist in this tissue. [...] PF-04455242 showed some unexpected results in the characterization studies performed here compared to previously described pharmacological properties [27, 36]. First, we found it to only have partial antagonist action, with maximal blockade of the U-69,593 response plateauing at approximately 50%. [...] Together, these observations indicate PF-04455242 is quite different from a neutral KOR selective antagonist.
- ^ Jacobsen L, Banerjee A, Byon W, Huang Y, Carson RE, Tomasi G, et al. (2010). "Central receptor occupancy and pharmacodynamic action of PF-04455242, a high affinity, selective kappa opioid receptor antagonist, in humans". Society for Neuroscience Abstract Viewer and Itinerary Planner. 40.
- ^ Verhoest PR, Basak AS, Parikh V, Hayward M, Kauffman GW, Paradis V, et al. (August 2011). "Design and discovery of a selective small molecule κ opioid antagonist (2-methyl-N-((2'-(pyrrolidin-1-ylsulfonyl)biphenyl-4-yl)methyl)propan-1-amine, PF-4455242)". Journal of Medicinal Chemistry. 54 (16): 5868–5877. doi:10.1021/jm2006035. PMID 21744827.
- ^ Grimwood S, Lu Y, Schmidt AW, Vanase-Frawley MA, Sawant-Basak A, Miller E, et al. (November 2011). "Pharmacological characterization of 2-methyl-N-((2'-(pyrrolidin-1-ylsulfonyl)biphenyl-4-yl)methyl)propan-1-amine (PF-04455242), a high-affinity antagonist selective for κ-opioid receptors". The Journal of Pharmacology and Experimental Therapeutics. 339 (2): 555–566. doi:10.1124/jpet.111.185108. PMID 21821697.
