Oleamide
| |
| Names | |
|---|---|
| Preferred IUPAC name
(9Z)-Octadec-9-enamide | |
| Other names
oleoyl-amide
Oleylamide 9-Octadecenamide (Z)-9-Octadecenamide 9,10-Octadecenoamide Oleic acid amide Cis-9,10-octadecenoamide | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.005.550 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C18H35NO | |
| Molar mass | 281.484 g·mol−1 |
| Appearance | Creamy solid[1] |
| Density | 0.879 g/cm3 |
| Melting point | 70 °C (158 °F; 343 K)<[2] |
| Boiling point | > 200 °C (392 °F; 473 K)[1] |
| Insoluble[1] | |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Flash point | > 200 °C (392 °F; 473 K)[1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
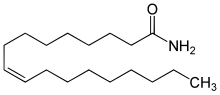
Oleamide is an organic compound with the formula CH3(CH2)7CH=CH(CH2)7CONH2.[3] It is the amide derived from the fatty acid oleic acid. It is a colorless waxy solid and occurs in nature. Sometimes labeled as a fatty acid primary amide (FAPA), it is biosynthesized from N-oleoylglycine.[4]
Biochemical and medical aspects
[edit]In terms of natural occurrence, oleamide was first detected in human plasma. It was later shown to accumulate in the cerebrospinal fluid during sleep deprivation and induces sleep in animals.[4][5]
It has been considered as a treatment for mood and sleep disorders, as well as cannabinoid-regulated depression.[6][7]
In terms of its sleep inducing effects, it is speculated that oleamide interacts with multiple neurotransmitter systems.[8][9] Some in-vitro studies show that cis-oleamide is an agonist for the cannabinoid receptor CB-1 with an affinity around 8 micromolar.[10] However, given oleamide's relatively low affinity for CB-1 and uncertainty about the concentration and biological role of oleamide in-vivo, it has been argued that it is premature to classify oleamide as an endocannabinoid.[11] At larger doses oleamide can lower the body temperature of mice by about 2 degrees, with the effect lasting about two hours.[9] The mechanism for this remains unknown.[9]
Oleamide has been found to enhance PPARα-dependent increase in doublecortin, a marker of neurogenesis in the hippocampus[12]
Oleamide is rapidly metabolized by fatty acid amide hydrolase (FAAH), the same enzyme that metabolizes anandamide.[13] It has been postulated that some effects of oleamide are caused by increased concentrations of anandamide brought about through the inhibition of FAAH.[9]
It has been claimed that oleamide increases the activity of choline acetyltransferase, an enzyme that is critical in the production of acetylcholine.[14]
Other occurrences
[edit]Oleamide has been found in Ziziphus jujuba, also known as Jujube fruit.[14]
Synthetic oleamide has a variety of industrial uses, including as a lubricant.[15]
Oleamide was found to be leaching out of polypropylene plastics in laboratory experiments, affecting experimental results.[16] Since polypropylene is used in a wide number of food containers such as those for yogurt, the problem is being studied.[17]
Oleamide is "one of the most frequent non-cannabinoid ingredients associated with Spice products."[18] Analysis of 44 products synthetic cannabinoid revealed oleamide in 7 of the products tested.[19]
See also
[edit]References
[edit]- ^ a b c d Oleamide at chemicalland21.com
- ^ "(9Z)-9-Octadecenamide | C18H35NO | ChemSpider".
- ^ "Oleamide".
- ^ a b McKinney, Michele K.; Cravatt, Benjamin F. (June 2005). "Structure and Function of Fatty Acid Amide Hydrolase". Annual Review of Biochemistry. 74 (1): 411–432. doi:10.1146/annurev.biochem.74.082803.133450. PMID 15952893. S2CID 23571858.
- ^ Cravatt, B.; Prospero-Garcia, O; Siuzdak, G; Gilula, N.; Henriksen, S.; Boger, D.; Lerner, R. (9 June 1995). "Chemical characterization of a family of brain lipids that induce sleep". Science. 268 (5216): 1506–1509. Bibcode:1995Sci...268.1506C. doi:10.1126/science.7770779. PMID 7770779. S2CID 32070839.
- ^ Methods of treating anxiety and mood disorders with oleamide – US Patent 6359010 Archived 2011-06-12 at the Wayback Machine
- ^ Mechoulam, Raphael; Fride, Ester; Hanu, Lumir; Sheskin, Tzviel; Bisogno, Tiziana; Di Marzo, Vincenzo; Bayewitch, Michael; Vogel, Zvi (September 1997). "Anandamide may mediate sleep induction". Nature. 389 (6646): 25–26. Bibcode:1997Natur.389R..25M. doi:10.1038/37891. PMID 9288961. S2CID 26371103.
- ^ Fedorova, I; Hashimoto, A; Fecik, RA; Hedrick, MP; Hanus, LO; Boger, DL; Rice, KC; Basile, AS (October 2001). "Behavioral evidence for the interaction of oleamide with multiple neurotransmitter systems". The Journal of Pharmacology and Experimental Therapeutics. 299 (1): 332–42. PMID 11561096.
- ^ a b c d Hiley, C. Robin; Hoi, Pui Man (13 April 2007). "Oleamide: A Fatty Acid Amide Signaling Molecule in the Cardiovascular System?: OLEAMIDE". Cardiovascular Drug Reviews. 25 (1): 46–60. doi:10.1111/j.1527-3466.2007.00004.x. PMID 17445087.
- ^ Leggett, JD; Aspley, S; Beckett, SR; D'Antona, AM; Kendall, DA; Kendall, DA (January 2004). "Oleamide is a selective endogenous agonist of rat and human CB1 cannabinoid receptors". British Journal of Pharmacology. 141 (2): 253–62. doi:10.1038/sj.bjp.0705607. PMC 1574194. PMID 14707029.
- ^ Fowler, Christopher J (January 2004). "Oleamide: a member of the endocannabinoid family?: Commentary". British Journal of Pharmacology. 141 (2): 195–196. doi:10.1038/sj.bjp.0705608. PMC 1574195. PMID 14691053.
- ^ Roy, Avik; Kundu, Madhuchhanda; Chakrabarti, Sudipta; Patel, Dhruv R; Pahan, Kalipada (December 2021). "Oleamide, a Sleep-Inducing Supplement, Upregulates Doublecortin in Hippocampal Progenitor Cells via PPARα". J Alzheimers Dis. 84 (4): 1747–1762. doi:10.3233/JAD-215124. PMC 10075226. PMID 34744082.
- ^ Maurelli, Stefano; Bisogno, Tiziana; De Petrocellis, Luciano; Di Luccia, Aldo; Marino, Gennaro; Di Marzo, Vincenzo (11 December 1995). "Two novel classes of neuroactive fatty acid amides are substrates for mouse neuroblastoma 'anandamide amidohydrolase'". FEBS Letters. 377 (1): 82–86. Bibcode:1995FEBSL.377...82M. doi:10.1016/0014-5793(95)01311-3. PMID 8543025. S2CID 7461775.
- ^ a b Heo, Ho-Jin; Park, Young-June; Suh, Young-Min; Choi, Soo-Jung; Kim, Mi-Jeong; Cho, Hong-Yon; Chang, Yun-Jeong; Hong, Bumshik; Kim, Hye-Kyung; Kim, Eunki; Kim, Chang-Ju; Kim, Byung-Gee; Shin, Dong-Hoon (January 2003). "Effects of Oleamide on Choline Acetyltransferase and Cognitive Activities". Bioscience, Biotechnology, and Biochemistry. 67 (6): 1284–1291. doi:10.1271/bbb.67.1284. PMID 12843655.
- ^ Surfactants : Westco Oleamide a Slip Agent In Polyethylene Films Archived January 27, 2007, at the Wayback Machine
- ^ McDonald, G. R.; Hudson, A. L.; Dunn, S. M. J.; You, H.; Baker, G. B.; Whittal, R. M.; Martin, J. W.; Jha, A.; Edmondson, D. E.; Holt, A. (7 November 2008). "Bioactive Contaminants Leach from Disposable Laboratory Plasticware". Science. 322 (5903): 917. Bibcode:2008Sci...322..917M. doi:10.1126/science.1162395. PMID 18988846. S2CID 35526901.
- ^ Mittelstaedt, Martin (6 November 2008). "Researchers Raise Alarm After Chemical Leak Found In Common Plastic". Globe and Mail. Retrieved 10 June 2013.
- ^ Fattore, Liana; Fratta, Walter (21 September 2011). "Beyond THC: The New Generation of Cannabinoid Designer Drugs". Frontiers in Behavioral Neuroscience. 5: 60. doi:10.3389/fnbeh.2011.00060. PMC 3187647. PMID 22007163.
- ^ Uchiyama, Nahoko; Kikura-Hanajiri, Ruri; Ogata, Jun; Goda, Yukihiro (May 2010). "Chemical analysis of synthetic cannabinoids as designer drugs in herbal products". Forensic Science International. 198 (1–3): 31–38. doi:10.1016/j.forsciint.2010.01.004. PMID 20117892.

