Wikipedia talk:WikiProject Elements/Archive 40
| This is an archive of past discussions about Wikipedia:WikiProject Elements. Do not edit the contents of this page. If you wish to start a new discussion or revive an old one, please do so on the current talk page. |
| Archive 35 | ← | Archive 38 | Archive 39 | Archive 40 | Archive 41 | Archive 42 | → | Archive 45 |
Standard temperature and pressure
At the risk of reopening what we discussed two years ago: what exactly are we using for our definition of STP? In {{Periodic table}} we say 0 °C and 1 atm, which is what IUPAC says it should be, but the chemistry world has not actually come to such a consensus and a lot of our data is not given for those conditions (e.g. densities of the elements (data page) contains a lot of use of 20 °C and 25 °C). 0 °C also has the issue that at this temperature Sn is in the alpha form and not a metal, as Sandbh noted last time. If memory serves we were using 25 °C for a while, so this may lead to a bit of confusion.
What brought this on this time is that R8R and I found a better figure for the melting point of Fr – but that estimate is 8.0 °C, so for the first time it actually makes a difference which definition of STP we go for. (Previously we didn't have any elements melting or boiling between 0 °C and 25 °C.) I was alerted to this by Incnis Mrsi updating the infobox here so that phase read "liquid or solid". Double sharp (talk) 14:18, 7 July 2019 (UTC)
- For the record, I, too, thought that STP was actually 20 °C, not 0. I suppose that we seem to be keen on following IUPAC whenever it is a good idea or not, and I think consistency is a good idea in general, so we may go with them. However, regardless of what we decide, I think it would be a good idea to change "STP" in the infobox to "STP (0 °C, 100 kPa)" (or whatever values we settle on).--R8R (talk) 15:05, 7 July 2019 (UTC)
- "which definition of STP": that abbreviation is solely for Standard conditions for temperature and pressure (by IUPAC), and it says 0 °C and 100 kPa. Also there is Standard state, and it can be different: "commonly 25 °C, less commonly 0 °C — there goes the standard. {{Chembox}} notes that in general "data is at standard state, 25 °C, unless otherwise specified". There also is the NIST NTP (20 °C).
- It is not for us to choose which standard we use, it should be defined in the source, and we should reproduce that. We could strive for using the STP values when available, and clarify when the sources do not use it, and explain the Sn, Fr situation (which is quite relevant IMO). When a data set like densities is measured with a different standard, that one should be added (to the data row). Anyway, I don't think saying "STP, 25 °C" can be correct ever. -DePiep (talk) 15:39, 7 July 2019 (UTC)
- (edit conflict) Interesting. I always understood STP to be 0 °C, which is consistent with the IUPAC definition; I would support such a change in the infoboxes simply for consistency, per R8R. However, regardless of what temperature (STP or NTP) we decide on, inconsistent data will be a problem. For example, the CRC handbook gives densities at 25 °C and magnetic susceptibility at 12-27 °C (285-300 K), while Wolfram gives them at STP and seems pretty reliable, but STP is not explicitly defined and I'm not sure if I can safely assume that 0 °C is used. ComplexRational (talk) 15:54, 7 July 2019 (UTC)
- Whoops… sorry, I was ignorant about the IUPAC standard. Incnis Mrsi (talk) 22:00, 7 July 2019 (UTC)
- Could someone update Melting points of the elements (data page) and List of chemical elements for Fr? I don't have the source at hand. -DePiep (talk) 08:53, 8 July 2019 (UTC)
 Done Double sharp (talk) 04:47, 9 July 2019 (UTC)
Done Double sharp (talk) 04:47, 9 July 2019 (UTC)- In the Fr case, should the infobox change or remove the "(at STP)" notion in the lefthand (label) side? -DePiep (talk) 08:57, 8 July 2019 (UTC)
- Could someone update Melting points of the elements (data page) and List of chemical elements for Fr? I don't have the source at hand. -DePiep (talk) 08:53, 8 July 2019 (UTC)
- As for tin… technically it can be metal at 0 °C (albeit the β form is thermodynamically unstable), look at “115 nΩ⋅m” in {{Infobox_tin}} – I guess it’s about β-tin, but why there are no data on the stable at 0 °C grey allotrope? Incnis Mrsi (talk) 22:00, 7 July 2019 (UTC)
- re
technically it can be metal at 0 °C
-- good, please do describe it, but that is not an STP nor OP issue. OP askes "what is STP &tc", not the temperature dependent effects you refer to. -DePiep (talk) 22:10, 7 July 2019 (UTC)- The resistivity Incnis Mrsi quotes for Sn is for 0 °C indeed (though WebElements gives 110 at 20–25 °C instead, according to that page). Of course you can have metallic β-Sn at 0 °C, as the transition is slow (well, until you get to something like −40 °C), but in general the standard state is supposed to be for the most stable allotrope, which is only true for Sn if you take the higher standard temperature. (Though we already have P as white phosphorus instead of the thermodynamically stable black form, so in some sense this is already moot.) Chemistry of Tin (edited by P. J. Smith) gives for resistivity 12.6 μΩ cm (alpha at 20 °C) and 300 μΩ cm (beta at 0 °C), but something seems quite obviously wrong with those figures. I suppose the conflicts are unavoidable, but we should probably at least state what conditions our figures are for, and perhaps make it clear if we want to say that Fr is liquid or solid at STP. Double sharp (talk) 03:55, 8 July 2019 (UTC)
- Grey tin is a zero-gap semiconductor, unlike e.g. germanium. Its conductivity should increase very strongly with temperature, hence 12.6 μΩ cm for 20 °C isn’t an obvious mistake. Incnis Mrsi (talk) 08:02, 8 July 2019 (UTC)
- While grey tin behaves like a semiconductor it is nevertheless regarded as a semimetal (with a zero band gap, as you say). There is an explanation of what's going on, here. Sandbh (talk) 02:44, 9 July 2019 (UTC)
- I see; thank you both for your explanations! Double sharp (talk) 15:44, 5 August 2019 (UTC)
- While grey tin behaves like a semiconductor it is nevertheless regarded as a semimetal (with a zero band gap, as you say). There is an explanation of what's going on, here. Sandbh (talk) 02:44, 9 July 2019 (UTC)
- Grey tin is a zero-gap semiconductor, unlike e.g. germanium. Its conductivity should increase very strongly with temperature, hence 12.6 μΩ cm for 20 °C isn’t an obvious mistake. Incnis Mrsi (talk) 08:02, 8 July 2019 (UTC)
- The resistivity Incnis Mrsi quotes for Sn is for 0 °C indeed (though WebElements gives 110 at 20–25 °C instead, according to that page). Of course you can have metallic β-Sn at 0 °C, as the transition is slow (well, until you get to something like −40 °C), but in general the standard state is supposed to be for the most stable allotrope, which is only true for Sn if you take the higher standard temperature. (Though we already have P as white phosphorus instead of the thermodynamically stable black form, so in some sense this is already moot.) Chemistry of Tin (edited by P. J. Smith) gives for resistivity 12.6 μΩ cm (alpha at 20 °C) and 300 μΩ cm (beta at 0 °C), but something seems quite obviously wrong with those figures. I suppose the conflicts are unavoidable, but we should probably at least state what conditions our figures are for, and perhaps make it clear if we want to say that Fr is liquid or solid at STP. Double sharp (talk) 03:55, 8 July 2019 (UTC)
- re
I understand IUPAC's position is that: "It must be stressed…that there is no reason why another value…should not be adopted for the reference temperature, so long as the author clearly states what has been done." SATP is standard ambient pressure and temperature, which is 25°C and 100 kPa. It does away with the tin issue. Sandbh (talk) 02:35, 9 July 2019 (UTC)
- I get the impression that this is not solved nor clarified. -DePiep (talk) 20:44, 9 August 2019 (UTC)
- Well, any attempt to put down a standard for most of our infobox values is doomed, because we have to follow the sources, and if you consult two sources you will probably find at least three standards. So in most cases I would suggest to explicitly say in our infoboxes what standard conditions are being used, as I think we already do on our data pages that are the reference for those infoboxes. For standard state, since this is the most straightforward, I would suggest adopting a room-temperature standard like 20 °C or 25 °C, because it is what our readers are probably expecting by now and avoids awkward questions about Sn. So Fr can be the third liquid. ^_^ Double sharp (talk) 05:00, 10 August 2019 (UTC)
- I get the impression that this is not solved nor clarified. -DePiep (talk) 20:44, 9 August 2019 (UTC)
List of standards
Lets collect the existion concepts. Worth an article like Standard states by pressure and temperature Standards in "standard" pressure and temperature.
- STP Standard conditions for temperature and pressure, defined by IUPAC. ("273.15 K and 105 Pa")
- Standard state -- not a definition, just a concept. ({{Chembox}} to be checked).
- SATP[1] mentioned by Sandbh, above, but is not in the source?
- -DePiep (talk) 22:14, 16 July 2019 (UTC)
- SATP: “Table of chemical thermodynamic properties”, National Bureau of Standards (NBS), Journal of Physics and Chemical Reference Data, 1982, Vol. 11, Supplement 2. Sandbh
Gold nanoseaweed
Here. Sandbh (talk) 02:21, 8 August 2019 (UTC)
- Cool! ^_^ Double sharp (talk) 05:09, 10 August 2019 (UTC)
Sidebar_periodic_table
Questions about {{Sidebar_periodic_table}} changes are posed at WT:CHEM: Wikipedia_talk:WikiProject_Chemistry#Sidebar periodic table. -DePiep (talk) 20:30, 11 August 2019 (UTC)
Theoretical instability of nuclides heavier than 92Zr
A few months ago, there was a discussion here regarding the conclusion that nuclides heavier than 92Zr are theoretically capable of spontaneous fission. While it is possible to mathematically justify this in terms of mass-energy conservation (using masses from {{NUBASE2016}}), no reliable sources directly assert this conclusion. To me, it appears that this conclusion is original research, though Magic9mushroom raises a fair point that WP:CALC supports the inclusion of this assertion as the result of a routine calculation. Seeing as WP:CALC refers to consensus on the quality and usability of the calculations, and I am not aware of any existing one, I'm not sure where to draw the line between CALC and OR (and thus the inclusion or removal of this content).
Here is the original question: User talk:ComplexRational#Your removal of "original research" from nuclear physics articles
@Double sharp, DePiep, YBG, Sandbh, R8R, and Utopes: Thoughts? ComplexRational (talk) 12:53, 13 August 2019 (UTC)
- I generally agree with your assertion that WP:CALC requires that calculations be to apply, obvious, indisputably correct, and can be easily drawn from the sources}. However, I'm unfamiliar with the topic matter here. Could someone walk me through it? What exactly to the WP:RS say? What is the calculation that is required? YBG (talk) 19:26, 13 August 2019 (UTC)
- The main concern here is that no RS seems to draw the conclusion that nuclides heavier than 92Zr are unstable. This was originally noted in many isotopes of X pages, though no citations were found and it was subsequently removed from some articles.
- As is mentioned in the post on my talk page, a nuclide is theoretically unstable when the decay products have less total energy than the nuclide itself, i.e. that the decay releases energy. In this example, the energy of nuclides (e.g. sourced to {{NUBASE2016}}) is analyzed and it is found, for example, that the decay of 93Nb into 46Ca and 47Sc releases energy, because the total energy of 93Nb exceeds the summed energies of these two daughter nuclides. At the same time, similar calculations find that no lighter nucleus is capable of such decay. Hence, this calculation (if Eparent > Eproducts, then decay is energetically allowed) seems to imply that this decay can spontaneously occur. No reliable source makes note of this, however, and I am also not sure if there are other factors that any publications may consider. The question, thus, is if the outcome of this calculation is sufficient to fulfill CALC, or if it is OR and should be removed. ComplexRational (talk) 19:40, 13 August 2019 (UTC)
- I should note that 93Nb is not the lightest nuclide energetically capable of splitting. It is the lightest observationally stable nuclide so capable (8Be splits into two alpha particles, though you can argue forever about whether that's alpha decay or SF; in any case, with a half-life under a femtosecond it's not observationally stable).
- The fundamental issue here is that "a form of decay is theoretically possible for a given nucleus if and only if reactant mass > product mass" is first-year nuclear physics and as such assumed knowledge in all modern nuclear physics papers. The mentions of that law itself over on Radioactive decay aren't cited and that needs to be fixed (IIRC Halliday/Resnick first-year physics textbook mentions it, although I don't have my own copy on-hand; I recall it mentioned in the nuclear chemistry section of several first-year chemistry textbooks as well), but the deduction from that law and a given set of nuclear masses is not in dispute (the exception being if the proposed form of decay itself is not known to be possible, such as for neutrinoless double beta decay; including that as possible without a source would definitely be inappropriate).
- I suppose the question is whether something that's a matter of course for those with a university education in the matter is considered sufficiently basic and indisputable to hit the common-sense/CALC exception to NOR. I say "yes". Magic9mushroom (talk) 03:21, 14 August 2019 (UTC)
- I think the answer is simple. You would use something like, "no nuclide with Z>40 and A>92 has a lower energy than a sum of energies of some lighter nuclides, whose total proton and neutron counts would equal those of the heavier nuclide;[ref NUBASE] in theory, this suggests they could be susceptible to SF, even if no such explicit claims have been made for some of these nuclei." Gives as much information and attributes it as properly as possible. This is the type of the resulting phrase I would expect to see, though exact content and phrasings are subject to discussion.--R8R (talk) 13:04, 15 August 2019 (UTC)
- This doesn't answer the question of what to put on the isotope list pages. Originally they were listed as "theoretically capable of spontaneous fission"; this has been removed from many of them. Also, your suggestion uses a bunch of weasel words to inject doubt despite no doubt existing here; I'm not seeing why this is a good thing. Magic9mushroom (talk) 05:42, 16 August 2019 (UTC)
- Indeed, any footnote in the isotopes pages needs to be a concise summary of this or any other answer to the question of CALC exempting this from NOR. This type of statement may be more useful in a broader nuclear physics article (stable nuclide and spontaneous fission come to mind). However, it is true that no explicit claims have been made, so regardless of the exact wording chosen, the text cannot imply that such predictions do exist. ComplexRational (talk) 14:02, 16 August 2019 (UTC)
- I am re-reading my own writing and I don't see anything that could be considered weasely here (although, as I said, wordings are up for discussion). Could you clarify that to me?
- My point is that if some calculations are routinely simple, then it can't hurt to mention that something is not a source-based data, but a routine calculation based on source data. So yes, you can use the outcome, for categorization as you mention, for instance, provided you specify that properly.--R8R (talk) 15:31, 16 August 2019 (UTC)
- I think it is clearer now. Would this amount to a footnote along the lines of "As this include has a greater mass/energy (one of them) than some lighter nuclides,[NUBASE] it is theoretically capable of spontaneous fission"?
- While this or a similar wording is sound, I still feel it's too clunky for footnotes, and is better off left only in overview articles or a footnote of a footnote (I.e. capable of SF has a footnote explaining the calculation). A more thorough explanation of the lack of precise predictions is necessary; as such, I feel that it is better done in prose, where it may be more easily clarified that this is CALC and (ideally) not SYNTH. ComplexRational (talk) 19:56, 16 August 2019 (UTC)
- Indeed, any footnote in the isotopes pages needs to be a concise summary of this or any other answer to the question of CALC exempting this from NOR. This type of statement may be more useful in a broader nuclear physics article (stable nuclide and spontaneous fission come to mind). However, it is true that no explicit claims have been made, so regardless of the exact wording chosen, the text cannot imply that such predictions do exist. ComplexRational (talk) 14:02, 16 August 2019 (UTC)
- This doesn't answer the question of what to put on the isotope list pages. Originally they were listed as "theoretically capable of spontaneous fission"; this has been removed from many of them. Also, your suggestion uses a bunch of weasel words to inject doubt despite no doubt existing here; I'm not seeing why this is a good thing. Magic9mushroom (talk) 05:42, 16 August 2019 (UTC)
- I think the answer is simple. You would use something like, "no nuclide with Z>40 and A>92 has a lower energy than a sum of energies of some lighter nuclides, whose total proton and neutron counts would equal those of the heavier nuclide;[ref NUBASE] in theory, this suggests they could be susceptible to SF, even if no such explicit claims have been made for some of these nuclei." Gives as much information and attributes it as properly as possible. This is the type of the resulting phrase I would expect to see, though exact content and phrasings are subject to discussion.--R8R (talk) 13:04, 15 August 2019 (UTC)
- User:R8R, what I'm getting at is that your original suggestion uses "suggests" and "could be", which creates the appearance of doubt where there isn't any. If SF would release energy, it is theoretically capable of SF.
- User:ComplexRational, I'm not sure why a note that it is CALC and not SYNTH would belong in the mainspace of the isotope-list articles, as policies are not part of content. On a talk page, yes. On the Stable nuclide page, the reasoning should be explained (although Wikipedia policies shouldn't). But the footnotes on the data pages don't need it. Magic9mushroom (talk) 12:24, 17 August 2019 (UTC)
- I apologize for the ambiguity; of course policies will not be explained in article space. What I meant is that there would be an explanation in normal prose that is both a clear presentation to readers and eliminates the possibility of interpretation as OR. I also felt that such an explanation might be too long for a footnote, but there may be a way to tweak it. ComplexRational (talk) 12:54, 17 August 2019 (UTC)
Categories and colors
Look:
- https://en.wikipedia.org/w/index.php?title=Template:Infobox_aluminium&action=history&offset=2019081312
- https://en.wikipedia.org/w/index.php?title=Template:Infobox_element&action=history&offset=2019081312
- https://en.wikipedia.org/w/index.php?title=Template:Element_color&action=history&offset=2019081312
While DePiep may cite some plausible rationale for the first two cases, {{element_color}} is not far from WP:OWN. Again, I added “p-block metal” as an alias for “post-transition metal” – they largely overlap. Who can be offended by an alias? Incnis Mrsi (talk) 12:38, 13 August 2019 (UTC)
- Not "offended". The category names as used are the result of some very thorough discussions (see box in top: "Metalloids and categorisation", 2013, Archive 15 and "Reclassifying_the_nonmetals", 2017/18). Also, they are preferably used consistently throughout this enwiki (while more detailed exceptions and variants can be described locally, i.e., in a relevant article).
- Using an alias at least reduces consistency, plus of course, if it is just an alias, why bother? Given the categorisation as applied on this enwiki, creating a separate category for a single element (while still being just an alias) does not look clarifying or helpful.
- Even stronger: the categories are listed in {{Infobox element/symbol-to-category}}, so as to improve consistency (maintained centrally). Making exceptions (in the infobox no less) is counterproductive. So far, the infobox handles all situations well, even SHE's and the theoretical elements.
- Anyway, if such an alias is to be introduced, that should be achieved through proposal & discussion. -DePiep (talk) 13:18, 13 August 2019 (UTC)
- If such an alias is to be introduced, that should be achieved through WP:consensus, which has not necessarily to be an active consensus. I added “p-block metal”, DePiep opposes it… but for which reason? There is a p-block metal aluminium which is technically not a post-transition metal, and the article’s body never claims (let alone for citations) it is post-transition. In reverse, group 12 elements are classified as post-transition metals but lie in d-block. For the remainder of both classes, they overlap and IMHO may be united, but not at expense of calling Al a “post-transition metal”. Incnis Mrsi (talk) 13:34, 13 August 2019 (UTC)
… but for which reason?
-- Those reasons are in my post above. -DePiep (talk) 13:45, 13 August 2019 (UTC)- Again, why there should not be an alias for the same category? “Creating a separate category” begs the question what is separation, and generic fluff about “to improve consistency (maintained centrally), [not] making exceptions” etc. must be dismissed as I already explained why there should be an alias. Incnis Mrsi (talk) 14:02, 13 August 2019 (UTC)
- There can be aliases. And they be treated as alias: a secondary name for the same concept (and so added in appropriate articles only). But the way you present it, it is a different name, used instead of (i.e., replacing) the original one and so creating a new category. Again, a replacement might be useful but only after consensus. BTW I did not find a response to the 'consistency' remark I made. (I note that in your second diff, there is this editsummary that did not point at all to any renaming issue, instead it looked like a systematical edit. That wrongfooted me). -DePiep (talk) 16:09, 13 August 2019 (UTC)
- wikt:renaming? Sorry, I’m dazed by the word. It is a misspell or a reference to the controversial post-transition class? 16:50, 13 August 2019 (UTC)
- As for “consistency”, the template already contains aliases, namely plurals. Technically—for wiki code—there is no difference between post-transition metals and p-block metal as between two instances of a deviation. The difference is staunchly semantical. Incnis Mrsi (talk) 16:57, 13 August 2019 (UTC)
- There can be aliases. And they be treated as alias: a secondary name for the same concept (and so added in appropriate articles only). But the way you present it, it is a different name, used instead of (i.e., replacing) the original one and so creating a new category. Again, a replacement might be useful but only after consensus. BTW I did not find a response to the 'consistency' remark I made. (I note that in your second diff, there is this editsummary that did not point at all to any renaming issue, instead it looked like a systematical edit. That wrongfooted me). -DePiep (talk) 16:09, 13 August 2019 (UTC)
- Again, why there should not be an alias for the same category? “Creating a separate category” begs the question what is separation, and generic fluff about “to improve consistency (maintained centrally), [not] making exceptions” etc. must be dismissed as I already explained why there should be an alias. Incnis Mrsi (talk) 14:02, 13 August 2019 (UTC)
- It is not unheard of to call Al a post-transition metal: see cite 78 on that article. While it demands special pleading as there are no transition elements before Al, aluminium surely behaves a lot like its heavier congeners starting with Ga which are post-transition by definition, so the abuse is reasonable (and can be handwaved by noting that group 13 at least comes to the right of the transition metals). We can argue all day about what the categories should contain anyway – the period 4 post-transition metals may include {Cu, Zn, Ga, Ge, As, Se}, depending on where the author you are reading thinks "transition" and "metal" most conveniently start and end for his or her purposes – and at the end of the day it is not really going to illuminate anything much by itself if we stick to just quibbling about terminology. What I'd like to do is: first, give the consistent convenient elementary answer for categorisation, call Al a post-transition metal to be consistent with the group, and then add a footnote saying that strictly speaking it is not but some authors do not use the term strictly. We can then actually get to the whole point of categorisation and discuss in the article what this difference means, viz. that while Al is a weak metal like its post-transition congeners Ga, In, and Tl, aluminium has a hard [Ne] noble gas core that causes it to have a strong affinity to oxygen and a consequently overly negative standard electrode potential (as Droog Andrey remarks in archive 33 it is not actually very electropositive), just like beryllium.
- On these grounds I have added cite 78 from post-transition metal to
{{Infobox aluminium}}, as well as a brief note stating that some authors exclude it on the grounds that Al is not actually after any transition metals. I have been planning for a rewrite of the chemistry section of Al for a few weeks now and will expound on what Al's unique situation as a p-block but not strictly post-transition metal actually means chemically there. Because of the important homology of Al with Ga, In, and Tl I would favour continuing with the "post-transition metal" categorisation while offering some explanation and sources for this use of the term. It may not be "technically correct" but you're not really going to find a better term for the whole category; having a proliferation of aliases without explanations would seem to encourage the misconception that "p-block metal" and "post-transition metal" are synonymous (they can be if you take group 12 as a transition group, but they don't have to be). Double sharp (talk) 13:59, 13 August 2019 (UTC)- @Double sharp: to what does it translate in respect to the alias in {{element color}}? Incnis Mrsi (talk) 14:07, 13 August 2019 (UTC)
- So far, there is no mainspace situation known where {{element color}} would be fed an alias. -DePiep (talk) 16:20, 13 August 2019 (UTC)
- @Incnis Mrsi: I do not favour an alias, as (1) an alias without explanation appears to the layman as a synonym, and (2) an alias with explanation can have its work done just as well by the original term with an explanation. I have added such a footnoted explanation to the place in period 3 element and
{{infobox aluminium}}where Al is called a post-transition metal. Double sharp (talk) 14:11, 13 August 2019 (UTC)
- @Double sharp: to what does it translate in respect to the alias in {{element color}}? Incnis Mrsi (talk) 14:07, 13 August 2019 (UTC)
- The nonmetals? Where in the hell is it currently relevant? Incnis Mrsi (talk) 14:02, 13 August 2019 (UTC)
- User:Incnis Mrsi I'm getting tired and distracted by your, errr, overenthousiastic posts. If you could turn those into a question, before hitting Save, one might find an answer. -DePiep (talk) 16:17, 13 August 2019 (UTC)
- Only now it occurs to me that in their OP, Incnis Mrsi wrote
While DePiep may cite some plausible rationale for the first two cases, {{element_color}} is not far from WP:OWN
[2] is an accusation (disguised as a suggestive smear) that is not baseed and not substantiated. And in my reply, I described the context clarifying even more. I strongly suggest that Incnis Mrsi strokes the smear. -DePiep (talk) 19:54, 13 August 2019 (UTC)- What is not substantiated? DePiep keeps aliases pleasant to DePiep—and their presence isn’t based on any kind of agreement—meanwhile deleting aliases which Incnis_Mrsi likes. How is it not OWN? I may mistake, but the incident currently looks namely in this light. Incnis Mrsi (talk) 05:26, 14 August 2019 (UTC)
- Your BF suggestion that I would WP:OWN the template. As I already expolained, the template is maintained based on consensus in this thread, and I linked two discussions (that took over 400 days each) that concluded such. Even before you posted this here, I already wrote in the es that your edit did not have consensus. So please strike your smear. -DePiep (talk) 05:37, 14 August 2019 (UTC)
- What the freaking BF – “bad faith”? No way: most cases or attempted ownership arise in good faith, yet ownership attitudes are discouraged. Incnis Mrsi (talk) 06:21, 14 August 2019 (UTC)
- Your BF suggestion that I would WP:OWN the template. As I already expolained, the template is maintained based on consensus in this thread, and I linked two discussions (that took over 400 days each) that concluded such. Even before you posted this here, I already wrote in the es that your edit did not have consensus. So please strike your smear. -DePiep (talk) 05:37, 14 August 2019 (UTC)
- What is not substantiated? DePiep keeps aliases pleasant to DePiep—and their presence isn’t based on any kind of agreement—meanwhile deleting aliases which Incnis_Mrsi likes. How is it not OWN? I may mistake, but the incident currently looks namely in this light. Incnis Mrsi (talk) 05:26, 14 August 2019 (UTC)
- Only now it occurs to me that in their OP, Incnis Mrsi wrote
- User:Incnis Mrsi I'm getting tired and distracted by your, errr, overenthousiastic posts. If you could turn those into a question, before hitting Save, one might find an answer. -DePiep (talk) 16:17, 13 August 2019 (UTC)
- If such an alias is to be introduced, that should be achieved through WP:consensus, which has not necessarily to be an active consensus. I added “p-block metal”, DePiep opposes it… but for which reason? There is a p-block metal aluminium which is technically not a post-transition metal, and the article’s body never claims (let alone for citations) it is post-transition. In reverse, group 12 elements are classified as post-transition metals but lie in d-block. For the remainder of both classes, they overlap and IMHO may be united, but not at expense of calling Al a “post-transition metal”. Incnis Mrsi (talk) 13:34, 13 August 2019 (UTC)
The question here appears to be "Should we have multiple synonyms in the template {{element color}}? To answer this, I ask another question: What are the benefits and detriments of having synonyms for template parameter values? It is beneficial to have synonyms for template parameter names and template parameter values in templates which have wide usage by a large number of editors who do not generally collaborate together. This allows different people to latch on to different parameter names or values which are most meaningful and memorable to them. The detriment is that it makes it much more complicated to search for the usage of specific parameter names or values when it becomes necessary to make some global changes. This is unavoidable with templates which have widespread usage - consider {{infobox}} or {{hatnote}}. But in this case we have a template which is used in a relatively small set of articles and used by a relatively small set of editors. This significantly reduces any benefit to synonyms. I would even go so far as to say that {{element color}} would be significantly improved if we were to eliminate all synonyms even the plurals. YBG (talk) 19:12, 13 August 2019 (UTC)
- While I could support the reasoning in broad lines, I disagree on your remark that this is about {{element color}}. {{element color}} is a metatemplate, and so by itself not directly visible. Its workings are derived from requirements for the visible, reader-end templates (like {{infobox element}} and {{periodic table}}). In short, the input can be a conformed category name, all fine. In this case, Incnis Mrsi introduced an alias for such an input in reader's view [3]. The main point is, that that alias is not vetted in a discussion (for example here). Thererfor the alias should not be used in mainspace, and so there is no requirement to have it added to metatemplates. IOW: first we need consensus about actually accepting and using the new name (why, when & how?), and from there meta-consequences can be drawn easily. Nicely, this is how WP:ELEMENTS works as long as I can remember. -DePiep (talk) 20:09, 13 August 2019 (UTC)
Hi @Incnis Mrsi: you wrote:
I added “p-block metal”, DePiep opposes it… but for which reason? There is a p-block metal aluminium which is technically not a post-transition metal, and the article’s body never claims (let alone for citations) it is post-transition. In reverse, group 12 elements are classified as post-transition metals but lie in d-block. For the remainder of both classes, they overlap and IMHO may be united, but not at expense of calling Al a “post-transition metal”. Incnis Mrsi (talk) 13:34, 13 August 2019 (UTC)
As DePiep noted, the current periodic table categories reflect consensus informed by 5+ years of discussion.
Yes, group 12 elements are PTM's that lie in the d-block. The IUPAC Red Book notes that although the group 3−12 elements are commonly referred to as the transition elements, the group 12 elements are not always included. And we know that the group 12 elements do not satisfy the IUPAC Gold Book definition of a transition metal.
I agree Al is a p-block metal; that group 12 are d-block metals; and that the post-transition metals are made up of d- and p-block metals.
I believe that having regard to the periodic table's most popular use, striving to show the complexity of the chemistry involved is more important than regimenting the periodic table into clean blocks. Sandbh (talk) 06:09, 16 August 2019 (UTC)
- Here Incnis Mrsi again changes the category for Al without consensus. They have been told and warned that this is incorrect editing. -DePiep (talk) 10:27, 17 August 2019 (UTC)
- What’s the problem: did the WikiProject state that in every navbox exactly the same wording should be used? Boron is a metalloid, gallium is a post-transition metal, and aluminium is a metal (unlike boron) and doesn’t have d10 (unlike gallium). In this context it’s perfectly reasonable to state that Al is a metal. Incnis Mrsi (talk) 10:42, 17 August 2019 (UTC)
- In short: yes. This WikiProject concluded that. It was described for you in this thread, including links to the discussions that reached the agreement. -DePiep (talk) 10:48, 17 August 2019 (UTC)
- (edit conflict) Sandbh makes an important point that chemical properties are most relevant for categorizing elements. While I agree that Al is a metal and that its exact categorization (p-block, post-transition, etc.) is disputed, we do not have a blanket metal category in the PT. If we classified Al as simply a metal in the navbox, it would be in a category of its own; this is the inconsistency that is not desired, and is the reason for labeling it as a post-transition metal. However, I'm wlling to discuss alternatives for the category name, if there are commonly accepted alternatives in which Al is not disputed. ComplexRational (talk) 10:55, 17 August 2019 (UTC)
- Which kind of alternative? Is the p-block metal link eligible? I am willing to settle on it, although deem it redundant as group 13 wholly is a p-group. Incnis Mrsi (talk) 11:06, 17 August 2019 (UTC)
- I’m not quite sure; p-block metal does not include Zn, Cd, and Hg (which are currently classified as post-transition metals). As a subset, it’s fine, but I wouldn’t consider renaming the whole category to p-block metal. ComplexRational (talk) 12:55, 17 August 2019 (UTC)
- We could extend the compromise I previously implemented at period 3 element and aluminium: label Al as a post-transition metal, but with an explanatory note. The note I added to the Al article says "As aluminium technically does not come after any transition metals in the periodic table, it is excluded by some authors from the set of post-transition metals. [cite] Nevertheless its weakly metallic behaviour is similar to that of its heavier congeners in group 13 gallium, indium, and thallium, which are post-transition metals by all definitions." I favour this because relabelling Al but not Ga, In, and Tl makes it look like its technically "pre-transition" status matters more for its chemistry than it really does. (Al and Ga are very similar.) Double sharp (talk) 13:14, 17 August 2019 (UTC)
- In that case, I'd keep the status quo in the navbox, and add the explanatory note (or a shortened version) as proposed. ComplexRational (talk) 19:36, 17 August 2019 (UTC)
- Look at WhatLinksHere/Template:Periodic table (group 13), guys. The only main-space page transclusing the template is exactly the same page for which “its weakly metallic behaviour similar to that of… gallium, indium, and thallium” is its immediate topic. Why to stuff the navbox with extra notes? Incnis Mrsi (talk) 19:55, 17 August 2019 (UTC)
- re
Why to stuff the navbox with extra notes?
: because your point (if I understand it well; you bring confusion with it too), your point is an exception to the grand category scheme, and a detail with that. So: we choose & keep the grand set of categories (names and principles), and carefully note exceptions & details in appropriate places. -DePiep (talk) 20:05, 17 August 2019 (UTC) - Because if we don't put a note in the navbox, people are going to have questions about whether "post-transition metal" can legitimately apply to Al like you did. Since these are legitimate questions, if we are going to carry on with using "post-transition metal" for Al (which makes the most chemical sense to avoid splitting it from Ga, In, and Tl), it behooves us to explain why this usage is not necessarily wrong and justify it. Double sharp (talk) 04:21, 18 August 2019 (UTC)
- re
- We could extend the compromise I previously implemented at period 3 element and aluminium: label Al as a post-transition metal, but with an explanatory note. The note I added to the Al article says "As aluminium technically does not come after any transition metals in the periodic table, it is excluded by some authors from the set of post-transition metals. [cite] Nevertheless its weakly metallic behaviour is similar to that of its heavier congeners in group 13 gallium, indium, and thallium, which are post-transition metals by all definitions." I favour this because relabelling Al but not Ga, In, and Tl makes it look like its technically "pre-transition" status matters more for its chemistry than it really does. (Al and Ga are very similar.) Double sharp (talk) 13:14, 17 August 2019 (UTC)
- I’m not quite sure; p-block metal does not include Zn, Cd, and Hg (which are currently classified as post-transition metals). As a subset, it’s fine, but I wouldn’t consider renaming the whole category to p-block metal. ComplexRational (talk) 12:55, 17 August 2019 (UTC)
- Which kind of alternative? Is the p-block metal link eligible? I am willing to settle on it, although deem it redundant as group 13 wholly is a p-group. Incnis Mrsi (talk) 11:06, 17 August 2019 (UTC)
The core of the issue is that in the literature, there is no consistency on a category name for the metals in groups 12 to 16. By consensus, we call them post-transition metals, since that term seemed to have the most academic credibility.
The term does not necessarily apply only to metals with d10 cores; Whitten et al. (2014 p. 1045) say it refers to the "the metals below the stepwise division of the periodic table in Groups 3A through 5A…These include aluminum [sic], gallium, indium, and thallium from Group 3A; tin and lead from Group 4A…". Here the authors are referring to the conventional form of the table, which is effectively universally used in the English world. Their mention of the metal-nonmetal line chimes with our categories, which are primarily categories of metallicity, showing a progression in metallic character from left to right:
[We used to show the Ln on top of the An, which is more accurate.]
The lead sentence of our article on the PTM simply says, "Post-transition metals are a set of metallic elements in the periodic table located between the transition metals to their left, and the metalloids to their right." This is a broader categorisation than the d10 requirement, provides an easier way of demarcating our table, and is consistent with our category legend.
Absent of any better proposal by Incnis Mrsi, I support the views of DePiep, Double sharp, and ComplexRational wrt to the current categorisation of Al as a PTM.
- Whitten KW et al. 2014, Chemistry, 10th ed., Brooks Cole/Cengage Learning, Belmont, CA
Sandbh (talk) 04:19, 18 August 2019 (UTC)
- My proposal would be to remove all transition/post-transition/p-block things from navboxes about groups. They show trends from period 2/3 to period 7, not trends from group 1 to group 17. It’s the text in the article (about the group) which should explain which kind of metals are encountered. Incnis Mrsi (talk) 09:40, 18 August 2019 (UTC)
- re Sandbh: Wow, what a post. -DePiep (talk) 22:50, 18 August 2019 (UTC)
I won't support a proposal that makes it harder to gain a quick appreciation of the subject matter i.e. by removing categories from navboxes about groups. Sandbh (talk) 23:34, 25 August 2019 (UTC)
Poly-/diatomic- nonmetals
See the last graphic in this item.
Did we get it right the first time? Uncanny.
Sandbh (talk) 12:48, 26 July 2019 (UTC)
- The last graphic seems to be based on words used in materials science papers, which explains some chemically strange findings (Fr is far away from the alkali metals because even though it is chemically like them it does not appear in the same sort of literature; the same thing happens with many radioactives). So I wouldn't give it that much weight. Double sharp (talk) 12:52, 26 July 2019 (UTC)
- Minor detail: it has a section title "Periodic_table v2.0" (sic). AFAIK, and YMMV, PT 2.0 was when the noble gases were added. 130 y ago. Quite pedantic. -DePiep (talk) 21:08, 27 August 2019 (UTC)
Looking at this again:
1. Their results likely draw on one of the biggest surveys of the literature ever attempted, for this kind of research.
2. The article says their database of [only] 3.3 million abstracts spanning 1922 to 2018, and drawing from 1,000 journals, was focussed on materials science, physics, and chemistry.
3. Key words were extracted using ChemDataExtractor ("Give it a journal article, and it will extract chemical names, properties, and spectra from the text so they can be imported into a database or spreadsheet.").
4. I agree there are a few anomalies like Fr for the very plausible reason you give. Is this not a case of "perfect is the enemy of good"?
5. Their results match with the JChemEd Kohonen map cited in our own archive 15. Anybody remember that one?
We've never had a comprehensive literature-based view of nonmetal categories, which is why we've always struggled doing a fair job with these. Now we have what is arguably the biggest perspective ever on the nonmetals, let alone the remarkable correlation with the rest of the periodic table.
If we confine our perspective on the periodic table to purely chemistry-based considerations, which we never have AFAIK, we'll being doing ourselves a disservice to our readers, not to mention the periodic table.
A 2019 editorial in Nature Materials ("Lessons from the periodic table") opined that:
"There has been a synergy between our evolving understanding of the periodic table and our understanding of materials. Element position within the periodic table and sustainability considerations plays a role in determining research activity and practical applications. The space in the periodic table for new materials combinations is vast. All elements will continue to be worthy of investigation."
The key words here are a "synergy" or a working together, as guided by the periodic table, and our "evolving" understanding of the latter.
Still, when nonmetals are categorised in the literature, the dichotomy of noble gases and reactive nonmetals is still prevalent. So I'll edit the nonmetal article by adding a citation for the Kohonen map and this latest effort, and I'll hoik up the league ladder standing for polyatomic/diatomic in the table of alternative categorisation schemes. And I'll see if I can reinstate the old table of comparative properties for polyatomic, diatomic, and monatomic nonmetals, further down the article, so as to add another perspective. Sandbh (talk) 07:15, 31 July 2019 (UTC)
- Placeholder. Double sharp (talk) 04:24, 17 August 2019 (UTC)
Check the position of astatine—deep in metal territory. Actually this is much more likely the case of At being associated with the radioactives. On a different note, the correlation with our own plot of electronegativity v melting point for the metals and metalloids is remarkable. Sandbh (talk) 23:20, 25 August 2019 (UTC)
- This reminds of the analogies between categories:
Metals Nonmetals Active (1–2; Ln, An) Diatomic Transition (most) Polyatomic Post-transition Weak (metalloids) Noble Noble gas (monatomic)
- Just as the transition metals represent a transition from the active metals to their right and the "poor" or post-transition metals to their left, so do the polyatomic nonmetals occupy a changeover zone between the diatomic nonmetals to their right and the weak nonmetals (metalloids) to the left.
- Usual disclaimer about boundary overlaps. For example, Al and the active metals; Ag as a post-transition metal; O as polyatomic (as ozone).
The left step periodic table (LST)
I’ve been looking at the LST for quite a while and...I don’t get why some folks prefer it:
| f1 | f2 | f3 | f4 | f5 | f6 | f7 | f8 | f9 | f10 | f11 | f12 | f13 | f14 | d1 | d2 | d3 | d4 | d5 | d6 | d7 | d8 | d9 | d10 | p1 | p2 | p3 | p4 | p5 | p6 | s1 | s2 | |||
| 1s | H | He | ||||||||||||||||||||||||||||||||
| 2s | Li | Be | ||||||||||||||||||||||||||||||||
| 2p 3s | B | C | N | O | F | Ne | Na | Mg | ||||||||||||||||||||||||||
| 3p 4s | Al | Si | P | S | Cl | Ar | K | Ca | ||||||||||||||||||||||||||
| 3d 4p 5s | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr | Rb | Sr | ||||||||||||||||
| 4d 5p 6s | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe | Cs | Ba | ||||||||||||||||
| 4f 5d 6p 7s | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn | Fr | Ra | ||
| 5f 6d 7p 8s | Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og | Uue | Ubn | ||
| f-block | d-block | p-block | s-block | |||||||||||||||||||||||||||||||
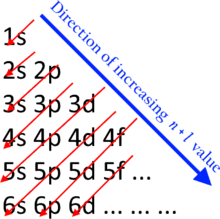
It has the following features: (a) it is more regular than the conventional table; (b) it has no gaps; (c) each element in every second row of each block starts a triad; (d) each element in each period has an n+l value matching the period number i.e. it matches the diagonal lines of the Madelung rule diagram (MRD); and (e) the first row anomaly becomes clearer i.e. s >> p > d > f.
(a) means the period lengths go 2, 2, 8, 8, 18, 18, 32, 32 whereas the conventional form goes 2, 8, 8, 18, 18, 32, 32;
(b) means e.g. there is no gap between Be and B;
(c) means e.g. that Tc, Re, and Bh form a triad;
(d) means e.g. that the n+l value of Ti, in period 5, is 3 + 2 = 5. Here, n is the principal quantum number (same as the orbital value of 3, in 3d); and l is the azimuthal quantum number, where s = 0; p = 1; d = 2; f =3). Sr, in the same period, is 5s = 5 + 0 = 5; and
(e) is nice.
Feature (d) doesn’t mean much since, like the MRD, the quantum numbers involved are based on idealised rather than actual differentiating electrons. For example, Zn is 3d = 3 + 2 = 5. Whereas in real life the differentiating electron in Zn = 4s = 4 + 0 = 4. In fact the MR is wrong in 20 places—it’s an approximation or idealisation, not a law carved in stone.
While the strongest objection to the LST is He over Be, that’s not so much of an issue. The electronegativity and ionisation energy of He can be extrapolated from the rest of the alkaline earth metals. The 1s2 electron configuration matches that of the rest of group 2. We know that He is capable of acting as a metal. One just needs to colour code it as a noble gas, on account of its closed shell.

Proponents of the LST say its regularity and elegance, as well as its symmetry (the four blocks fit naturally into a tetrahedron or, the LST can be rearranged into the ADOMAH form) mean that it is an optimal form of periodic table.
I say that:
- these features are derived from an idealisation rather than actual physical parameters i.e. idealised v actual quantum numbers;
- as such they mean nothing in terms of an optimal periodic table;
- while the electron configuration filling sequence has some irregularities one can still see a general regularity that accords with the MR diagram;
- the general regularity hints at a hidden symmetry in the applicable laws of nature; and
- this hidden (or broken) symmetry manifests as the asymmetric conventional form.
Here’s a relevant quote:
- While the laws of Nature, “are simple, symmetrical, and elegant, the real world isn’t. It’s messy and complicated…The reason is clear. We do not observe the laws of Nature: we observe their outcomes. Since these laws find their most efficient representation as mathematical equations, we might say that we see only the solutions of those equations not the equations themselves. This is the secret which reconciles the complexity observed in Nature with the advertised simplicity of her laws. Outcomes are much more complicated than laws; solutions much more subtle than equations. For, although a law of Nature might possess a certain symmetry, this does not mean that all the outcomes of the law need manifest that same symmetry.” (Barrow 2008)
It is as if the periodic table lies at the bottom of a wine bottle; the symmetry of the bottle’s base is clear from the top of the dimple in the centre, but it is hidden from any point in the valley surrounding the central dimple.* Assigning >special< significance to the LST (or the ADOMAH variation) on the basis of its elegance, regularity, symmetry, or coherence with the MRD, while well intentioned, is misguided and needlessly detracts from the clarity of the chemical relationships among the elements and their compounds.
- ∗ I borrowed this analogy from the EB article on Subatomic particles, Hidden symmetry section, by Sutton C, 2017

Possibly the next biggest objection to the LST is that it makes a train wreck of the left to right metal to nonmetal progression, and associated trends.
As Ball (2010) said:
- “I think it is a poor deal to trade subjective aesthetics (which clearly not everyone shares) for the long-standing traditional navigational axes that chemists use around the PT.”
Weighing up the features and drawbacks of the LST, I don’t understand how they can be reconciled.
The conventional form of the periodic table is about as good as any other; hence it is retained for want of a better alternative.
Can anyone shed light on the merits of the left-step table, such that it becomes preferred to the conventional form?
References
- Ball P 2010, comments to “The disappearing spoon” review, homunculus blog, viewed 19 May 2019, https://www.blogger.com/comment.g?blogID=26741618&postID=2980643491692822828&bpli=1&pli=1
- Barrow JD 2008, New theories of everything, Oxford University Press, Oxford, pp. 136–140
See also
- Ball P 2014, “Beauty ≠ truth”, Aeon, 19 May
- Stewart PJ 2018, “Tetrahedral and spherical representations of the periodic system”, Foundations of Chemistry, vol. 20, no. 2, pp. 111–120, open access
Sandbh (talk) 20:21, 14 June 2019 (UTC)
Comments by DePiep
First and quick replies: I like the Left step.
- It is Mendelevian. M's periodicity is in there, it's just reordering columns.
- It even supports shell filling better (s, p, d, f pattern). So while groups don't apply any more, the shells/blocks are regular (up to a point).
- It helps me, a layman, better understanding the orbitals & shells. After all, the PT is 2D while QM is 4D.
- -DePiep (talk) 00:53, 15 June 2019 (UTC)
- Thanks for that. I was interested to know what others thought about this table. I intend to submit an article for publication about it.
- It’s almost, but not quite, Mendelevian. If you arrange the elements in one long line then La shows the next recurrence of properties after Y. The table appears to ignore this example of the periodic law since it shows Lu, rather than La, under Y. I don’t want to turn this observation into an argument about group 3; I just think it’s interesting to note this apparent discrepancy.
- The groups are still there it’s just that the numbering goes 3 to 18, then 1 and 2. My reaction would be to wonder why the groups couldn’t be arranged from 1 to 18?
- On your third item, how do you feel about this diagram(?):
1s 2s 2p 3s 3p 4s 3d 4p 5s 4d 5p 6s 4f 5d 6p 7s 5f 6d 7p
- Sandbh (talk) 11:28, 17 June 2019 (UTC)
- re "how do you feel about this diagram": Just as correct (Z is climbing, valence periodicity kept, nice shell filling regularity), though it lost the elegance of the overall build-up (the steps). Not just elegance, the build-up is core to shell filling if I have it right. The reduction of step-up simplicity is a consequence of you trying to keep the original group order (valences I-VIII+0) present. However, IMO that is not needed & extra hard to explain. We'd better use two tables: "When we go by valences, we get the Mendeleevian 18-column PT [our current standard]; and when we go by shell filling we get Janet's left step. Both are most regular by their ordering principle.". -DePiep (talk) 17:23, 20 June 2019 (UTC)
- Sandbh (talk) 11:28, 17 June 2019 (UTC)
- For explaining the shell filing pattern, it's probably more helpful to have three of these:
Pattern 1 1s 2s 2p 3s 3p 4s 3d 4p 5s 4d 5p 6s 4f 5d 6p 7s 5f 6d 7p Pattern 2 Pattern 3 1s 1s 2s 2p 2s 2p 3s 3p 3s 3p 4s 3d 4p 4s 3d 4p 5s 4d 5p 5s 4d 5p 6s 4f 5d 6p 6s 4f 5d 6p 7s 5f 6d 7p 7s 5f 6d 7p
- P1 is the elements lined up in a row, according to Z. This is hard to grasp however one can point to a kind of recurrence among the s's, the p's, the d's and the f's. And it's interesting to note that s appears for the first time before p appears before d appears before f.
- P2 arranges P1 into something more compact, and that has the elegance of a build up.
- P3 spaces things out a bit so that the s's, the p's, the d's and the f's, are lined up. Hey presto, that is the shape of the modern PT, and you can see why there are gaps. There is still some irregularity, since the numbers going along each of the last four rows aren't the same. To explain this we can observe that the numbers in the s column and the p column are the same, and that the numbers in the f and d columns are lower because here electrons are being added to inner shells (the first inner shell in the case of the d's; and the second inner shell in the case of the f's, rather than outer shells as is the case with the s and p columns.
- P3, I think, serves both the purposes of valence, and shell filling.
- Here is the Janet pattern sequence:
Pattern 1
1s 2s 2p 3s 3p 4s 3d 4p 5s 4d 5p 6s 4f 5d 6p 7s 5f 6d 7p
Pattern 2J
1s
2s
2p 3s
3p 4s
3d 4p 5s
4d 5p 6s
4f 5d 6p 7s
5f 6d 7p 8s
- Hey presto, that is the shape of the LST.
- Some differences between the modern PT and the LST is that the modern PT preserves the group numbering sequence; the metal-nonmetal dichotomy; and reading from left to right. However the explanation of the modern PT takes one more step.
- Does it look easier to use the modern PT plus the three-pattern explanation, rather than two periodic tables? Sandbh (talk) 00:54, 24 June 2019 (UTC)
- The way I think about constructing the usual PT from the subshell sequence is that you align the blocks (pedagogically obvious) and start a new row when a new s-subshell is filled (chemically obvious). Then you make an exception for helium, to align this exceptional full subshell (chemically obvious on a level that the La/Lu and Ac/Lr differences aren't). The chemically obvious position of the noble gases and the "wall" between a final s-shell and the inner shells below it is why I think the usual PT is more effective than the Janet table. Double sharp (talk) 04:56, 24 June 2019 (UTC)
Comments by Double sharp
- Regarding the first point, I will restrict myself to two sentences to avoid making this about group 3 again ^_^: if you arrange the elements in one long line, then Sc shows the next recurrence of properties after Al, not Ga, and indeed Mendeleev expected it to take the position right under Al. So there is more to it than that, which I would summarise as saying that we try not to rock the boat by making weirdly-shaped blocks too much, unless not doing so would be the greater boat-rocking (helium, predicted superheavies far past Og). I would instead criticise the Janet table on the grounds that the way it is arranged ignores the fact that chemically speaking, the break does not come between different values of n+ℓ, but instead after a noble gas configuration is reached, because that cannot be chemically breached in "normal" compounds. (Unless relativistic effects contrive to make that happen for E119 and maybe E120, but those are surely not "normal".) Since the group numbers originally stem from valences, I think this criticism of the Janet table is in agreement with your statement about group numbering. Double sharp (talk) 13:37, 17 June 2019 (UTC)
- Re your first point: In the second 1871 version of his table Mendeleev showed two tranches for group III, i.e. Al-(eka-Al)-In-Tl; and Sc-Yt-Di. What to do after so-called Di was the puzzle due to the similarity of the other lanthanides. In the event he put Ce in group IV at which point the wheels started falling off. Anyway, Mendeleev was looking for the next applicable recurrence after Yt, which he mistook for Di (atomic weight 138), which actually turned out to be a mixture of Pr and Nd. He got the atomic weight well-enough right (La = 138.91), he just got the element wrong. Had he known La had the lowest atomic weight I expect he would have recognised it as the next applicable recurrence after Yt.
- On the weirdly-shaped blocks issue, I made a post about that a while back in archive 32; it's the one by Poliakoff on group 3, and his response that, "What we are interested in is what Nature is like not how easy it is to draw it."
- I recently found a similar line of thought that suggested to me that we are looking for or expecting elegance in the wrong place:
- "[While] the laws of Nature are simple, symmetrical, and elegant, the real world isn’t. It’s messy and complicated…The reason is clear. We do not observe the laws of Nature: we observe their outcomes. Since these laws find their most efficient representation as mathematical equations, we might say that we see only the solutions of those equations not the equations themselves. This is the secret which reconciles the complexity observed in Nature with the advertised simplicity of her laws. Outcomes are much more complicated than laws; solutions much more subtle than equations. For, although a law of Nature might possess a certain symmetry, this does not mean that all the outcomes of the law need manifest that same symmetry.”(Barrow 2008, p. 138)
- That is to say, it is the interactions of these laws that lead to the blemishes and anomalies of the periodic table.
- Barrow JD (2008), New theories of everything: The quest for ultimate explanation, Oxford University Press, Oxford
- Sandbh (talk) 07:23, 19 June 2019 (UTC)
Group 3 revisited
- On the first point: I'd take Mendeleev's written period numbers as the guide, so Sc is the next element after Al. And the reason why I would consider it such is that otherwise we'd have to assume he considered Mn to be the next recurrence of properties after F, when Cl seems to be a much more obvious candidate. [added after YBG's and R8R's replies: Remember, for Mendeleev all the period 2-period 3 pairs were drawn with each in a different tranche, so Sc was eka-boron while Ga was eka-aluminium. Double sharp (talk) 04:05, 20 June 2019 (UTC)]
- On the blocks issue: to me, the periodic table as shown is not purely a law, it is the periodic law illustrated in a way that makes the most sense pedagogically and practically. (Or if you like, the interaction of multiple such laws that usually get all subsumed under the roof of periodicity.) As such, I think part of it is indeed about how we choose to draw it. Helium is helium (*) regardless of how we draw it, but depending on what we want to show we may want to put it somewhere or somewhere else. It just seems to me that if you want a more or less optimal table, you want the one that fits as many use cases as possible, and then surely He over Ne wins out.
- (*) I am sure R8R will come to gently scold me like he did last time for ignoring that we made up the grouping of 3He and 4He and the other six mayflies we made ourselves, but at least we can say that the proton number 2 that brings them together is fairly straightforward. Double sharp (talk) 15:46, 19 June 2019 (UTC)
- @Double sharp: I'm intrigued by your comments
depending on what we want to show
andas many use cases as possible
. It would be interesting to list the different things one might wish to show, and then for each individual "thing to show", note the PT implications and perhaps the optimal PT for that particular case. Having a specific list might help us better understand the overall structure of the Whac-A-Mole game we are playing. YBG (talk) 19:46, 19 June 2019 (UTC)- I have noted this interesting quote: "What we are interested in is what Nature is like not how easy it is to draw it." And it seems like it's so great on the first glance but I think this is either insincere or not very profound (I'm leaning towards the second alternative). Because you will inevitably turn to drawing. And our periodic table is, inevitably, also an approximation. Our way to describe the reality. If, say, you really establish that Sc-Y-La-Ac is better than Sc-Y-Lu-Lr with the difference in the sixth decimal place, does -La-Ac really make a better alternative to the simpler almost-just-as-good -Lu-Lr, given that -La-Ac is still inevitably merely our concept to describe the reality rather than the reality itself? (Was that difference in the sixth decimal place really on the decisive criterion?) I really don't think there's a good answer to that in the sense of having a better conceiving of the reality. Merely in terms on what we could agree on at best, if anything.
- Or speaking differently, drawing PTs does not substitute looking for the better understanding of the nature and vice versa. But you can't escape the need for the former in favor for the latter (unless you seek to undermine the whole idea of the periodic table, that is).--R8R (talk) 21:56, 19 June 2019 (UTC)
- Yeah, the facts that you have to draw a PT somehow and that such a drawing will inevitably be an oversimplification of nature are what I was trying to get at, but you put it a lot more eloquently than I did. ^_^ Double sharp (talk) 04:09, 20 June 2019 (UTC)
- @Double sharp: I'm intrigued by your comments
- Double sharp, here are Mendeleev's group and period assignments, for groups III and VII:
Period III VII 2 B F 3 Al Cl 4 (Sc) Mn 5 Ga Br 6 Yt (Tc) 7 In J 8 Di --
- There's no basis to consider Mn as the next element after F, when Mendeleev quite clearly considered this to be Cl. My original supposition that he would have recognised La as the next applicable recurrence after Yt, thus still stands.
- I agree about the weirdly shaped blocks issue, which is what we have with the current (partially) split s-block. So the question is what's the story that Nature is telling us about the composition of group 3? I don't want to reopen that topic so much. If we have a choice, then yes let's use the version that's easiest to teach and represent. Let's not, however, prioritise teaching and practicality considerations over Nature's story. I suggest the teaching and practicality differences are small, in any event. Sandbh (talk) 05:06, 20 June 2019 (UTC)
- But if we are considering Di, instead of In, as the next recurrence in Mendeleev's table after Yt, that suggests that we are following the tranches, in which case F and Cl are not in the same tranche and it's instead F and Mn that are relevant. Either that or we consider the tranches to not be important and consider Cl to be next element after F, but in that case Sc is the next element after Ga, and In after Yt.
- To me, Sc-Y-La has good points, and Sc-Y-Lu has good points too, and they're close enough for the argument to rest at least partially on what you consider most important to display. In other words, I think that yes, we have a choice, and that Nature is telling us that periodicity is not so simple that you can project it down to a 2D arrangement of elements and preserve everything. For me, Sc-Y-La would be all very well if the divide between the d-block and the f-block was really stark, so that the choice was really obvious like it is for putting He over Ne, but as it stands, the irregularity it creates in the table's appearance looks like it is creating a mountain out of a molehill. The table is always going to be partially right and partially wrong: it can only ever hope to show primary periodicity and you will have to add in all the secondary factors (yes, including first-row anomaly s << p < d < f) yourselves. So if it is going to be oversimplified no matter how we draw it, we might as well draw it to look really simple, instead of adding something that looks really fiddly. Double sharp (talk) 06:39, 20 June 2019 (UTC)
- Yes, we are considering Di as the next recurrence in Mendeleev's table after Yt. Group III bifurcates after B, as group VII bifurcates after F:
- III
- Aluminium tranche: Al-(Ga)-In
- Scandium tranche: Sc-Yt-Di
- VII
- Chlorine tranche: Cl-Br-J (In volume 1 of The Principles of Chemistry, Mendeleev said that F belongs the halogens, along with Cl, Br, and J)
- Manganese branch: Mn-(Tc)-(--)
- Postscript: In the 5th (1891) edition of The Principles of Chemistry, Mendeleev shows La under Yt. Sandbh (talk) 23:39, 20 June 2019 (UTC)
- Post-postscript: He also refers to the analogues of Al as Ga, In and Tl. Sandbh (talk) 23:51, 20 June 2019 (UTC)
- While I'm sure that he said that F is a halogen like Cl, Br, and I, the fact remains that this is not what the tranches he draws in his 1871 table show, as they align F with Mn. Probably at that time he assumed that since there were similar "double-rows" from K to Br and from Rb to I, the sequence from Li to Cl could be treated analogously. And indeed that is what we see in Mendeleev's own 18-column arrangement from the same time (go to 3:28 in this video from Periodic Videos): Li through F are put in modern groups 1 through 7, and Na through Cl in modern groups 11 through 17. (H is put atop Na.) Not only that, but his final published table (go to 9:10) has exactly the same vertical alignments, with F and Mn justified to the left of their cells, but Cl, Br, and I to the right. Now of course he doesn't use A or B labels in it, but given the way he used "eka-" in "eka-boron" I think both the one-row and two-row cycles seemed important to him, and he could consider both B-Sc and B-Al (resp. F-Mn and F-Cl) recurrences in some sense. Double sharp (talk) 07:35, 21 June 2019 (UTC)
Reframing the argument
In terms of the periodic law, I think it looks like this:
- Premise 1: If the elements are lined up in one row, from 1 to 118, an approximate repetition of properties is evident, at varying but regular intervals i.e. after 2, 8, 8, 18, 18, 32, and 32 elements.
- Premise 2: An interval increase from 2 to 8, 8 to 18, or 18 to 32 occurs with the filling of a new kind of orbital.
For example, counting forward two places from #2 He lands on #4 Be. No further action required. Counting forwards two places from #4 Be lands on #6 C. This is a p element so the interval increases to eight and lands on #12 Mg.
Observations
For #37 Rb and #38 Sr, the intervals are 18 elements long representing the filling of intervening s, p, and d orbitals. The 18 element interval was triggered by #21 Sc.
For #39 Y, counting forward 18 elements lands on #57 La, representing the filling of intervening s, p, and d orbitals.
For #40 Zr, counting forward 18 elements lands on Ce at which point the the filling of the f shell starts. The interval therefore increases to 32 elements and ends on #72 Hf.
Counting forward 32 elements from Y, to land on Lu, is a non-example as no new orbital type is encountered in moving the default 18 elements forward.
Conclusion
For IUPAC, the periodic law underpinned by the filling of the s, p, d and f orbitals supports La under Y, and He over Be.
Do I have this right now? Sandbh (talk) 00:07, 25 June 2019 (UTC)
Mendeleev's line

Here’s a picture of the elements in one long line. You'll have to open the image, and then click on it again to get a readable size.
It shows the approximate repetition of properties, at varying intervals.
The big intervals are 2, 8, 18, and 32, as triggered by the appearance of an s, p, d, or f electron, at H, B, Sc, and La respectively. These intervals are coloured.
There are a few lesser repetitions along the way, at intervals of 7, 9, 10, 13, 14, 20, 22 or 34 elements. Included here are e.g. the diagonal relationship along the first row elements, and the knight’s move relationship originating in the lower right of the d-block. These relationships are not coloured. Some of the repetitions are those that occur after the first repetition e.g. Cu-Ag-In-Tl.
As can be seen, the next repetition after Y is La.
The key question is why would La be skipped in favour of Lu? Sandbh (talk) 08:12, 28 June 2019 (UTC)
- Because 4f becomes chemically relevant at La even if it doesn't appear in the ground state until Ce (you have to consider excited states at energies within the range of bond energies); because 4f is not significantly chemically relevant at Lu (less than 3d in Zn); because the interplay of 4fn and 4fn+1 configurations characteristic of 4f chemistry is possible for La but is mathematically impossible for Lu; because the chemical and physical properties of Lu are closer to Hf–Hg than La is to Hf–Hg (the other sure 5d elements); and because the "delayed collapse" effect becomes stronger and stronger at higher atomic numbers, so you have to take it into account for why La's f-involvement is weaker than you expect it to be. In other words, I think saying that a block starts when its characteristic electron appears in the ground state only is too coarse. Double sharp (talk) 10:45, 28 June 2019 (UTC)
You must have been on fire (in a good way) when you wrote that!
- “4f becomes chemically relevant at La even if it doesn't appear in the ground state until Ce (you have to consider excited states at energies within the range of bond energies);”
- Do you have any good examples of 4f becoming chemically relevant at La? I expect such involvement is likely to be extremely marginal.
- See below. Double sharp (talk) 06:50, 1 July 2019 (UTC)
- Do you have any good examples of 4f becoming chemically relevant at La? I expect such involvement is likely to be extremely marginal.
- “4f is not significantly chemically relevant at Lu (less than 3d in Zn);”
- The ionic radius, and hence the chemical behaviour of the Ln ions proper, including Lu, is impacted by the presence of f electrons. We know this because the lanthanide contraction runs from Ce3+, with the configuration [Xe]4f1, to Lu3+ with [Xe]4f14. This is not the case for lanthanum.
- Sure it is, if a bit degenerately: as you add f-electrons the ion gets smaller, and that surely happens going from 0 to 1 f-electron (La3+ to Ce3+) just as it happens going from 1 to 2 (Ce3+ to Pr3+). The difference is that until Yb the 4f electrons are surely active but at Lu they surely are not: they are core electrons any way you think about it. (Otherwise we could just as well note that the radii of Hf, Ta, and so on are also impacted by the presence of f electrons in the core.) Most lanthanides can be seen in both [Xe]4fn5d16s2 (trivalent) and 4fn+16s2 (divalent) configuration: yes, this is difficult for lanthanum because of the delayed collapse, but at least it is mathematically possible. You can't do it at all for Lu because then n = 14, n + 1 = 15, and the 4f shell can only hold fourteen electrons. That already makes me think that if we have to pull out one of La and Lu from the series, it makes more sense to pull out Lu, even if 4f involvement in La is marginal. We can just see it as the affliction of delayed collapse, which explains why it seems to be more severe in 5f; whereas if we were to follow the 5f collapse just as we did the 4f collapse, Th is also a weak f-block member (albeit a bit stronger than La), and the block gets "staggered" later each row. The same thing happens with the d-block (true transition chemistry with variable oxidation states and all that only starts at Db in the 6d row, group 5 instead of group 3 or 4). Double sharp (talk) 06:50, 1 July 2019 (UTC)
- The ionic radius, and hence the chemical behaviour of the Ln ions proper, including Lu, is impacted by the presence of f electrons. We know this because the lanthanide contraction runs from Ce3+, with the configuration [Xe]4f1, to Lu3+ with [Xe]4f14. This is not the case for lanthanum.
- “the interplay of 4fn and 4fn+1configurations characteristic of 4f chemistry is possible for La but is mathematically impossible for Lu;”
- For La do you have any good examples of this interplay? I expect such interplay is likely to be extremely marginal.
- The important thing is the existence and energy of such configurations. As I gave the last time: the type of 4f bonding (10.1039/C6DT00358C) in Ln2O3 depends on the energy levels of the 4fn and 4fn+1 states. Here La patterns with with Gd–Er in the band model, but Lu is an outlier because there are no 4fn+1 states and spin-orbit coupling is eliminated. I'd rather have La with both configurations (even if one is marginal) than Lu with only one in the f-block, where everybody else can show both. Double sharp (talk) 06:50, 1 July 2019 (UTC)
- For La do you have any good examples of this interplay? I expect such interplay is likely to be extremely marginal.
- “…the chemical and physical properties of Lu are closer to Hf–Hg than La is to Hf–Hg (the other sure 5d elements);”
- We know that Restrepo (2018) found that La shares similarities with transition metals and with lanthanides, while Lu is more similar to lanthanides than transition metals, based on the stoichiometry of binary compounds. We know this is consistent with Scerri in his red book (2007): "…there are in fact two big ideas in chemistry. They are chemical periodicity and chemical bonding, and they are deeply interconnected" (p. xiii). This goes back all the way to Mendeleev.
- We know this accords with the observations, some 50 years ago, of Spedding and Beadry (1968, p. 377), who wrote, "Since metallic lutetium resembles closely erbium and holmium, except that it melts at a slightly higher temperature and is essentially non-magnetic, the details of producing, purifying and fabricating it are almost identical with those described under Holmium."
- There is another more recent work by Glawe et al. (2016), based on the degree of chemical substitionability between pairs of elements, which suggests La is better fit with the transition metals whereas Lu is a better fit with the lanthanides (see Figure 1 in their article).
- We're not comparing La and Lu to the lanthanides, but to the early transition metals, because we want to see which one is a better fit for the d-block. Then it's obvious that Lu is much closer to the 5d transition metals (just look at density, melting point, atomic radius, electronegativity, hardness, bulk modulus, etc.) from basic physical properties. I'm inclined to think that what Restrepo is considering is not all that important when comparing elements from different groups (like La/Hf vs Lu/Hf), because then the stoichiometry will almost by definition disagree. Double sharp (talk) 06:50, 1 July 2019 (UTC)
- There is another more recent work by Glawe et al. (2016), based on the degree of chemical substitionability between pairs of elements, which suggests La is better fit with the transition metals whereas Lu is a better fit with the lanthanides (see Figure 1 in their article).
- In archive 38 you argued that “…the weight of evidence seems to support Lu-Lr as more similar to Hf through Hg and Rf through Cn; so the d-block is more homogeneous with Lu and Lr in group 3.”
- Even if this was true it would make the f-block less homologous. And the d-block would become less homologous in terms of differentiating electrons due to the pesky p electron in Lr.
- Both La and Lu are very similar to the rest of the lanthanides chemically, so I don't consider it to be a problem. And considering that the 7p differentiating electron in Lr seems to have about zero effect on its chemistry I also don't consider that a problem either. Double sharp (talk) 06:50, 1 July 2019 (UTC)
- Even if this was true it would make the f-block less homologous. And the d-block would become less homologous in terms of differentiating electrons due to the pesky p electron in Lr.
- We shouldn’t forget the other, related, arguments for La under Y:
- “…if you look at atomic properties, the trend B–Al–Sc–Y–La–Ac (e.g. electronegativity and ionisation energies) is a smooth decline…These intraperiod resemblances are naturally swamped by the closer intragroup resemblances; but periodicity is based on trends, not just similarities…"
- That's a quote by you, from Archive 33. Your first sentence seems to contradict your second. Intraperiod resemblances are indeed swamped by intragroup trends. And we know that intragroup trends favour La under Y, given these trends match those in groups 1 and 2. As we know, Laing (2009, p. 12) said a reasonable case could be made for La below Y on the basis of comparing the pairs Ca-Sc and Sr-Y with Ba-La.”
- Yes, but you can draw analogies also with the group 4++ trends and Sc/Y/Lu: a reasonable case could be made for Lu below Y on the basis of comparing the pairs Sc-Ti and Y-Zr with Lu-Hf. As we remarked it then hinges on whether you think group 3 should pattern with the pre-transition groups or the transition groups. And as Droog Andrey noted, if you think group 3 patterns with the pre-transition groups, you then have to ask "why not B-Al-Sc-Y-La, then"? And if you then reply "because B and Al are p1 but the rest are d1", it then starts becoming a game of finding when 4f and 5f really become chemically active. That's how we first decided on Sc/Y/La, based on condensed-phase arguments and the delayed collapse. But because in the condensed phase Be and Mg have p-involvement, and because delayed collapses raise uncomfortable questions about the 6d and 5g blocks, I would now insist on also asking: when do they become chemically inactive? And the answer seems to be Lu/Lr rather than Hf/Rf. That way, the f14 column patterns better with the other block-ending columns where the electrons are still removable in at least some of the elements: Yb and No can use their f-electrons (albeit No and E156 don't want to because of relativistic effects), but Lu, Lr, and E157 all cannot. Double sharp (talk) 06:50, 1 July 2019 (UTC)
- That's a quote by you, from Archive 33. Your first sentence seems to contradict your second. Intraperiod resemblances are indeed swamped by intragroup trends. And we know that intragroup trends favour La under Y, given these trends match those in groups 1 and 2. As we know, Laing (2009, p. 12) said a reasonable case could be made for La below Y on the basis of comparing the pairs Ca-Sc and Sr-Y with Ba-La.”
- “the "delayed collapse" effect becomes stronger and stronger at higher atomic numbers, so you have to take it into account for why La's f-involvement is weaker than you expect it to be.”
- I agree.
- Great! ^_^ But I would treat this as a second-order perturbation. In other words, you expect the block to start here, but it's a bit weak in the first few elements because of delayed collapses (Ac in 5f, Lr in 6d, E121 in 5g). Double sharp (talk) 06:50, 1 July 2019 (UTC)
- I agree.
- "In other words, I think saying that a block starts when its characteristic electron appears in the ground state only is too coarse."
- It’s consistent with all my responses above. It's the simplest rubric I can think of that produces consistent results viz. "The best education is found in gaining the utmost information from the simplest apparatus". (Whitehead AN 1929, The aims of education and other essays, The Free Press, New York, p. 37).
- But if the cost is that a block then has to extend to the point where its characteristic electrons are core electrons, then I'd rather have weakly involved excited states at the start instead. Double sharp (talk) 06:52, 1 July 2019 (UTC)
- It’s consistent with all my responses above. It's the simplest rubric I can think of that produces consistent results viz. "The best education is found in gaining the utmost information from the simplest apparatus". (Whitehead AN 1929, The aims of education and other essays, The Free Press, New York, p. 37).
Sandbh (talk) 04:43, 1 July 2019 (UTC)
Terminology
- re Mendeleev's "groups" (I-VIII) and "periods" Double sharp and Sandbh mention. I don't know if this changes the statements you made, but the fact is that M. did not use or define "period"; in 1871 he defined "Reihe" (German for "row"). Sandbh's table here confuses those two and their numbers. In general, Reihe number =/= period number; only so for Reihe=period 1, 2, 3.
- 1871 to 2019: Reihe 1 = period 1, Reihe 2 = period 2, Reihe 3 = period 3 aka 3ACAS, Reihe 4 = period 4AIUPAC, Reihe 5 = period 4BIUPAC. Same for pairs Reihe 6, 7 (make period 5), Reihe 8/9/10/11 (make period 6; complication because lanthanides come in play), final Reihe 12 returns in period 7. Note that Reihe 3 is period 3, all 3ACAS and "3A,BIUPAC, with each valence only once".
- Our modern group 3, whichever way detailed, appears only from Reihe 4 (=period part 4AIUPAC), as Sc with valence III, then in Reihe 6 (=period part 5AIUPAC) with Y. I skipped Reihe 5 (period part 4B) and 7 (period part 5B), Ga and In, the other valence III in the periods.
- Take home: a Reihe is not a period. Those who changed the PT from Reihen (shorty form) to periods (long form) performed a complicated and smart task (so hard to explain/describe even afterwards today). Then the word 'period' was created usefully. The periodic law then became a double-periodic or how to say this: a valence (I-VII) appears twice in a single period. Bold: for the topic at hand i.e. Janet's Left Step, this change into long form is more important than Janet's change, because the long form introduced block forms & fillings in the PT, we now easily recognise in regular and Janet's PTs. -DePiep (talk) 09:58, 20 June 2019 (UTC)
- Yes, this is why I noted that the next recurrence of properties after Al is Sc and that Mendeleev puts it in the next row in the same column. You have a recurrence of valence before a whole period is completed, and given that his groups included things like F and Mn in the same tranche, clearly valence is important to Mendeleev's assignments. (That's why modern short-form periodic tables break the pattern for the Ln and the An.) Double sharp (talk) 10:38, 20 June 2019 (UTC)
- I do not exactly grasp why the 1871 PT would enlighten us wrt the Left Step PT (except for detailing the recent group 3 discussion, I understand?).
- To illustrate how Mendeleev's 1871 PT morphed into the classic long PT (ca. 1930?, say pre block f), I started {{Periodic table (Reihen and periods)}}. It invites us to see how multiple Reihen could be squeezed into one modern period. Interesting detail visible: in modern period 6, in groups III and IV, we see how Mendeleev was handling the then known Ln's and An's (?Di, ?Ce, ?Er, ?La, Th, U; +predicted Pa): he had to use extra rows for period 6 to maintain both increasing atomic weight & valence periodicity. -DePiep (talk) 17:32, 20 June 2019 (UTC)
- Yes, you're right. The main issue is detailing the recent group 3 chat. Sandbh (talk) 23:26, 20 June 2019 (UTC)
- Scerri The PT (2007), p. 111/fig. 4.4 has a reproduction of an English version anno 1871! Reihe is translated as Series, which is more to the point than just row. (Series = has a pattern, row = graphical/tablewise only). -DePiep (talk) 18:48, 10 July 2019 (UTC)
- Yes, you're right. The main issue is detailing the recent group 3 chat. Sandbh (talk) 23:26, 20 June 2019 (UTC)
- Yes, this is why I noted that the next recurrence of properties after Al is Sc and that Mendeleev puts it in the next row in the same column. You have a recurrence of valence before a whole period is completed, and given that his groups included things like F and Mn in the same tranche, clearly valence is important to Mendeleev's assignments. (That's why modern short-form periodic tables break the pattern for the Ln and the An.) Double sharp (talk) 10:38, 20 June 2019 (UTC)
- Sandbh Please do not introduce "LST" for the stepped one. Keep using "PT", so for example it can be "LSPT". Thanks ;-) -DePiep (talk) 21:27, 7 July 2019 (UTC)
Halides
In Jensen’s 1982 article proposing Lu in group 3 he included a table comparing several properties of Sc, Y, Lu and La.
One of these properties was for the crystalline structure of the trichlorides. This entry showed Sc, Y and Lu having the same structure whereas La differed, hence Jensen was suggesting Lu was a better fit in group 3.
I looked up the structures of the chlorides in Jensen’s source (Wells 1975, Structural inorganic chemistry) and found:
- Sc = Y = Lu = YCl3; La = UCl3
So there is a match here with Jensen.
Later work (1994) on the structure of ScCl3 shows it as the BiI3 layered type, which makes more sense:
- Sc = BiI3; Y = Lu = YCl3; La = UCl3
Going back to Wells, he shows the fluorides as follows:
- Sc = ReO3; Y = Lu = YF3; La = LaF3
So there is a similar pattern to the chlorides.
Neither pattern suggests anything in support of either Lu or La.
The puzzle is why Jensen chose to leave out the fluorides given they were also listed in Wells, or provided no explanation for doing so.
Writing in From Mendeleev to Oganesson (pre-print chapter here) Scerri and Parsons (2018) wrote:
"…we believe that Jensen has been too selective in the evidence he puts forward in order to support his case. We will show that if other forms of data are appealed to, the case becomes somewhat less compelling."
Sandbh (talk) 23:11, 14 July 2019 (UTC)
- It seems this has not popped up anywhere or I have missed it, but here's the actual book as it was printed. Scerri's bit on group 3 is available without limitations.--R8R (talk) 18:44, 20 July 2019 (UTC)
2nd ionisation energies?
Following on from the above subsection, the other puzzle is why Jensen chose to use the 2nd ionisation energies of Sc Y Lu and La, rather than their 3rd IE’s). Scerri and Parsons write:
“For example, it is not at all clear why Christyakov [whom Jensen used as a source] and following him Jensen, have chosen to focus only on the sum of the first two ionization energies of the elements in question rather than say the sum of the first three ionization energies. The latter quantity is in fact more relevant given the overwhelming tendency of actinides [sic] to form ions with a +3 charge.”
@R8R: Would you be able to look up Christyakov and see what he said, if anything, about this? The source is Christyakov, V. M., Zh. Obshch. Khim., 38(2), 209 (1968). Thank you. Sandbh (talk) 23:25, 14 July 2019 (UTC)
- Hi. I've tried but been unlucky so far. I may try more in the future but I suspect this is not to be found online.--R8R (talk) 10:01, 20 July 2019 (UTC)
Ion configurations
In Foundations of inorganic chemistry (2018) the author (Wulfsberg) says most chemists encounter ions of the elements far more frequently than their gaseous atoms in a high vacuum.
So he teaches the electron configurations of ions, "in precipitation chemistry and the solid-state chemistry of salts", since ions don’t have anomalous configurations.
This prompted me to make a list of ion configurations:
s-block
Cations have an s0 configuration
Anions e.g. Na− have an s2 configuration
p-bock
Cations are s0 (or s2)
Anions have a p6 configuration
d-block
Cations are group number less charge, which gives a range of d0 to d10
Anions have complete d and s shells e.g. Au−
f-block
With La starting, cations are f0 to f13
With Ce starting, are f0 to f14.
In terms of regularity, the f-block starting with La looks odd since f14, as Lu3+, has been shunted into the d-block.
All of this can be washed away by saying it never the less isn’t possible to distinguish between the two group 3 options on physical, chemical, or electronic properties.
Still, it does look odd. I don't know; it makes me think that Lu arguments are more aligned with e.g. quantum mechanics, physics, and the Madelung rule, whereas La arguments seem to be more practical. Maybe. Sandbh (talk) 06:12, 25 July 2019 (UTC)
- Well, we also have Yb2+ and No2+ to give us f14 even if we cut Lu and Lr off, just as Ce4+ and Th4+ give us f0 even if we cut La and Ac off. We have one major oxidation state in the Ln (+3), but Ce, Sm, Eu, and Yb (especially Ce and Eu) often show another one (+4 for Ce, +2 for the others). For nobelium +2 is even the major oxidation state. Double sharp (talk) 07:38, 28 July 2019 (UTC)
- P.S. Perhaps we are focusing too much on the constant +3 across the lanthanides, because the actinides do not have this. There are similarly contractions from YIII to AgIII (and really across the whole table, as that is basic high-school chemistry), but no one cares about that because +3 is not the major state for all of those elements. Here is what Seaborg had to say:
It is important to realize that the electronic structures listed in Table 6 are those of the neutral (unionized) gaseous atoms, whereas it is the electronicstructure of the ions and compounds that we are chiefly concerned with in chemistry. The relationship of the electronic structure of the gaseous atom of an element to that of its compounds can be rather complicated. For example, in the case of the actinide and lanthanide elements, one would not necessarily predict the predominance of the III oxidation state from the electronic· structures of the gaseous atoms; there are usually only two so-called "valence electrons," the 7s or 6s electrons, which might indicate a preference for the II oxidation state.
Apparently, specific factors in the crystal structure of, and the aquation (hydration) energies of, the compounds and ions are important in determining the stability of the III oxidation state. Thus, the characteristic tripositive oxidation state of the lanthanide elements is not related directly to the number of "valence electrons" outside the 4f subs hell, but is the somewhat accidental result of a nearly constant small difference between large energy terms (ionization potentials on the one hand, and hydration and crystal energies on the other) which persists over an interval of fourteen atomic numbers. Therefore, if we could somehow have a very extended Periodic Table of Elements containing numerous "f" transition series, we might expect that the 5f, rather than the 4f, elements would be regarded as more nearly representative of such f series.
- And indeed we have some inkling of this from calculations, where the 6f elements are definitely a lot more like the actinides than the lanthanides, with unprecedentedly high oxidation states represented. The 5g elements are more like the 4f ones, because of a lack of radial nodes. Unfortunately we do not seem to have more recent calculations on the 6g elements than the ones on E173 and E184 we quote at extended periodic table; at the beginning 6g is for sure active at E173, but at E184 it seems that at least 7f and 8d are higher in energy and drown it into the core, so we cannot say too much yet without knowing what happens in between. (And here there is a significant difference in that we no longer have a covering shell.) Double sharp (talk) 03:28, 29 July 2019 (UTC)
Short solution
Here's the short solution to the group 3 question:

It’s based on the supposition by Scerri and Parsons, that it's not possible to differentiate between Sc Y La Ac and Sc Y Lu Lr on the grounds of electron configurations or chemical or physical properties (2018, pp. 145–146).
It's implemented by adding a group 3 label to La Ac:

That's it.
We've discussed this approach before and it was well received. I'd like to submit it to the IUPAC Group 3 Project.
Any further thoughts?
- Scerri E and Parsons W 2018, "Which elements belong in group 3", in Mendeleev to Oganesson: A multidisciplinary perspective on the periodic table, E Scerri and G Restrepo (eds), Oxford University Press, New York, pp. 140–15.
-- Sandbh (talk) 00:13, 6 July 2019 (UTC)
- Well, I like it. We can label them 3 (f) and 3 (d), and note that to some extent you can think of group 13 as 3 (p). (To some extent group 12 might be 2 (d), as well.) Double sharp (talk) 03:41, 6 July 2019 (UTC)
Thank you! It's been pointed out to me that IUPAC and the chemical community may not like to revisit group numbering since they went through a whole lot of pain in arriving at the current 1--18 arrangement of a unique number for each numbered group.
Still, I am not proposing a new numbering scheme, or to add a completely new group number.
I like your suggestion a lot. It's quite clever. I am worried that this could give rise to the impression that there are two group 3's. [or 3 if we have 3(p)] Also that 3(f) would have two elements with no f electrons. Even though, in this arrangement they would be part of the f-block. Sandbh (talk) 06:08, 7 July 2019 (UTC)
- The second one doesn't bother me that much, since 4f/5f are theoretically relevant for La/Ac, just delayed like 6d is for Lr. The first one may indeed cause some misunderstandings, but I think you could deal with it by saying that "OK, we show Sc/Y/Lu/Lr on our periodic table because it looks simpler and it's not really any worse at showing trends, but both Sc-Y-La and Sc-Y-Lu show some reasonable trends that we can look at sometimes and for convenience we give them both names." Greenwood and Earnshaw show trends like B-Al-Sc and C-Si-Ti too when relevant, so there's a precedent for equivocating on which branch of the old "group 3" vs. "group III" we mean. (I realise it's naughty but for this kind of thing I think the old A-B nomenclature should still have its place, as it corresponds to real chemical facts. I'd just rather replace "A" and "B" with the schematic block letters when this is relevant, like "3 (p)", because no one can agree on whether IIIB is the one with Sc or the one with Ga. But I understand that chemists may not want to go through all that pain again, even if my suggestion just adds a few alternate legal names for the groups.) Double sharp (talk) 07:43, 7 July 2019 (UTC)
- So we should not name (label) them 3(f) and 3(d) all right. After all, they are not different groups, that is the main outcome. But to describe and refer to the different columns in texts like this, pointing out by saying the "3(f) column", "3(d) column" is acceptable IMO, preventing confusion. -DePiep (talk) 09:04, 8 July 2019 (UTC)
- Yes, something like that will probably happen. As you say there is no 3f group and no 3d group. There is only one group 3. It just happens to have two tranches after Y. I don't think I'll say anything about this in the IUPAC submission as it looks too weedy. If you want to refer to a particular tranche of group 3, perhaps 3L for the left and 3R for the right? Not much in it. I wouldn't want the revisit the past confusion around A and B labels. Another reason to avoid the whole thing is that on the PT you'd still only see two 3's. Sandbh (talk) 03:23, 9 July 2019 (UTC)
- So we should not name (label) them 3(f) and 3(d) all right. After all, they are not different groups, that is the main outcome. But to describe and refer to the different columns in texts like this, pointing out by saying the "3(f) column", "3(d) column" is acceptable IMO, preventing confusion. -DePiep (talk) 09:04, 8 July 2019 (UTC)
- Do I understand that with this approach, there is no need to draw Sc, Y above L
ua, Ac any more ever (still both columns having the "3" label)? Or is there a not-so-short-solution where this would be meaningful (as a physical/chemical statement)? -DePiep (talk) 11:10, 7 July 2019 (UTC)
Yes, I'd expect that's largely right. In a chemistry book chapter you might want to have Sc Y La Ac beside Sc Y Lu Lr to show what is going on. You might want to show Sc-Y-La-Lu-Ac-Lr, or
+ La-Ac
Sc-Y-|
+ Lu-Lr
and add some trend lines (continued in subsection after next)
Split d-block, not
- @Double sharp: The short solution doesn't prevent a split d-block since Sc and Y could be moved over La, just as easily, with a 3 over Sc and a 3 over Lu. However, since the two options can't be differentiated on physical, chemical, or electronic grounds, Sc Y Lu Lr is preferred as it doesn't require the artifice of a split d-block. Sandbh (talk) 07:10, 26 July 2019 (UTC)
- @Sandbh: Yes, this is pretty much why I favour Sc-Y-Lu now, albeit with support for marking La and Ac as a separate branch of group 3. Double sharp (talk) 07:19, 26 July 2019 (UTC)
- @Double sharp: Excellent. The only remaining issue is possible confusion on whether Ce to Yb and their An congeners also belong to group 3. Sandbh (talk) 07:33, 26 July 2019 (UTC)
- @Sandbh: Well, I would want to say that they are not strictly group 3 members because of significantly stronger f-orbital involvement, but that in many situations it is useful to consider some of them alongside the more strictly defined group 3 elements for comparative chemistry. Double sharp (talk) 07:39, 26 July 2019 (UTC)
- @Double sharp: That might work. It's certainly the case for the lighter An. Sandbh (talk) 12:25, 26 July 2019 (UTC)
- Actually, the distinctive chemistry of the light Ln is a good enough reason to keep them out of group 3. Sandbh (talk)
Trendline charts
-
1st IE
-
Densities
-
3rd IE
-
Melting points
-
Ionic radii, 6 coordinate
-
Electron affinity
The 1st IE chart, and the density chart, show how smooth these properties are.
The 3d IE chart shows the impact of the lanthanide contraction.
The melting point chart is interesting. It looks like Lu and Lr benefit significantly from the greater pulling power on their delocalised electrons than is the case with La and Ac, due to poor shielding by the f electrons. I didn't even know this was the case with Ac. I had read that the low mp of La might've been due to the presence of some f character, but maybe not.
Ionic radius has a large impact on chemical behaviour, and once again you can see the result of the poor shielding from the f electrons, in Lu and Lr.
At the end of the day you can do dozens of these charts (gets a bit tricky with Lr as you'd have to use many predicted values). Sometimes you can't tell the difference; sometimes it looks better as La-Ac; sometime it looks better as Lu-Lr. The end result is likely to be that you can't practically distinguish between the two sets of Sc-Y-La-Ac and Sc-Y-Lu-Lr.
I find the graphs to be wonderful. Now I can see what's going on, much more plainly, and marvel at the impact of the Ln and An contractions.
Having been more inclined to support -La-Ac, I'm completely comfortable with the two tranche proposal, even with La Ac out on the right: that seems appropriate given their status as Ln and Ac progenitors.
Am very pleased with all this. The comparative chemistry opportunities look to be rich. Winners all round. Sandbh (talk) 08:07, 8 July 2019 (UTC)
- EA chart. I added an electron affinity chart. So now there are 2 smooth graphs, 2 graphs showing Lu and Lr with higher values, and two graphs with La and Ac having higher values. Sandbh (talk) 01:58, 9 July 2019 (UTC)
15-wide footnote
Could this option support a 15-wide footnote(?):
1 2 3 4
H
Li Be
Na Mg
K Ca Sc Ti
Rb Sr Y Sr
Cs Ba Ln* Hf
Fr Ra An^ Rf
3 3 *Lanthanides La Ce......Yb Lu ^Actinides Ac Th......No Lr
- Table: Demo 1
Group 3 has two tranches under Y, and the Ln and An could be regarded as two horizontal series in group 3. Here, the trends going down Sc Y La Ac most closely match groups 1 and 2, while the trends going down Sc Y Lu Lr most closely match the trends going down groups 4 to about 10.
IUPAC might be attracted to a proposal which still facilitates a 15-element wide footnote whereas those who like to bring out the blocks more can use a 14-element wide footnote. Sandbh (talk) 11:07, 18 July 2019 (UTC)
- "IUPAC might be attracted to ... " — I thought the presentation form was not to be involved in the Group 3 Question. Anyway, here is my reply: NO. This one is wrong, inconsistent, self-contradicting and exactly the option that was excluded in the Scerri-taskforce remit.
- As the scheme is drawn, above it unambigously states that "Ln*" and "An^" are all and completely in group 3, because they are under the "3" column header. Then not putting a "3" above the columns Ce ... Yb below makes the statement inconsistent, contradicting even. We do not need a graph that requires an explanation into "but you must read it different from what it actually shows & says".
- FFS, start with the correct 32-column one you already have, and if someone wants to cut out the Ln+An's so desparately (why anyway? why not cut out the metalloids? why not the rare earths?), at least be sure after the cutting to not merge columns, empty or not) that are not similar. How difficult can it be? Even going back to a Mendeleevian 10-column form, extended with f-block Reihen, would be more correct. -DePiep (talk) 12:58, 18 July 2019 (UTC)
Some people like to cut out the Ln+An on chemical similarity grounds, and to keep the width of the table more manageable. Is this better(?):
1 2 3 4
H
Li Be
Na Mg
K Ca Sc Ti
Rb Sr Y Sr
Cs Ba* Hf
Fr Ra^ Rf
3 3 *Lanthanides La Ce......Yb Lu ^Actinides Ac Th......No Lr
- Table: Demo 2
Yes, this is out of scope of the project. It may help grease the wheel. Sandbh (talk) 02:05, 19 July 2019 (UTC)
- (Redacted) Please read my replies, recent and old, saying: "if you put a 3 above a column, the whole column is 3".-DePiep (talk) 22:38, 20 July 2019 (UTC)
- @Sandbh: re "Is this better(?)": you can self-check, because it is not a secret operation ;-). The footnote in/out operation must be possible with scissors and glue. Starting with this scheme, move the footnote back into the top table. In this case, cut the top table vertically between "2" and "3" with scissors, then move columns "2" and "3" apart, 15 empty columns wide. Then cut out the 15-column footnote (including the "3"s), and place it in the new space, glue it. The check is: is there any element-in-group situation that is wrong?. AFAIK, not. It's just that you have three columns heading "3" and two empty cells below Sc/Y (as you have at the beginning).
- There is one uneasy detail: the text says "Ba*" in one cell, and it is unclear how exactly the footnote replacement is to be positioned. One could correctly conclude that the 15-column footnote should be "in" the cell with Ba somehow (so all in group 2!). This is not what we want to suggest, but it is the essence of the asterisk (=a placeholder here, but a reference/authors comment etc it means elsewhere). SAme for An^of course.
- This would be a correct form both with/without using the footnote construct:
1 2 3 4
H
Li Be
Na Mg
K Ca Sc Ti
Rb Sr Y Sr
Cs Ba Ln* Hf
Fr Ra An^ Rf
3 3 *Ln: Lanthanides La Ce......Yb Lu ^An: Actinides Ac Th......No Lr
- Table: Demo 2, adjusted
- I see no solution if you want this graph: have two elements below Sc/Y and put all Ln's An's in the footnote (15-column). One could add Sc/Y in the footnote in one of the group-3 columns 100% correctly (making it a 4-period footnote), but that would be "Rare earth elements" nnot "Lanthanides" then.
- Of course this may be a bit strange to older people who learned the PT in classical form with the footnote always present, but it appears that this modern group 3 presentation requires some graphic changes in there. I remember seeing even an extended PT by Pyykkö, where this group 3+footnotes was drawn a bit compromised (unclear). -DePiep (talk) 07:05, 23 July 2019 (UTC)
Introducing: Droog Andrey's depiction (extended PT)

There are some arguments for drawing the gap in between groups 2 and 3 a little bit funkily. It is kind of like the notation in Chopin's Nocturne Op. 15 No. 2 which made Donald Byrd's page on interesting music notation: there are too many relationships we wish to show, and drawing them all produces what literally appears to be nonsense and must be interpreted a little bit imaginatively, but the end result has more explanatory power than something literally correct. It is how we can say that yes, theoretically the periodic table is 32 columns and if we take a footnote out, it should not matter whether we put it under the main body, above the main body, on another page altogether, or on Saturn; but there are some secondary-periodicity relationships like Ac–U ~ Lu–W that are irresistible to show through a specific alignment of the extra footnotes. And it is also why I think the best solution I've seen yet for dealing with period 8 is Droog Andrey's, because even if it is literally very unclear about how the 121–156 stretch ends up in the main table (you have to do some imaginative gluing), its mastery of the chemical properties involved is indisputable as 139–156 indeed ought to act like heavier congeners of 121–138 (adding a relatively inert filled 5g subshell), and 121–138 and 139–156 are respectively analogous in their chemistry to the Ln and An (57–70 and 89–102) respectively even if they are supposed to both be in one period. Plus, it makes the aspect ratio nice and neatly aligns La and Ac with group 3 as well (not 121, but you can't have everything, I suppose). Double sharp (talk) 15:21, 23 July 2019 (UTC)
- Table: Demo 3
- I like Droog Andrey's format. Picking up from DePiep's comments, I believe the 15 form would look like:
1 2 3 4
+--+
|H |
+--+--+
|Li|Be|
+--+--+
|Na|Mg|
+--+--+ +--+--+
|K |Ca| |Sc|Ti|
+--+--+ +--+--+
|Rb|Sr| |Y |Zr|
+--+--+ +--+--+
|Cs|Ba|* |Hf|
+--+--+ +--+
|Fr|Ra|** |Rf|
+--+--+ +--+
3
+--+--+- -+--+--+
* |La|Ce| etc |Yb|Lu|
+--+--+- -+--+--+
**|Ac|Th| etc |No|Lr|
+--+--+- -+--+--+
- Sandbh (talk) 01:25, 25 July 2019 (UTC)
- Yes. For clarity, one could add a redundant "3" above Lu, Lr. One can still add the Ln, An category names, preferably below.
- Re Droog Andrey's PT: glad you all like it, to me it looks simple & not overly constructed (no graphic tricks needed). IIRC, Fricke and Pyykkö would yield something different (that is, different structure or different element order required). Anyway, up & until E118 (E120 even) this one accomodates the issue at hand (group 3 &tc).
- Just one question with it: how do the two footnotes align between them? Which
 -elements are under/in column La, Ac? (... looking this up ...) Answer from Talk:Extended_periodic_table/Archive_3#Location_of_the_6f_series: column is La, Ac, E143, so nicely right-aligning. (one could glue the two footnotes like this).
-elements are under/in column La, Ac? (... looking this up ...) Answer from Talk:Extended_periodic_table/Archive_3#Location_of_the_6f_series: column is La, Ac, E143, so nicely right-aligning. (one could glue the two footnotes like this). - But wait. The image here says that E121 and E139 are in the same column (suggesting same electron fillng pattern), but the original, very very long PT says:
 =single-line (121–156; and color change really, in Archive 3?). For this, the
=single-line (121–156; and color change really, in Archive 3?). For this, the  footnote could be split horizontally (add whitespace line), ... or drawn in full, 36-wide. I think once people go into that extended area, one better accept a less practical but more informative form. Those initiated can secretly use a compact form+mindmaps. -DePiep (talk) 07:19, 25 July 2019 (UTC)
footnote could be split horizontally (add whitespace line), ... or drawn in full, 36-wide. I think once people go into that extended area, one better accept a less practical but more informative form. Those initiated can secretly use a compact form+mindmaps. -DePiep (talk) 07:19, 25 July 2019 (UTC)
- It's something like the pseudo-alignment of Ce/Th and group 4 in our tables. E121 and E139 (and so on with E122 and E140, E123 and E141, etc. until E138 and E156) have similarities and the overly long 36-wide footnote is split there in half to show this (the similarity is that they differ by an inner 5g18 shell). Of course they are not literally in the same group, but then neither are Rf and Ce. So you can think of the idea as being that "OK, if we have to distort that 54-column table so that it fits on a page and doesn't ask for nested footnotes, we will make the distortions point to something meaningful". I agree that a bit of whitespace between the rows 121–138 and 139–156 would clarify things. As for the colour change: unfortunately, while there is a chemical difference between the elements in the 120s and their homologues in the 140s, the transition from 5g being the active subshell being filled to 6f is not very clear (we need more accurate predictions to know when 5g stops being chemically active, and when purple should change to green). It would be possible to keep the compressed layout and recolour 143–156 to green for consistency and a hint of how to do the gluing, but we should note that this is for now a formal assignment that Droog Andrey calls in that archive "only a rough pattern". The homology may be a bit formal at times, like how E148 with its predicted +12 goes below Pu which seems to be stuck at +7, but considering that Sm is stuck at +4 at the very most this is clearly not a new issue with f-block homologies. Double sharp (talk) 12:59, 25 July 2019 (UTC)
- The two User:Droog Andrey PT's are not consistent: Pt172expanded.png <-> Pt172new. Please solve this first. -DePiep (talk) 21:12, 27 July 2019 (UTC)
- DePiep says: The one an only good route is: draw the PT as wide as possible. Draw as many columns & groups & blocks as you need to state your point. Then publish it. Then lesser deities can fold it into lessfewer columns and start using footnotes and make their mistakes. But do not compromise beforehand. -DePiep (talk) 21:18, 27 July 2019 (UTC)
- They are not really inconsistent if you think of the 32-column form as likewise asking for a right-aligned gluing of the superactinide footnote (as you interpreted the 18-column form), desiring not to keep the chemically similar superactinides visually apart. But even if they can be interpreted as such, I think that is more a feature than a bug, looking at what those elements probably are like, with a multi-shell 5g/6f inner transition series that is simply without precedent in how the g- and f-blocks are smeared together. No less than Seaborg suggested that the superactinides be simply shown as a third long transition series under the lanthanides and actinides; although he apologised for it by saying that "this is admittedly an approximation and a matter of finding the best compromise" and called it an "oversimplification of a complicated situation", following a block structure thoroughly demands information we do not have (and when we do have it it will likely contradict whatever block structure we choose because it's not all that clear when 5g drowns fully into the core). Whatever f-block homologies you show are likely to be purely formal only (perhaps worse than the homology of Nd to U), outside perhaps rare oxidation states (U is indeed like Nd in the +3 state, but that is simply not a major state for U) and I would much rather have a messy 18-column table that at least makes clear the clear homologies (119, 120, and 157–172 are nice homologues of Rb–Xe), and that adequately gets the point that in between 120 and 157 we have a very long transition series, somewhat analogous to the lanthanides and actinides but not quite coinciding with either. Double sharp (talk) 03:33, 29 July 2019 (UTC)
Mosaic near Mendeleev monument
I see this table near the Mendeleev monument in St Petersburg shows group III as Sc Y La Lu Ac. Integrating the rest of the Ln into the main body of the table, other than in a superficial manner, starts to break down with Ce in group IV. But for group III it works very well indeed.
I feel reassured. Sandbh (talk) 01:51, 30 July 2019 (UTC)
- The "mosaic" is from 1934, based on M's 1871 PT. In the mosaic, the blue elements were discovered & added later, usually appearing as a "—" in 1871.See Template:Periodic table (Reihen and periods) for a first transition of PT 1871 from-short-to-long form (long being: using A and B recurring valence-groups; Periods not Reihen).
- re "[the mosaic] shows group III as Sc Y La Lu Ac" is a bit stretched: Lu and Ac were unknown in 1871, the mosaic also shows B in that group III, and Mendeeev mentioned "?Er" in Reihe 10 (1934: appears in VII). "?La" was in Reihe 10, group IV (later La was placed in (nonelement) position "?Di").
- All in all, this grouping was based on chemistry (valence), not physics (blocks, shell filing). And I thought this whole long excercise stemmed from the physical news (energy levels), so I am confused as how Mendeleev could bring an argument into this new approach. -DePiep (talk) 16:03, 30 July 2019 (UTC)
The mosaic is apparently based on the 8th (last) edition of Mendeleev's book Principles of Chemistry (1906). Since the mosaic dates from 1934 the designer presumably attempted to treat the Ln in the spirit of Mendeleev's approach to the periodic table which was, as you say, based more on valence. Mendeleev's table of 1906 showed La under Y, Ce under Zr, and Yb under La. Ce under Zr is kind of OK given we know the existence of Ce(IV) oxide. Yb under La is OK given Yb(III) oxide. In 1934 the mosaic designer had to fit all of the Ln in to the table, and the way they did it seemed to be consistent with the rest of the table. For example Gd ends up in the square under La (but displaced to the right) which would've been consistent with what was known about the Ln at the time.
The whole long group 3 exercise stemmed from Jensen's 1982 article in JChemEd in which he argued for Lu in group 3 on physical, chemical, and electronic property grounds. I think we've gone from Lu to La, to Lu, and now(?) La and Lu (which is how the 1934 mosaic shows it). It's ironic that the picture of the mosaic was on the cover of Scerri's Red Book on the periodic table (2007). So, the answer has been staring at us for the last 12 years, which is quite funny. Sandbh (talk) 01:04, 31 July 2019 (UTC)
- (m): Mv's "1906" would have included the noble gases, wouldn't it? -DePiep (talk) 22:49, 31 July 2019 (UTC)
Yes, indeed. See here. Sandbh (talk) 02:21, 1 August 2019 (UTC)
- I'm still a bit puzzled. here, in section #Tracing_the_idea:_1922_to_2018, you mention this mosaic PT as "1934". But if I understand your post here right, it was a 1906 (scientific) publication by Mendeleev. I've not checked the changes (differences) the mosaic-artist made in 1934 (changes wrt the 1906 form), but anyway why would this "1934" count as a scientific statement? -DePiep (talk) 07:21, 22 August 2019 (UTC)
This site here, which includes a picture of the statue of DIM, notes involvement by the Russian Academy of Sciences. That's about what I'd expect for someone of DIM's stature, and I'm happy with that for reliability purposes. DIM was never able to properly integrate the Ln into his table, even in the 8th (last) edition. Sandbh (talk) 12:44, 23 August 2019 (UTC)
Tracing the idea: 1922 to 2018

In this article from 1952 the author, Coryell, shows a step pyramid style table, with solid and dashed tie lines to show primary and secondary relationships.
Coryell (1912–1971) was an American chemist and one of the discoverers of promethium.
In his table, just two elements are shown by him as having two solid tie lines: yttrium, to La-Ac and to Lu-Ac; and silicon, to Ti-Zr-Hf and to Ge-Sn-Pb.
These days Ti-Zr-Hf-Rf make up group 4 whereas C-Si-Ge-Sn-Pb-Fl make up group 14. Rightly so given group 4 is the first to exhibit characteristic transition metal properties. Coryell also shows solid tie lines between Hf-Th, Ta-Pa, and W-U. These would now be rendered in dashed form.
If his table was mapped to a 32- or 18-column form, group 3 would presumably be shown as bifurcating after Y.

This is consistent with:
| 1922 | Bohr's system, with bifurcations at Na, Mg, and Y |
| 1932 | Bacher & Goudsmith, who show an Lu table but add, "La may well have been put in the place occupied by Lu." |
| 1934 | Mendeleev’s mosaic table |
| 1939 | Foster LS 1939, “Why not modernise textbooks also? I. The periodic table”, Journal of Chemical Education, vol. 16, no. 9, doi.org/10.1021/ed016p409. In group 3 the box under Y is “La {58–70}* Lu”, where * = the rare earths. |
| 1947 | Stedman's planar arrangement of his conic system |
| 1964 | Sanderson RT 1964, "A rational periodic table", Journal of Chemical Education, vol. 41, no. 4, pp. 187–189 |
| 2006 | Silberberg’s table (as discussed) |
| 2007 | Scerri's Red Book cover, which has M's 1934 mosaic in the top right corner |
| 2018 | Scerri and Parson’s suggestion re physical, chemical and electronic properties being inconclusive |
| 2018 | Joshi et al. finding that La, Ac, Lu and Lr behaved the same way in Zintl clusters |
It's good to be able to establish this thread. Sandbh (talk) 02:20, 7 August 2019 (UTC)
Janet's table (1928)
See this table, here, figure 1.
It's from Mazurs (1974, p. 77) who says it’s one of Janet’s tables. I post this with some trepidation, noting Mazurs’ aptitude for fanciful interpretations and embellishments. The reference is Janet C 1928, La classification hélicoidale des éléments chimiques, Beuvais, table VII-17.
There are two group IIIb markers. The 9 o’clock one is Sc Y Lu Lw. The 1130 one is Sc Y.
Group IIIb appears to bifurcate after Y, into an Lu line and an La line. This may be the most elegant representation of group 3B (European) with two branches that I’ve seen.
If you see where B and Al are, it almost looks like group IIIa bifurcates after Al, into the Ga line and the Sc line.
If Mazurs drew it right, this is a remarkable table. Sandbh (talk) 01:51, 22 August 2019 (UTC)
Horizontal triads
In the periodic table there is a recurring sequence of maximum oxidation numbers of +2, +3, and +4. This happens for the following sets of consecutive elements i.e. horizontal triads (P = period):
P +2 +3 +4 ========== 2 Be B C ---------- 3 Mg Al Si ---------- 4 Ca Sc Ti 4 Zn Ga Ge ---------- 5 Sr Y Zr 5 Cd In Sn ---------- 6 Ba La Ce 6 Hg Tl Pb ---------- 7 Ra Ac Th
The recurring sequence doesn't work for Yb (+3) Lu (+3) Hf (+4); nor for No (+3) Lr (+3) Rf (+4). So whereas Sc, Y, La, and Ac are the middle elements of such triads, Lu and Lr are not.
This looks like a good argument for Sc Y La Ac in group 3, or a bifurcated group 3. Sandbh (talk) 03:33, 8 August 2019 (UTC)
- It also probably doesn't work for Cn (+4) Nh (+3) Fl (+2). And in my opinion we should mostly be looking at the elements in question, not their neighbours; otherwise we get into fun questions about the noble gases, whose maximum valence is not 0 from Kr onwards; and while we get a nice F (−1) Ne (0) Na (+1) triad, the next one is Cl (+7) Ar (0) K (+1). (And this in a model of great group trends! ^_^) In other words, we need to look at all characteristic valences, not just maxima, because otherwise O and F don't follow the oxide formulae Mendeleev put above their columns. Double sharp (talk) 03:37, 8 August 2019 (UTC)
Yes, agree re Cn etc.
I thought M based his table on maximum valencies? Sandbh (talk) 07:32, 8 August 2019 (UTC)
- For the most part, yes. But obviously there were some common-sense considerations as well for O and F, and some handwaving for most of "group VIII" (only Ru and Os among the elements he put in it actually form the MO4 that he wrote above that column). Double sharp (talk) 08:02, 8 August 2019 (UTC)
Errr, well yes. I agree. And does this pragmatic approach not also apply to the relevance of the maximum oxidation pattern? That is, you question the bona-fides on the one hand and on the other hand provide arguments supporting the bona fides. Sandbh (talk) 06:49, 10 August 2019 (UTC)
- Because it starts to get a bit strange when the maximum oxidation state is uncharacteristic. For nobelium it is not the most stable state, just like for silver, and most of the late 3d metals are happy to stay at +2 even if we can coax them into going further. And I also don't see why the neighbouring elements should matter much if the relevant ones all show the relevant +3 state. Suppose those reports of HgIV were confirmed: does that weaken the obvious periodicity of Tl going under In? The +4-+5-+6 pattern occurs centred on P, V, As, Nb, Sb, Ta, Bi, Pa, and Db, but not for N, Pr, or Mc; does that make N somehow illegitimate as a congener of the heavier pnictogens? For cases like the chalcogens, I would say that frankly the entire −2/0/+2/+4/+6 complex ought to be considered characteristic, to stop our heads from exploding at O and Po being in the same group. That, which in itself already considers the inert pair and later inert quartet effects, neatly explains why we can dare to put Lv in group VIB even if it should be stuck mostly at +2, with +4 only with the strongest oxidisers, and +6 not at all; because each step from O to S, from S to Se, from Se to Te, from Te to Po, and from Po to Lv, makes individual sense. Such a portrait of Lv seems a reasonable continuation of the trend down from Po, which mostly prefers +2 and +4 and is unhappy to get to +6, after all.
- So I would argue that since +2 is also characteristic for Yb and No, even if they can go up to +3 as well, this should not stop us from considering Yb-Lu-Hf and No-Lr-Rf as well under this umbrella. Gd and Cm can equally well be considered as half-period congeners as well, given the obvious special status of Eu with the half-filled subshell that mimics Mn in the 3d series (a return to the low +2 oxidation state), even if +4 is weaker for Tb than it is for Ce. If we want pragmatism, we might as well consider every manifestation of periodicity, and not restrict ourselves to those following a rule like "pick the maximum", "pick the minimum", "pick only rarities", "pick only the common", etc. Double sharp (talk) 07:27, 10 August 2019 (UTC)
Three kinds of symmetry
For the Ln there are three kinds of symmetry:
1. Klemm’s preference for Ce to Gd, and Tb to Lu. This is based on the configurations of the +3 ions, i.e. f1 to f7; and f8 to f14. It is consistent with the Ln contraction, which starts in Ce and concludes in Lu. It preserves Bent’s +2 +3 +4 periodicity.
2. The old school preference for La to Eu and Gd to Yb, based on concept of the cerite earths and the yttrium earths.
3. The "gadolinium break" symmetry, per Laing (2009), in which the properties of the Ln mirror each other to either side of Gd. For example, the absorption bands of their trivalent cations shift progressively from red to UV in Pr to Gd; and from Gd to Tm they go back to red (Remy 1956):
57 La colourless 70 Lu colourless
58 Ce colourless (uv) 69 Yb colourless (uv)
59 Pr yellow green 68 Tm pale green
60 Nd red violet 67 Er pink
61 Pm violet-pink 66 Ho brownish yellow
62 Sm yellow 65 Dy pale yellow green
63 Eu nearly colourless 64 Tb nearly colourless
------------------------------------------------
64 Gd
colourless
- Klemm W 1929, "Measurements on divalent and quadrivalent rare earth compounds. II. A system of rare earths based on periodic property changes of their ions", Zeitschrift für anorganische und allgemeine Chemie, vol. 184, p. 345
- —— 1930, "Measurements on divalent and quadrivalent rare earth compounds. IV. Supplement to the communication: A system of rare earths", Zeitschrift für anorganische und allgemeine Chemie, vol. 184, p. 29
- Laing M 2009, "Gadolinium: Central metal of the lanthanoids," J. Chem. Educ., vol. 86, no. 2, p. 188, https://pubs.acs.org/doi/abs/10.1021/ed086p188
- Remy H 1956, Treatise on inorganic chemistry, vol. 2, Elsevier, Amsterdam, p. 484
The three symmetries look like this:
+----------+ 1 +----------+ | | | | La Ce………Eu Gd Tb………Yb Lu | | | | | +--------+ 2 +----------+ | | | +-------------3--------------+
- 1= Klemm symmetry; 2 = Old school 3; = Laing.
I think this is the source of much of the confusion that arises out of the group 3 question, including in discussions between @Double sharp: @Droog Andrey: and I.
I reckon the situation is nicely acquitted, using an Lu table, by admitting both La and Lu into group 3:
Sandbh (talk) 04:06, 6 September 2019 (UTC)
- Is there an introduction to this symmetry pattern? Does it have to do with ADOMAH (QM-4-parameter) PT, mentioned in the Left Step PT section above? Can we call a element or an element border the axis of symmetry? (Then: axis of symmetry. Gd-Tb border: Klemm; Eu-Gd border: old school; Gd cell: Laing). -DePiep (talk) 07:23, 8 September 2019 (UTC)
I think the symmetry pattern comes from the oxidation states generally observed for the Ln…
+3 +4 +2 +2 La Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu
…or:
+3 +4 +2 +2 La Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu
It doesn't have anything in particular to do with ADOMAH, that I can see.
Yes, I agree with your setting out of the axis of Ln symmetry as: Gd-Tb border: Klemm; Eu-Gd border: old school; Gd cell: Laing. Sandbh (talk) 22:34, 8 September 2019 (UTC)
Internet database of periodic tables
Here. Sandbh (talk) 04:17, 6 September 2019 (UTC)
Query re development of thread
- In general, I am confused on why my opnion ("nice elegance in Left step PT, while still Mendeleevian" [i.e., periodic law intact]) caused these issues/questions. So far, I understand from all this that Janet's is not wrong, just depicting the periodic law from a different approach. (In the 19th century, had they developed physical experiments instead of or before the actual chemical ones, the original PT would have been a Janet's form with us pondering now why the valences I-VIII so weirdly start in the middle of a period ;-) ). Also, this thread diving into the group 3 issue is unexpected, and I don't think this or any PT presentation can prove modern group 3 statements, we are just trying on "how would 'our' group 3 look like if we use this presentation setup". -DePiep (talk) 17:12, 20 June 2019 (UTC)
Yes, I agree Janet was originally looking at the table from a different i.e. aesthetic approach. Later he realised that his table matched quite well with the electron configuration filling sequence, so much so that he presumed that where there were discrepancies that these must have been errors of measurement and that the true configuration must be as his table said. It turned out that he was wrong; there had been no errors of measurement. In modern day terms this translates to saying the picture with the diagonals, which goes by various names including the n+l rule, the Madelung rule, and aufbau approximation is in fact only approximation and that, as such, it contains 20 errors i.e. it predicts the wrong electron configuration filling sequence 20 times.
The group 3 discussion is just an interesting exploration about the periodic law and the recurrence of properties, and whether this is observed in the case of group 3. To my knowledge we haven't talked about this before and I feel it'd be worthwhile talking it through, to see if there's anything in it. Sandbh (talk) 22:51, 20 June 2019 (UTC)
Oganesson predicted to be a semiconductor
Here. Predicted to have a band gap of 1.5 eV. May qualify as a metalloid, given its intermediate IE, but subject to it having a 1.8 to 2.2 EN; compare 2.2 for radon. Extraordinary! Double sharp: I'll respond re carbonyls shortly. Sandbh (talk) 01:00, 7 August 2019 (UTC)
Full text here. Sandbh (talk) 01:30, 7 August 2019 (UTC)
- What would we do without you to tell us about the latest papers? ^_^
- Well, on the electronegativity scale of Droog Andrey and his collaborator for his wonderful periodic table, Og is at 1.61 while Rn is at 1.94. (To some extent the electronegativity of Kr, Xe, and Rn is overrated by the Pauling scale and should be less than those of the adjacent halogens.) This value is just between those of Fe and Ru on this scale at 1.60 and 1.62 respectively, while Rn's value is between those of Au (1.93) and Ge (1.96). But if you want Pauling-like values: the halogen values on that scale are not too far from the Pauling values (Pauling, F: 3.98, Cl: 3.16, Br: 2.96, I: 2.66, At, 2.2; Kul'sha and Kolevich, F: 4.00, Cl: 3.06, Br: 2.86, I: 2.58, At: 2.09, Ts: 1.76). If we assume that the trend on the Pauling scale of almost identical electronegativities across the pairs Br–Kr, I–Xe, and At–Rn holds for Ts–Og as well, and correct the difference of about 0.1 between the scales, we arrive at an extrapolated Pauling electronegativity of around 1.86 for Og, within your discretionary range. (And actually it is still between those of Fe and Ru on the Pauling scale, which are 1.83 and 2.2 respectively.) Maybe not too surprising, given oganesson's analogies to group 14, and noting that the electronegativities of Si, Ge, Sn, and Pb are all within this discretionary range. Double sharp (talk) 03:15, 8 August 2019 (UTC)
- P.S. You may like 10.1002/anie.200800827, the first paper they cited. Double sharp (talk) 03:32, 8 August 2019 (UTC)
- I have updated the Og article with this new prediction. Double sharp (talk) 03:46, 8 August 2019 (UTC)
- P.S. You may like 10.1002/anie.200800827, the first paper they cited. Double sharp (talk) 03:32, 8 August 2019 (UTC)
Excellent.
Setting aside the first row elements, I can only see four instances of EN varying by more than 0.4, so there is a possibility that Og may have an EN of 1.8 or more. Now, the predicted IE of Og is 860.1 kJ/mol. And the predicted EA of Og at 5.6 kJ/mol. So the expected Pauling EN of Og, using the Mulliken conversion formula of yields a 1.89 value, which is dramatically close to your own 1.86 value.
On this basis the metalloid status for Og is on the upside rather than the downside.
In our Og article should we change its status to "Unknown chemical properties, probably a semiconductor, possibly a metalloid"? Sandbh (talk) 07:27, 8 August 2019 (UTC)
- Doesn't turning Og into a metalloid in our scheme create the same problem that kicked off all the long talks here, viz. that it creates problems for "noble gas" as a category just like At creates problems for "halogen"? Anyway the article still calls Og a "superheavy noble gas" even while simultaneously predicting for it some really ignoble behaviour, so I think that because of the low importance of Ts and Og compared to the other members of their group, people will still be calling them a halogen and a noble gas respectively even if their chemistry is like that of Ga and Sn respectively. Note how the article ends: "...Og is indeed a very unusual group 18 element and an atypical noble gas. However, while breaking with well establish group trends, Og appears to be a typical member of the seventh period, the most prototypical aspect of which may be just that: the breaking of group trends." (Albeit not really until Cn onwards, to be fair.) So while I think At is worrying enough that we made the right move in deleting the halogens as a category (since you can actually use it for things), Og is not something to worry about at the moment.
- (P.S. Even Cn is a reasonable homologue of Hg, after all: the melting and boiling points keep falling and the inertness of the closed-shell configuration follows. Mc and Lv are even better congeners of Bi and Po: it's Nh, Fl, Ts, and Og that start to get truly weird, with perhaps better homology if you arrange the 7th period as ending "111Rg, 112Cn, 117Ts, 118Og, 115Mc, 116Lv, 113Nh, 114Fl"!) Double sharp (talk) 04:30, 9 August 2019 (UTC)
- BTW, with this formula we can calculate Pauling electronegativities for a few more superheavies from the data at Electron affinity (data page): Rg 2.50, Nh 1.71, Mc 1.32, Lv 1.65, Ts 1.97, Og 1.90, Uue 1.23(!), Ubn 1.30(!), Ubu 1.14. Double sharp (talk) 07:52, 8 August 2019 (UTC)
- 2.50 is too high for Rg. In RgB molecule some negative charge is on the boron side. Droog Andrey (talk) 15:42, 9 August 2019 (UTC)
- @Droog Andrey: Well, I did say below not to put too much faith into them. ^_^ Not only do E119 and E120 look obviously wrong (hence my exclamation marks), but Pauling electronegativity does not always match chemical electronegativity (I recall you mentioned Mo and W as such cases, which have overly high electronegativities because Mo–Mo and W–W bonds are strong with high bond order). With an electron affinity for Rg closer to those of Cu and Ag than that of Au I would be more willing to predict something more like 2.0 on the Pauling scale where Cu is 1.90, Ag is 1.93, and Au is 2.54. (And I see you have EN(Cu) = 1.77, EN(Ag) = 1.79, EN(Au) = 1.93, EN(Rg) = 1.99 on your scale with endpoints of EN(Cs) = 0.70 and EN(Ne) = 4.50, noting EN(F) = 4.00 like the original Pauling scale. I guess aurophilicity is responsible for why Au looks so high on the Pauling scale.) Double sharp (talk) 16:02, 9 August 2019 (UTC)
- 2.50 is too high for Rg. In RgB molecule some negative charge is on the boron side. Droog Andrey (talk) 15:42, 9 August 2019 (UTC)
- I wouldn't object to noting this alongside unknown chemical properties in the infobox - we do it elsewhere in line with predictions. Regarding categories, there should not be a problem if Og turns out to be a metalloid; as I understand it, the properties of the first six noble gases are clearly similar to each other and distinguished from other nonmetals, whereas At and even I exhibit some noteworthy differences relative to the lighter group 17 elements. If Og turns out to be an ignoble semiconductor, it can be placed with the metalloids; the similarity of the other noble gases will remain and there is nothing wrong with breaking the uniform characterization in group 18. In any case, relativistic effects will probably cast doubt on the categorization of many SHEs, and it will be a while before Og is investigated and this dilemma is discussed in detail in the literature. I agree that it's no big deal right now. ComplexRational (talk) 09:18, 8 August 2019 (UTC)
I agree with all the feedback. Keep the noble gas group name. Keep showing Og as an unknown chemical properties box. I’d now change its description in the info box to “Unknown chemical properties, traditionally regarded as a noble gas, predicted to be a metallic-looking semi-conductor;[ref] possibly a metalloid[note]”.
Those predicted EN values are remarkable! Sandbh (talk) 11:05, 8 August 2019 (UTC)
- Well, let's not put too much faith into them, since I also got a more reasonable value of 0.86 for E119 by converting a quoted predicted Mulliken value (see ununennium). That is close to 0.82 for Rb, as it ought to be, rather than being higher even than that for Li. ^_^ I've updated the infobox for Og to read "Unknown chemical properties, but traditionally regarded as a noble gas; likely a semiconductor, possibly a post-transition metal or metalloid". (The possibly of metallicity was discussed in a paper you sent me; I've also updated the Nh and Fl articles for their possible metalloid status. All this is in a way expected for the role reversal that seems to happen with the group 13/17 and 14/18 pairs in the 7p elements.) I suppose it's time for more peeving about how IUPAC should really have kept the -ium suffix for Ts and Og, rather than just following the groups, but it's too late now: I will hold out hope that if we must apply those suffixes, we end up doing so for E171 and E172, and not for E167 and E168 which ought to instead be pretty good congeners of In and Sn.
- P.S. Re ComplexRational: iodine is a pretty normal halogen. It does show some incipient metallic behaviour (it is a semiconductor in the direction of its planes and looks pretty shiny), but it patterns very well with F, Cl, and Br. The chemistry of At is however often qualitatively different, which makes it exciting in about the same way Fr isn't (apologies to Douglas Adams for that turn of phrase), but which also makes it a pain to study because you cannot assume that an astatine compound will be carried with its corresponding iodine compound. Its behaviour will often be similar but not quite the same. Part of this may be the extreme tracer scale, because IIRC the tracer chemistry of iodine is quite different from its bulk chemistry and is more like that we see for At. Double sharp (talk) 14:36, 8 August 2019 (UTC)
- Re the peeving: apparently, according to Heinz W. Gäggeler, tennessium and oganessium were in fact proposed, but they were changed to tennessine and oganesson. (I still think it was a mistake to do this for elements whose closest chemical homologues are probably gallium and tin respectively. We could have gotten so many schoolkids interested in relativistic effects when they inevitably asked why the suffixes didn't show the expected consistency!) Double sharp (talk) 15:31, 8 August 2019 (UTC)
Thanks for all that. I had a go at fine-tuning the Og info box, re its element category. Sandbh (talk) 04:02, 9 August 2019 (UTC)
- Is the term "noble gas" going to end up becoming obsolete sometime this century because of Og? It would be interesting. ― Дрейгорич / Dreigorich Talk 22:06, 10 August 2019 (UTC)
- @Дрейгорич: I seriously doubt it. He, Ne, Ar, Kr, Xe, and Rn are so much more common and they are good noble gases; the fact that At is probably metallic hasn't done anything to kill off the term "halogen", because it is so useful when describing the set {F, Cl, Br, I} even if At has an ambiguous status. Even if we somehow discover some metastable isotope of Og that we can actually use and make it the next semiconductor wonder-material I suspect "noble gas" will just keep meaning He through Rn and Og will be called something else, probably a metalloid or metal. Double sharp (talk) 04:11, 11 August 2019 (UTC)
- Very interesting. Thanks. ― Дрейгорич / Dreigorich Talk 04:20, 11 August 2019 (UTC)
- @Дрейгорич: I seriously doubt it. He, Ne, Ar, Kr, Xe, and Rn are so much more common and they are good noble gases; the fact that At is probably metallic hasn't done anything to kill off the term "halogen", because it is so useful when describing the set {F, Cl, Br, I} even if At has an ambiguous status. Even if we somehow discover some metastable isotope of Og that we can actually use and make it the next semiconductor wonder-material I suspect "noble gas" will just keep meaning He through Rn and Og will be called something else, probably a metalloid or metal. Double sharp (talk) 04:11, 11 August 2019 (UTC)
Hmm. If At is a metal it won't upset "halogen" as a group name. If Og is a metal then calling group 18 the noble gases won't work anymore, nor will "noble nonmetals". What then will group 18 be called? Sandbh (talk) 05:26, 12 September 2019 (UTC)
- Probably "noble gases" will simply mean the first six members of group 18 only. Oganesson can, I suppose, still be an aerogen: with its strong radioactivity, and a predicted melting point of about 50 °C that is very low for a metal, it will presumably volatilise itself even more effectively than Po. Double sharp (talk) 06:40, 12 September 2019 (UTC)
- I propose "octogen" but helium's a problem. Hmmm... "telogen", meaning "end" or "last" (of its period), perhaps? ― Дрейгорич / Dreigorich Talk 07:25, 12 September 2019 (UTC)
- You could always join the helium-in-group-2 club (see the periodic table on my userpage)! ^_^ Telogen is not bad etymologically, but it does have another meaning in another field. Double sharp (talk) 14:53, 12 September 2019 (UTC)
- Ahahaha, thanks, but no thanks. I'm on team "Why Is Hydrogen Even Above Lithium In The First Place When It Has Almost Nothing In Common With The Other Group 1 Elements" myself. ― Дрейгорич / Dreigorich Talk 06:31, 13 September 2019 (UTC)
- You could always join the helium-in-group-2 club (see the periodic table on my userpage)! ^_^ Telogen is not bad etymologically, but it does have another meaning in another field. Double sharp (talk) 14:53, 12 September 2019 (UTC)
- I propose "octogen" but helium's a problem. Hmmm... "telogen", meaning "end" or "last" (of its period), perhaps? ― Дрейгорич / Dreigorich Talk 07:25, 12 September 2019 (UTC)
The position of hydrogen in group 1 is reasonably well settled. Its usual valency is +1. Like lithium, it has a significant covalent chemistry. It can stand in for alkali metals in typical alkali metal structures. It is capable of forming alloy-like hydrides, featuring metallic bonding, with some transition metals. Its anomalous properties can be explained by its electron configuration. Sandbh (talk) 10:35, 13 September 2019 (UTC)
- Oh right, chemical properties. Forgot about those. I was thinking "we have metals on this side and then we have a nonmetal on the wrong side". ― Дрейгорич / Dreigorich Talk 05:45, 14 September 2019 (UTC)









