Carbon monoxide
| |||
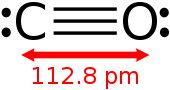
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Carbon monoxide
| |||
| Other names
Carbonic oxide gas
Carbon protoxide Oxide of carbon Protoxide of carbon Carbonous oxide Carbonous acid gas Carbon(II) oxide Breath of carbon Oxygenated carbon Carbate Carbonyl Water gas Hydrocarbon gas Fuel gas Rauchgas Carbonic inflammable air Heavy inflammable air White damp Fire Damp Powder Gas Illuminating gas Dowson gas Mond gas Power gas Producer gas Blast furnace gas Coal gas Phlogiston Car gas | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| 3587264 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.010.118 | ||
| EC Number |
| ||
| 421 | |||
| KEGG | |||
| MeSH | Carbon+monoxide | ||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
| UN number | 1016 | ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| CO | |||
| Molar mass | 28.010 g·mol−1 | ||
| Appearance | Colorless | ||
| Odor | Odorless | ||
| Density |
| ||
| Melting point | −205.02 °C (−337.04 °F; 68.13 K) | ||
| Boiling point | −191.5 °C (−312.7 °F; 81.6 K) | ||
| 27.6 mg/L (25 °C) | |||
| Solubility | soluble in chloroform, acetic acid, ethyl acetate, ethanol, ammonium hydroxide, benzene | ||
Henry's law
constant (kH) |
1.04 atm·m3/mol | ||
| −9.8·10−6 cm3/mol | |||
Refractive index (nD)
|
1.0003364 | ||
| 0.122 D | |||
| Thermochemistry | |||
Heat capacity (C)
|
29.1 J/(K·mol) | ||
Std molar
entropy (S⦵298) |
197.7 J/(K·mol) | ||
Std enthalpy of
formation (ΔfH⦵298) |
−110.5 kJ/mol | ||
Std enthalpy of
combustion (ΔcH⦵298) |
−283.0 kJ/mol | ||
| Pharmacology | |||
| V04CX08 (WHO) | |||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
Poisonous by inhalation[1] | ||
| GHS labelling: | |||
  
| |||
| Danger | |||
| H220, H331, H360, H372, H420 | |||
| P201, P202, P210, P251, P260, P261, P264, P270, P281, P304+P340, P308+P313, P311, P314, P321, P377, P381, P403, P403+P233, P405, P501 | |||
| NFPA 704 (fire diamond) | |||
| Flash point | −191 °C (−311.8 °F; 82.1 K) | ||
| 609 °C (1,128 °F; 882 K) | |||
| Explosive limits | 12.5–74.2% | ||
| Lethal dose or concentration (LD, LC): | |||
LC50 (median concentration)
|
| ||
LCLo (lowest published)
|
| ||
| NIOSH (US health exposure limits):[1] | |||
PEL (Permissible)
|
TWA 50 ppm (55 mg/m3) | ||
REL (Recommended)
|
| ||
IDLH (Immediate danger)
|
1200 ppm | ||
| Safety data sheet (SDS) | ICSC 0023 | ||
| Related compounds | |||
Other anions
|
Carbon monosulfide | ||
Other cations
|
Silicon monoxide Germanium monoxide Tin(II) oxide Lead(II) oxide | ||
Related carbon oxides
|
Carbon dioxide Carbon suboxide Oxocarbons | ||
| Supplementary data page | |||
| Carbon monoxide (data page) | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Carbon monoxide (chemical formula CO) is a poisonous, flammable gas that is colorless, odorless, tasteless, and slightly less dense than air. Carbon monoxide consists of one carbon atom and one oxygen atom connected by a triple bond. It is the simplest carbon oxide. In coordination complexes, the carbon monoxide ligand is called carbonyl. It is a key ingredient in many processes in industrial chemistry.[5]
The most common source of carbon monoxide is the partial combustion of carbon-containing compounds. Numerous environmental and biological sources generate carbon monoxide. In industry, carbon monoxide is important in the production of many compounds, including drugs, fragrances, and fuels.[6] Upon emission into the atmosphere, carbon monoxide affects several processes that contribute to climate change.[7]
Indoors CO is one of the most acutely toxic contaminants affecting indoor air quality. CO may be emitted from tobacco smoke and generated from malfunctioning fuel burning stoves (wood, kerosene, natural gas, propane) and fuel burning heating systems (wood, oil, natural gas) and from blocked flues connected to these appliances.[8] Carbon monoxide poisoning is the most common type of fatal air poisoning in many countries.[9][8][10]
Carbon monoxide has important biological roles across phylogenetic kingdoms. It is produced by many organisms, including humans. In mammalian physiology, carbon monoxide is a classical example of hormesis where low concentrations serve as an endogenous neurotransmitter (gasotransmitter) and high concentrations are toxic resulting in carbon monoxide poisoning. It is isoelectronic with both cyanide anion CN− and molecular nitrogen N2.
Physical and chemical properties
[edit]Carbon monoxide is the simplest oxocarbon and is isoelectronic with other triply bonded diatomic species possessing 10 valence electrons, including the cyanide anion, the nitrosonium cation, boron monofluoride and molecular nitrogen. It has a molar mass of 28.0, which, according to the ideal gas law, makes it slightly less dense than air, whose average molar mass is 28.8.
The carbon and oxygen are connected by a triple bond that consists of a net two pi bonds and one sigma bond. The bond length between the carbon atom and the oxygen atom is 112.8 pm.[11][12] This bond length is consistent with a triple bond, as in molecular nitrogen (N2), which has a similar bond length (109.76 pm) and nearly the same molecular mass. Carbon–oxygen double bonds are significantly longer, 120.8 pm in formaldehyde, for example.[13] The boiling point (82 K) and melting point (68 K) are very similar to those of N2 (77 K and 63 K, respectively). The bond-dissociation energy of 1072 kJ/mol is stronger than that of N2 (942 kJ/mol) and represents the strongest chemical bond known.[14]
The ground electronic state of carbon monoxide is a singlet state[15] since there are no unpaired electrons.
| Temperature (°C) | Temperature (K) | Density (kg/m3) | Specific heat (J/g °C) | Dynamic viscosity (cg/m s) | Kinematic viscosity (cm2/s) | Thermal conductivity (cW/m °C) | Thermal diffusivity (cm2/s) | Prandtl number |
|---|---|---|---|---|---|---|---|---|
| -73.15 | 200 | 1.6888 | 1.045 | 1.27 | 0.0752 | 1.7 | 0.0963 | 0.781 |
| -53.15 | 220 | 1.5341 | 1.044 | 1.37 | 0.0893 | 1.9 | 0.119 | 0.753 |
| -33.15 | 240 | 1.4055 | 1.043 | 1.47 | 0.105 | 2.06 | 0.141 | 0.744 |
| -13.15 | 260 | 1.2967 | 1.043 | 1.57 | 0.121 | 2.21 | 0.163 | 0.741 |
| 6.85 | 280 | 1.2038 | 1.042 | 1.66 | 0.138 | 2.36 | 0.188 | 0.733 |
| 26.85 | 300 | 1.1233 | 1.043 | 1.75 | 0.156 | 2.5 | 0.213 | 0.73 |
| 46.85 | 320 | 1.0529 | 1.043 | 1.84 | 0.175 | 2.63 | 0.239 | 0.73 |
| 66.85 | 340 | 0.9909 | 1.044 | 1.93 | 0.195 | 2.78 | 0.269 | 0.725 |
| 86.85 | 360 | 0.9357 | 1.045 | 2.02 | 0.216 | 2.91 | 0.298 | 0.725 |
| 106.85 | 380 | 0.8864 | 1.047 | 2.1 | 0.237 | 3.05 | 0.329 | 0.729 |
| 126.85 | 400 | 0.8421 | 1.049 | 2.18 | 0.259 | 3.18 | 0.36 | 0.719 |
| 176.85 | 450 | 0.7483 | 1.055 | 2.37 | 0.317 | 3.5 | 0.443 | 0.714 |
| 226.85 | 500 | 0.67352 | 1.065 | 2.54 | 0.377 | 3.81 | 0.531 | 0.71 |
| 276.85 | 550 | 0.61226 | 1.076 | 2.71 | 0.443 | 4.11 | 0.624 | 0.71 |
| 326.85 | 600 | 0.56126 | 1.088 | 2.86 | 0.51 | 4.4 | 0.721 | 0.707 |
| 376.85 | 650 | 0.51806 | 1.101 | 3.01 | 0.581 | 4.7 | 0.824 | 0.705 |
| 426.85 | 700 | 0.48102 | 1.114 | 3.15 | 0.655 | 5 | 0.933 | 0.702 |
| 476.85 | 750 | 0.44899 | 1.127 | 3.29 | 0.733 | 5.28 | 1.04 | 0.702 |
| 526.85 | 800 | 0.42095 | 1.14 | 3.43 | 0.815 | 5.55 | 1.16 | 0.705 |
Bonding and dipole moment
[edit]The strength of the C-O bond in carbon monoxide is indicated by the high frequency of its vibration, 2143 cm-1.[18] For comparison, organic carbonyls such as ketones and esters absorb at around 1700 cm-1.
Carbon and oxygen together have a total of 10 electrons in the valence shell. Following the octet rule for both carbon and oxygen, the two atoms form a triple bond, with six shared electrons in three bonding molecular orbitals, rather than the usual double bond found in organic carbonyl compounds. Since four of the shared electrons come from the oxygen atom and only two from carbon, one bonding orbital is occupied by two electrons from oxygen, forming a dative or dipolar bond. This causes a C←O polarization of the molecule, with a small negative charge on carbon and a small positive charge on oxygen. The other two bonding orbitals are each occupied by one electron from carbon and one from oxygen, forming (polar) covalent bonds with a reverse C→O polarization since oxygen is more electronegative than carbon. In the free carbon monoxide molecule, a net negative charge δ– remains at the carbon end and the molecule has a small dipole moment of 0.122 D.[19]
The molecule is therefore asymmetric: oxygen is more electron dense than carbon and is also slightly positively charged compared to carbon being negative.

Carbon monoxide has a computed fractional bond order of 2.6, indicating that the "third" bond is important but constitutes somewhat less than a full bond.[20] Thus, in valence bond terms, –C≡O+ is the most important structure, while :C=O is non-octet, but has a neutral formal charge on each atom and represents the second most important resonance contributor. Because of the lone pair and divalence of carbon in this resonance structure, carbon monoxide is often considered to be an extraordinarily stabilized carbene.[21] Isocyanides are compounds in which the O is replaced by an NR (R = alkyl or aryl) group and have a similar bonding scheme.
If carbon monoxide acts as a ligand, the polarity of the dipole may reverse with a net negative charge on the oxygen end, depending on the structure of the coordination complex.[22] See also the section "Coordination chemistry" below.
Bond polarity and oxidation state
[edit]Theoretical and experimental studies show that, despite the greater electronegativity of oxygen, the dipole moment points from the more-negative carbon end to the more-positive oxygen end.[23][24] The three bonds are in fact polar covalent bonds that are strongly polarized. The calculated polarization toward the oxygen atom is 71% for the σ-bond and 77% for both π-bonds.[25]
The oxidation state of carbon in carbon monoxide is +2 in each of these structures. It is calculated by counting all the bonding electrons as belonging to the more electronegative oxygen. Only the two non-bonding electrons on carbon are assigned to carbon. In this count, carbon then has only two valence electrons in the molecule compared to four in the free atom.
Occurrence
[edit]Carbon monoxide occurs in various natural and artificial environments. Photochemical degradation of plant matter for example generates an estimated 60 million tons/year.[27] Typical concentrations in parts per million are as follows:
| Concentration (ppmv[a]) | Source |
|---|---|
| 0.1 | Natural atmosphere level (MOPITT)[29] |
| 0.5–5 | Average level in homes[30] |
| 5–15 | Near properly adjusted gas stoves in homes, modern vehicle exhaust emissions[31][citation needed] |
| 17 | Atmosphere of Venus |
| 100–200 | Exhaust from automobiles in the Mexico City central area in 1975[32] |
| 700 | Atmosphere of Mars |
| <1,000 | Car exhaust fumes after passing through catalytic converter[33] |
| 5,000 | Exhaust from a home wood fire[34] |
| 30,000–100,000 | Undiluted warm car exhaust without a catalytic converter[33] |
| |
Atmospheric presence
[edit]
Carbon monoxide (CO) is present in small amounts (about 80 ppb) in the Earth's atmosphere. Most of the rest comes from chemical reactions with organic compounds emitted by human activities and natural origins due to photochemical reactions in the troposphere that generate about 5 × 1012 kilograms per year.[35] Other natural sources of CO include volcanoes, forest and bushfires, and other miscellaneous forms of combustion such as fossil fuels.[36] Small amounts are also emitted from the ocean, and from geological activity because carbon monoxide occurs dissolved in molten volcanic rock at high pressures in the Earth's mantle.[37] Because natural sources of carbon monoxide vary from year to year, it is difficult to accurately measure natural emissions of the gas.
Carbon monoxide has an indirect effect on radiative forcing by elevating concentrations of direct greenhouse gases, including methane and tropospheric ozone. CO can react chemically with other atmospheric constituents (primarily the hydroxyl radical, •OH) that would otherwise destroy methane.[38] Through natural processes in the atmosphere, it is oxidized to carbon dioxide and ozone. Carbon monoxide is short-lived in the atmosphere (with an average lifetime of about one to two months), and spatially variable in concentration.[39]
Due to its long lifetime in the mid-troposphere, carbon monoxide is also used as a tracer for pollutant plumes.[40]
Astronomy
[edit]Beyond Earth, carbon monoxide is the second-most common diatomic molecule in the interstellar medium, after molecular hydrogen. Because of its asymmetry, this polar molecule produces far brighter spectral lines than the hydrogen molecule, making CO much easier to detect. Interstellar CO was first detected with radio telescopes in 1970. It is now the most commonly used tracer of molecular gas in general in the interstellar medium of galaxies, as molecular hydrogen can only be detected using ultraviolet light, which requires space telescopes. Carbon monoxide observations provide much of the information about the molecular clouds in which most stars form.[41][42]
Beta Pictoris, the second brightest star in the constellation Pictor, shows an excess of infrared emission compared to normal stars of its type, which is caused by large quantities of dust and gas (including carbon monoxide)[43][44] near the star.
In the atmosphere of Venus carbon monoxide occurs as a result of the photodissociation of carbon dioxide by electromagnetic radiation of wavelengths shorter than 169 nm. It has also been identified spectroscopically on the surface of Neptune's moon Triton.[45]
Solid carbon monoxide is a component of comets.[46] The volatile or "ice" component of Halley's Comet is about 15% CO.[47] At room temperature and at atmospheric pressure, carbon monoxide is actually only metastable (see Boudouard reaction) and the same is true at low temperatures where CO and CO
2 are solid, but nevertheless it can exist for billions of years in comets. There is very little CO in the atmosphere of Pluto, which seems to have been formed from comets. This may be because there is (or was) liquid water inside Pluto.
Carbon monoxide can react with water to form carbon dioxide and hydrogen:
- CO + H2O → H
2 + CO
2
This is called the water-gas shift reaction when occurring in the gas phase, but it can also take place (very slowly) in an aqueous solution. If the hydrogen partial pressure is high enough (for instance in an underground sea), formic acid will be formed:
- CO + H2O → HCOOH
These reactions can take place in a few million years even at temperatures such as found on Pluto.[48]
Pollution and health effects
[edit]Urban pollution
[edit]Carbon monoxide is a temporary atmospheric pollutant in some urban areas, chiefly from the exhaust of internal combustion engines (including vehicles, portable and back-up generators, lawnmowers, power washers, etc.), but also from incomplete combustion of various other fuels (including wood, coal, charcoal, oil, paraffin, propane, natural gas, and trash).
Large CO pollution events can be observed from space over cities.[49]
Role in ground level ozone formation
[edit]Carbon monoxide is, along with aldehydes, part of the series of cycles of chemical reactions that form photochemical smog. It reacts with hydroxyl radical (•OH) to produce a radical intermediate •HOCO, which rapidly transfers its radical hydrogen to O2 to form peroxy radical (HO2•) and carbon dioxide (CO2).[50] Peroxy radical subsequently reacts with nitrogen oxide (NO) to form nitrogen dioxide (NO2) and hydroxyl radical. NO2 gives O(3P) via photolysis, thereby forming O3 following reaction with O2. Since hydroxyl radical is formed during the formation of NO2, the balance of the sequence of chemical reactions starting with carbon monoxide and leading to the formation of ozone is:
- CO + 2O2 + hν → CO2 + O3
(where hν refers to the photon of light absorbed by the NO2 molecule in the sequence)
Although the creation of NO2 is the critical step leading to low level ozone formation, it also increases this ozone in another, somewhat mutually exclusive way, by reducing the quantity of NO that is available to react with ozone.[51]
Indoor air pollution
[edit]Carbon monoxide is one of the most acutely toxic indoor air contaminants. Carbon monoxide may be emitted from tobacco smoke and generated from malfunctioning fuel burning stoves (wood, kerosene, natural gas, propane) and fuel burning heating systems (wood, oil, natural gas) and from blocked flues connected to these appliances.[8] In developed countries the main sources of indoor CO emission come from cooking and heating devices that burn fossil fuels and are faulty, incorrectly installed or poorly maintained.[52] Appliance malfunction may be due to faulty installation or lack of maintenance and proper use.[8] In low- and middle-income countries the most common sources of CO in homes are burning biomass fuels and cigarette smoke.[52]
Mining
[edit]Miners refer to carbon monoxide as "whitedamp" or the "silent killer". It can be found in confined areas of poor ventilation in both surface mines and underground mines. The most common sources of carbon monoxide in mining operations are the internal combustion engine and explosives; however, in coal mines, carbon monoxide can also be found due to the low-temperature oxidation of coal.[53] The idiom "Canary in the coal mine" pertained to an early warning of a carbon monoxide presence.[54]
Health effects
[edit]Carbon monoxide poisoning is the most common type of fatal air poisoning in many countries. Acute exposure can also lead to long-term neurological effects such as cognitive and behavioural changes. Severe CO poisoning may lead to unconsciousness, coma and death. Chronic exposure to low concentrations of carbon monoxide may lead to lethargy, headaches, nausea, flu-like symptoms and neuropsychological and cardiovascular issues.[9][8][10]
Chemistry
[edit]Carbon monoxide has a wide range of functions across all disciplines of chemistry. The four premier categories of reactivity involve metal-carbonyl catalysis, radical chemistry, cation and anion chemistries.[55]
Coordination chemistry
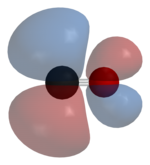
[edit]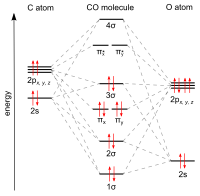

Most metals form coordination complexes containing covalently attached carbon monoxide. These derivatives, which are called metal carbonyls, tend to be more robust when the metal is in lower oxidation states. For example iron pentacarbonyl (Fe(CO)5) is an air-stable, distillable liquid. Nickel carbonyl is an example of a metal carbonyl complex that forms by the direct combination of carbon monoxide with the metal:C. Elschenbroich (2006). Organometallics. VCH. ISBN 978-3-527-29390-2.
- Ni + 4 CO → Ni(CO)4 (1 bar, 55 °C)
These volatile complexes are often highly toxic. Some metal–CO complexes are prepared by decarbonylation of organic solvents, not from CO. For instance, iridium trichloride and triphenylphosphine react in boiling 2-methoxyethanol or DMF to afford IrCl(CO)(PPh3)2.
As a ligand, CO binds through carbon, forming a kind of triple bond. The lone pair on the carbon atom donates electron density to form a M-CO sigma bond. The two π* orbitals on CO bind to filled metal orbitals. The effect is related to the Dewar-Chatt-Duncanson model. The effects of the quasi-triple M-C bond is reflected in the infrared spectrum of these complexes. Whereas free CO vibrates at 2143 cm-1, its complexes tend to absorb near 1950 cm-1.

Organic and main group chemistry
[edit]In the presence of strong acids, alkenes react with carboxylic acids. Hydrolysis of this species (an acylium ion) gives the carboxylic acid, a net process known as the Koch–Haaf reaction.[56] In the Gattermann–Koch reaction, arenes are converted to benzaldehyde derivatives in the presence of CO, AlCl3, and HCl.[57]
A mixture of hydrogen gas and CO reacts with alkenes to give aldehydes. The process requires the presence of metal catalysts.[58]
With main group reagents, CO undergoes several noteworthy reactions. Chlorination of CO is the industrial route to the important compound phosgene. With borane CO forms the adduct H3BCO, which is isoelectronic with the acylium cation [H3CCO]+. CO reacts with sodium to give products resulting from C−C coupling such as sodium acetylenediolate 2Na+
·C
2O2−
2. It reacts with molten potassium to give a mixture of an organometallic compound, potassium acetylenediolate 2K+
·C
2O2−
2, potassium benzenehexolate 6K+
C
6O6−
6,[59] and potassium rhodizonate 2K+
·C
6O2−
6.[60]
The compounds cyclohexanehexone or triquinoyl (C6O6) and cyclopentanepentone or leuconic acid (C5O5), which so far have been obtained only in trace amounts, can be regarded as polymers of carbon monoxide. At pressures exceeding 5 GPa, carbon monoxide converts to polycarbonyl, a solid polymer that is metastable at atmospheric pressure but is explosive.[61][62]
Laboratory preparation
[edit]Carbon monoxide is conveniently produced in the laboratory by the dehydration of formic acid or oxalic acid, for example with concentrated sulfuric acid.[56][57][63] Another method is heating an intimate mixture of powdered zinc metal and calcium carbonate, which releases CO and leaves behind zinc oxide and calcium oxide:
- Zn + CaCO3 → ZnO + CaO + CO
Silver nitrate and iodoform also afford carbon monoxide:
- CHI3 + 3AgNO3 + H2O → 3HNO3 + CO + 3AgI
Finally, metal oxalate salts release CO upon heating, leaving a carbonate as byproduct:
- Na
2C
2O
4 → Na
2CO
3 + CO
Production
[edit]Thermal combustion is the most common source for carbon monoxide. Carbon monoxide is produced from the partial oxidation of carbon-containing compounds; it forms when there is not enough oxygen to produce carbon dioxide (CO2), such as when operating a stove or an internal combustion engine in an enclosed space.
A large quantity of CO byproduct is formed during the oxidative processes for the production of chemicals. For this reason, the process off-gases have to be purified.
Many methods have been developed for carbon monoxide production.[64]
Industrial production
[edit]A major industrial source of CO is producer gas, a mixture containing mostly carbon monoxide and nitrogen, formed by combustion of carbon in air at high temperature when there is an excess of carbon. In an oven, air is passed through a bed of coke. The initially produced CO2 equilibrates with the remaining hot carbon to give CO.[65] The reaction of CO2 with carbon to give CO is described as the Boudouard reaction.[66] Above 800 °C, CO is the predominant product:
- CO2 (g) + C (s) → 2 CO (g) (ΔHr = 170 kJ/mol)
Another source is "water gas", a mixture of hydrogen and carbon monoxide produced via the endothermic reaction of steam and carbon:
- H2O (g) + C (s) → H2 (g) + CO (g) (ΔHr = 131 kJ/mol)
Other similar "synthesis gases" can be obtained from natural gas and other fuels.
Carbon monoxide can also be produced by high-temperature electrolysis of carbon dioxide with solid oxide electrolyzer cells.[67] One method developed at DTU Energy uses a cerium oxide catalyst and does not have any issues of fouling of the catalyst.[68][69]
- 2 CO2 → 2 CO + O2
Carbon monoxide is also a byproduct of the reduction of metal oxide ores with carbon, shown in a simplified form as follows:
- MO + C → M + CO
Carbon monoxide is also produced by the direct oxidation of carbon in a limited supply of oxygen or air.
- 2 C + O2 → 2 CO
Since CO is a gas, the reduction process can be driven by heating, exploiting the positive (favorable) entropy of reaction. The Ellingham diagram shows that CO formation is favored over CO2 in high temperatures.
Use
[edit]Chemical industry
[edit]Carbon monoxide is an industrial gas that has many applications in bulk chemicals manufacturing.[70] Large quantities of aldehydes are produced by the hydroformylation reaction of alkenes, carbon monoxide, and H2. Hydroformylation is coupled to the Shell higher olefin process to give precursors to detergents.
Phosgene, useful for preparing isocyanates, polycarbonates, and polyurethanes, is produced by passing purified carbon monoxide and chlorine gas through a bed of porous activated carbon, which serves as a catalyst. World production of this compound was estimated to be 2.74 million tonnes in 1989.[71]
- CO + Cl2 → COCl2
Methanol is produced by the hydrogenation of carbon monoxide. In a related reaction, the hydrogenation of carbon monoxide is coupled to C−C bond formation, as in the Fischer–Tropsch process where carbon monoxide is hydrogenated to liquid hydrocarbon fuels. This technology allows coal or biomass to be converted to diesel.
In the Cativa process, carbon monoxide and methanol react in the presence of a homogeneous iridium catalyst and hydroiodic acid to give acetic acid. This process is responsible for most of the industrial production of acetic acid.
Metallurgy
[edit]Carbon monoxide is a strong reductive agent and has been used in pyrometallurgy to reduce metals from ores since ancient times. Carbon monoxide strips oxygen off metal oxides, reducing them to pure metal in high temperatures, forming carbon dioxide in the process. Carbon monoxide is not usually supplied as is, in the gaseous phase, in the reactor, but rather it is formed in high temperature in presence of oxygen-carrying ore, or a carboniferous agent such as coke, and high temperature. The blast furnace process is a typical example of a process of reduction of metal from ore with carbon monoxide.
Likewise, blast furnace gas collected at the top of blast furnace, still contains some 10% to 30% of carbon monoxide, and is used as fuel on Cowper stoves and on Siemens-Martin furnaces on open hearth steelmaking.
Proposed use as a rocket fuel
[edit]Carbon monoxide has been proposed for use as a fuel on Mars by NASA researcher Geoffrey Landis. Carbon monoxide/oxygen engines have been suggested for early surface transportation use as both carbon monoxide and oxygen can be straightforwardly produced from the carbon dioxide atmosphere of Mars by zirconia electrolysis, without using any Martian water resources to obtain hydrogen, which would be needed to make methane or any hydrogen-based fuel.[72]
Landis also proposed manufacturing the fuel from the similar carbon dioxide atmosphere of Venus for a sample return mission, in combination with solar-powered UAVs and rocket balloon ascent.[73]
Biological and physiological properties
[edit]Physiology
[edit]Carbon monoxide is a bioactive molecule which acts as a gaseous signaling molecule. It is naturally produced by many enzymatic and non-enzymatic pathways,[74] the best understood of which is the catabolic action of heme oxygenase on the heme derived from hemoproteins such as hemoglobin.[75] Following the first report that carbon monoxide is a normal neurotransmitter in 1993,[54] carbon monoxide has received significant clinical attention as a biological regulator.
Because of carbon monoxide's role in the body, abnormalities in its metabolism have been linked to a variety of diseases, including neurodegenerations, hypertension, heart failure, and pathological inflammation.[76] In many tissues, carbon monoxide acts as anti-inflammatory, vasodilatory, and encouragers of neovascular growth.[77] In animal model studies, carbon monoxide reduced the severity of experimentally induced bacterial sepsis, pancreatitis, hepatic ischemia/reperfusion injury, colitis, osteoarthritis, lung injury, lung transplantation rejection, and neuropathic pain while promoting skin wound healing. Therefore, there is significant interest in the therapeutic potential of carbon monoxide becoming pharmaceutical agent and clinical standard of care.[78]
Medicine
[edit]Studies involving carbon monoxide have been conducted in many laboratories throughout the world for its anti-inflammatory and cytoprotective properties.[79] These properties have the potential to be used to prevent the development of a series of pathological conditions including ischemia reperfusion injury, transplant rejection, atherosclerosis, severe sepsis, severe malaria, or autoimmunity.[78] Many pharmaceutical drug delivery initiatives have developed methods to safely administer carbon monoxide, and subsequent controlled clinical trials have evaluated the therapeutic effect of carbon monoxide.[80]
Microbiology
[edit]Microbiota may also utilize carbon monoxide as a gasotransmitter.[81] Carbon monoxide sensing is a signaling pathway facilitated by proteins such as CooA.[82][83][84] The scope of the biological roles for carbon monoxide sensing is still unknown.
The human microbiome produces, consumes, and responds to carbon monoxide.[74] For example, in certain bacteria, carbon monoxide is produced via the reduction of carbon dioxide by the enzyme carbon monoxide dehydrogenase with favorable bioenergetics to power downstream cellular operations.[85][74] In another example, carbon monoxide is a nutrient for methanogenic archaea which reduce it to methane using hydrogen.[86]
Carbon monoxide has certain antimicrobial properties which have been studied to treat against infectious diseases.[74]
Food science
[edit]Carbon monoxide is used in modified atmosphere packaging systems in the US, mainly with fresh meat products such as beef, pork, and fish to keep them looking fresh. The benefit is two-fold, carbon monoxide protects against microbial spoilage and it enhances the meat color for consumer appeal.[87] The carbon monoxide combines with myoglobin to form carboxymyoglobin, a bright-cherry-red pigment. Carboxymyoglobin is more stable than the oxygenated form of myoglobin, oxymyoglobin, which can become oxidized to the brown pigment metmyoglobin. This stable red color can persist much longer than in normally packaged meat. Typical levels of carbon monoxide used in the facilities that use this process are between 0.4% and 0.5%.[87]
The technology was first given "generally recognized as safe" (GRAS) status by the U.S. Food and Drug Administration (FDA) in 2002 for use as a secondary packaging system, and does not require labeling. In 2004, the FDA approved CO as primary packaging method, declaring that CO does not mask spoilage odor.[88] The process is currently unauthorized in many other countries, including Japan, Singapore, and the European Union.[89][90][91]
Weaponization
[edit]In ancient history, Hannibal executed Roman prisoners with coal fumes during the Second Punic War.[54]
Carbon monoxide had been used for genocide during the Holocaust at some extermination camps, the most notable by gas vans in Chełmno, and in the Action T4 "euthanasia" program.[92]
History
[edit]Prehistory
[edit]Humans have maintained a complex relationship with carbon monoxide since first learning to control fire circa 800,000 BC. Early humans probably discovered the toxicity of carbon monoxide poisoning upon introducing fire into their dwellings. The early development of metallurgy and smelting technologies emerging circa 6,000 BC through the Bronze Age likewise plagued humankind from carbon monoxide exposure. Apart from the toxicity of carbon monoxide, indigenous Native Americans may have experienced the neuroactive properties of carbon monoxide through shamanistic fireside rituals.[54]
Ancient history
[edit]Early civilizations developed mythological tales to explain the origin of fire, such as Prometheus from Greek mythology who shared fire with humans. Aristotle (384–322 BC) first recorded that burning coals produced toxic fumes. Greek physician Galen (129–199 AD) speculated that there was a change in the composition of the air that caused harm when inhaled, and many others of the era developed a basis of knowledge about carbon monoxide in the context of coal fume toxicity. Cleopatra may have died from carbon monoxide poisoning.[54]
Pre–industrial revolution
[edit]Georg Ernst Stahl mentioned carbonarii halitus in 1697 in reference to toxic vapors thought to be carbon monoxide. Friedrich Hoffmann conducted the first modern scientific investigation into carbon monoxide poisoning from coal in 1716. Herman Boerhaave conducted the first scientific experiments on the effect of carbon monoxide (coal fumes) on animals in the 1730s.[54]
Joseph Priestley is considered to have first synthesized carbon monoxide in 1772. Carl Wilhelm Scheele similarly isolated carbon monoxide from charcoal in 1773 and thought it could be the carbonic entity making fumes toxic. Torbern Bergman isolated carbon monoxide from oxalic acid in 1775. Later in 1776, the French chemist de Lassone produced CO by heating zinc oxide with coke, but mistakenly concluded that the gaseous product was hydrogen, as it burned with a blue flame. In the presence of oxygen, including atmospheric concentrations, carbon monoxide burns with a blue flame, producing carbon dioxide. Antoine Lavoisier conducted similar inconclusive experiments to Lassone in 1777. The gas was identified as a compound containing carbon and oxygen by William Cruickshank in 1800.[54][93]
Thomas Beddoes and James Watt recognized carbon monoxide (as hydrocarbonate) to brighten venous blood in 1793. Watt suggested coal fumes could act as an antidote to the oxygen in blood, and Beddoes and Watt likewise suggested hydrocarbonate has a greater affinity for animal fiber than oxygen in 1796. In 1854, Adrien Chenot similarly suggested carbon monoxide to remove the oxygen from blood and then be oxidized by the body to carbon dioxide.[54] The mechanism for carbon monoxide poisoning is widely credited to Claude Bernard whose memoirs beginning in 1846 and published in 1857 phrased, "prevents arterials blood from becoming venous". Felix Hoppe-Seyler independently published similar conclusions in the following year.[54]
Advent of industrial chemistry
[edit]Carbon monoxide gained recognition as an essential reagent in the 1900s.[5] Three industrial processes illustrate its evolution in industry. In the Fischer–Tropsch process, coal and related carbon-rich feedstocks are converted into liquid fuels via the intermediacy of CO. Originally developed as part of the German war effort to compensate for their lack of domestic petroleum, this technology continues today. Also in Germany, a mixture of CO and hydrogen was found to combine with olefins to give aldehydes. This process, called hydroformylation, is used to produce many large scale chemicals such as surfactants as well as specialty compounds that are popular fragrances and drugs. For example, CO is used in the production of vitamin A.[94] In a third major process, attributed to researchers at Monsanto, CO combines with methanol to give acetic acid. Most acetic acid is produced by the Cativa process. Hydroformylation and the acetic acid syntheses are two of myriad carbonylation processes.
See also
[edit]- Breath carbon monoxide – Level of carbon monoxide in exhaled breath
- Carbon monoxide (data page) – Chemical data page
- Carbon monoxide detector – Device that measures carbon monoxide (CO)
- Criteria air pollutants – US EPA limits on certain air pollutants
- List of highly toxic gases
References
[edit]- ^ a b NIOSH Pocket Guide to Chemical Hazards. "#0105". National Institute for Occupational Safety and Health (NIOSH).
- ^ a b "Carbon monoxide". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- ^ Richard, Pohanish (2012). Sittig's Handbook of Toxic and Hazardous Chemicals and Carcinogens (2 ed.). Elsevier. p. 572. ISBN 978-1-4377-7869-4. Retrieved 5 September 2015.
- ^ "Carbon Monoxide - CAMEO Chemicals". cameochemicals.noaa.gov. US NOAA Office of Response and Restoration.
- ^ a b Bierhals, Jürgen (2001). "Carbon Monoxide". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a05_203. ISBN 3527306730.
- ^ Bierhals, Jürgen (2001). "Carbon Monoxide". Ullmann's Encyclopedia of Industrial Chemistry. doi:10.1002/14356007.a05_203. ISBN 3527306730.
- ^ Voiland, Adam. "Fourteen years of carbon monoxide from MOPITT". Climate Change: Vital Signs of the Planet. Retrieved 2022-03-04.
- ^ a b c d e Myers, Isabella (February 2022). The efficient operation of regulation and legislation: An holistic approach to understanding the effect of Carbon Monoxide on mortality (PDF). CO Research Trust.
- ^ a b "Carbon monoxide: toxicological overview". UK Health Security Agency. 24 May 2022. Retrieved 2024-04-17.
- ^ a b Dent, Matthew R.; Rose, Jason J.; Tejero, Jesús; Gladwin, Mark T. (2024-01-29). "Carbon Monoxide Poisoning: From Microbes to Therapeutics". Annual Review of Medicine. 75 (1): 337–351. doi:10.1146/annurev-med-052422-020045. ISSN 0066-4219. PMC 11160397. PMID 37582490.
- ^ Gilliam, O. R.; Johnson, C. M.; Gordy, W. (1950). "Microwave Spectroscopy in the Region from Two to Three Millimeters". Physical Review. 78 (2): 140–144. Bibcode:1950PhRv...78..140G. doi:10.1103/PhysRev.78.140.
- ^ Haynes, William M. (2010). Handbook of Chemistry and Physics (91 ed.). Boca Raton, Florida, USA: CRC Press. p. 9–33. ISBN 978-1-43982077-3.
- ^ Haynes, William M. (2010). Handbook of Chemistry and Physics (91 ed.). Boca Raton, Florida, USA: CRC Press. p. 9–39. ISBN 978-1-43982077-3.
- ^ Common Bond Energies (D) and Bond Lengths (r). wiredchemist.com
- ^ Vidal, C. R. (28 June 1997). "Highly Excited Triplet States of Carbon Monoxide". Archived from the original on 2006-08-28. Retrieved August 16, 2012.
- ^ Holman, Jack P. (2002). Heat transfer (9th ed.). New York, NY: McGraw-Hill Companies, Inc. pp. 600–606. ISBN 9780072406559. OCLC 46959719.
- ^ Incropera 1 Dewitt 2 Bergman 3 Lavigne 4, Frank P. 1 David P. 2 Theodore L. 3 Adrienne S. 4 (2007). Fundamentals of heat and mass transfer (6th ed.). Hoboken, NJ: John Wiley and Sons, Inc. pp. 941–950. ISBN 9780471457282. OCLC 62532755.
{{cite book}}: CS1 maint: numeric names: authors list (link) - ^ Little, L. H.; Amberg, C. H. (1962). "Infrared Spectra of Carbon Monoxide and Carbon Dioxide Adsorbed on Chromia–Alumina and on Alumina". Canadian Journal of Chemistry. 40 (10): 1997–2006. doi:10.1139/v62-306.
- ^ Scuseria, Gustavo E.; Miller, Michael D.; Jensen, Frank; Geertsen, Jan (1991). "The dipole moment of carbon monoxide". J. Chem. Phys. 94 (10): 6660. Bibcode:1991JChPh..94.6660S. doi:10.1063/1.460293.
- ^ Martinie, Ryan J.; Bultema, Jarred J.; Vander Wal, Mark N.; Burkhart, Brandon J.; Vander Griend, Douglas A.; DeKock, Roger L. (2011-08-01). "Bond Order and Chemical Properties of BF, CO, and N2". Journal of Chemical Education. 88 (8): 1094–1097. Bibcode:2011JChEd..88.1094M. doi:10.1021/ed100758t. ISSN 0021-9584. S2CID 11905354.
- ^ Ulrich, Henri (2009). Cumulenes in click reactions. Wiley InterScience (Online service). Chichester, U.K.: Wiley. p. 45. ISBN 9780470747957. OCLC 476311784.
- ^ Lupinetti, Anthony J.; Fau, Stefan; Frenking, Gernot; Strauss, Steven H. (1997). "Theoretical Analysis of the Bonding between CO and Positively Charged Atoms". J. Phys. Chem. A. 101 (49): 9551–9559. Bibcode:1997JPCA..101.9551L. doi:10.1021/jp972657l.
- ^ Blanco, Fernando; Alkorta, Ibon; Solimannejad, Mohammad; Elguero, Jose (2009). "Theoretical Study of the 1:1 Complexes between Carbon Monoxide and Hypohalous Acids". J. Phys. Chem. A. 113 (13): 3237–3244. Bibcode:2009JPCA..113.3237B. doi:10.1021/jp810462h. hdl:10261/66300. PMID 19275137.
- ^ Meerts, W; De Leeuw, F.H.; Dymanus, A. (1 June 1977). "Electric and magnetic properties of carbon monoxide by molecular-beam electric-resonance spectroscopy". Chemical Physics. 22 (2): 319–324. Bibcode:1977CP.....22..319M. doi:10.1016/0301-0104(77)87016-X.
- ^ Stefan, Thorsten; Janoschek, Rudolf (2000). "How relevant are S=O and P=O Double Bonds for the Description of the Acid Molecules H2SO3, H2SO4, and H3PO3, respectively?". Journal of Molecular Modeling. 6 (2): 282–288. doi:10.1007/PL00010730. S2CID 96291857.
- ^ Global Maps. Carbon Monoxide. earthobservatory.nasa.gov
- ^ Khalil, M.A.K; Pinto, J.P; Shearer, M.J (1999). "Atmospheric carbon monoxide". Chemosphere - Global Change Science. 1 (1–3): ix–xi. Bibcode:1999ChGCS...1D...9K. doi:10.1016/S1465-9972(99)00053-7.
- ^ Source for figures: Carbon dioxide, NOAA Earth System Research Laboratory, (updated 2010.06). Methane, IPCC TAR table 6.1 Archived 2007-06-15 at the Wayback Machine, (updated to 1998). The NASA total was 17 ppmv over 100%, and CO2 was increased here by 15 ppmv. To normalize, N2 should be reduced by about 25 ppmv and O2 by about 7 ppmv.
- ^ Committee on Medical and Biological Effects of Environmental Pollutants (1977). Carbon Monoxide. Washington, D.C.: National Academy of Sciences. p. 29. ISBN 978-0-309-02631-4.
- ^ Green W. "An Introduction to Indoor Air Quality: Carbon Monoxide (CO)". United States Environmental Protection Agency. Retrieved 2008-12-16.
- ^ Gosink, Tom (1983-01-28). "What Do Carbon Monoxide Levels Mean?". Alaska Science Forum. Geophysical Institute, University of Alaska Fairbanks. Archived from the original on 2008-12-25. Retrieved 2007-12-01.
- ^ Singer, Siegfried Fred (1975). The Changing Global Environment. Springer. p. 90. ISBN 978-9027704023.
- ^ a b "Carbon Monoxide Poisoning: Vehicles (AEN-208)". abe.iastate.edu. Retrieved 11 February 2018.
- ^ Gosink T (January 28, 1983). "What Do Carbon Monoxide Levels Mean?". Alaska Science Forum. Geophysical Institute, University of Alaska Fairbanks. Archived from the original on December 25, 2008. Retrieved December 16, 2008.
- ^ Weinstock, B.; Niki, H. (1972). "Carbon Monoxide Balance in Nature". Science. 176 (4032): 290–2. Bibcode:1972Sci...176..290W. doi:10.1126/science.176.4032.290. PMID 5019781. S2CID 25223868.
- ^ Seinfeld, John; Pandis, Spyros (2006). Atmospheric Chemistry and Physics: From Air Pollution to Climate Change. John Wiley & Sons. ISBN 978-0-471-72018-8.
- ^ Sigel, Astrid; Sigel, Roland K. O. (2009). Metal-Carbon Bonds in Enzymes and Cofactors. Royal Society of Chemistry. p. 243. ISBN 978-1-84755-915-9.
- ^ White, James Carrick; et al. (1989). Global climate change linkages: acid rain, air quality, and stratospheric ozone. Springer. p. 106. ISBN 978-0-444-01515-0.
- ^ Drummond, James (February 2, 2018). "MOPITT, Atmospheric Pollution, and Me: A Personal Story". Canadian Meteorological and Oceanographic Society. Retrieved August 1, 2018.
- ^ Pommier, M.; Law, K. S.; Clerbaux, C.; Turquety, S.; Hurtmans, D.; Hadji-Lazaro, J.; Coheur, P.-F.; Schlager, H.; Ancellet, G.; Paris, J.-D.; Nédélec, P.; Diskin, G. S.; Podolske, J. R.; Holloway, J. S.; Bernath, P. (2010). "IASI carbon monoxide validation over the Arctic during POLARCAT spring and summer campaigns". Atmospheric Chemistry and Physics. 10 (21): 10655–10678. Bibcode:2010ACP....1010655P. doi:10.5194/acp-10-10655-2010.
- ^ Combes, Françoise (1991). "Distribution of CO in the Milky Way". Annual Review of Astronomy & Astrophysics. 29: 195–237. Bibcode:1991ARA&A..29..195C. doi:10.1146/annurev.aa.29.090191.001211.
- ^ Hamed, M. (2021). "Multiwavelength dissection of a massive heavily dust-obscured galaxy and its blue companion at z~2". Astronomy & Astrophysics. 646: A127. arXiv:2101.07724. Bibcode:2021A&A...646A.127H. doi:10.1051/0004-6361/202039577. S2CID 231639096.
- ^ Khan, Amina. "Did two planets around nearby star collide? Toxic gas holds hints". Los Angeles Times. Retrieved March 9, 2014.
- ^ Dent, WR, Wyatt, MC, Roberge, A, Augereau, JC, Casassus, S, Corder, S, Greaves, JS, de Gregorio-Monsalvo, I, Hales, A, Jackson, AP, Hughes, AM, Meredith A, Lagrange, AM, Matthews, B, Wilner, D (March 6, 2014). "Molecular Gas Clumps from the Destruction of Icy Bodies in the β Pictoris Debris Disk". Science. 343 (6178): 1490–1492. arXiv:1404.1380. Bibcode:2014Sci...343.1490D. doi:10.1126/science.1248726. PMID 24603151. S2CID 206553853. Retrieved March 9, 2014.
- ^ Lellouch, E.; de Bergh, C.; Sicardy, B.; Ferron, S.; Käufl, H.-U. (2010). "Detection of CO in Triton's atmosphere and the nature of surface-atmosphere interactions". Astronomy and Astrophysics. 512: L8. arXiv:1003.2866. Bibcode:2010A&A...512L...8L. doi:10.1051/0004-6361/201014339. ISSN 0004-6361. S2CID 58889896.
- ^ Greenberg, J. Mayo (1998). "Making a comet nucleus". Astronomy and Astrophysics. 330: 375. Bibcode:1998A&A...330..375G.
- ^ Yeomans, Donald K. (2005). "Comets (World Book Online Reference Center 125580)". NASA. Archived from the original on 29 April 2005. Retrieved 18 August 2022.
About 80 percent of the ice is water ice, and frozen carbon monoxide makes up another 15 percent.
- ^ Christopher Glein and Hunter Waite (May 11, 2018). "Primordial N2 provides a cosmo chemical explanation for the existence of Sputnik Planitia, Pluto". Icarus. 313: 79–92. arXiv:1805.09285. Bibcode:2018Icar..313...79G. doi:10.1016/j.icarus.2018.05.007. S2CID 102343522.
- ^ Pommier, M.; McLinden, C. A.; Deeter, M. (2013). "Relative changes in CO emissions over megacities based on observations from space". Geophysical Research Letters. 40 (14): 3766. Bibcode:2013GeoRL..40.3766P. doi:10.1002/grl.50704.
- ^ Reeves, Claire E.; Penkett, Stuart A.; Bauguitte, Stephane; Law, Kathy S.; Evans, Mathew J.; Bandy, Brian J.; Monks, Paul S.; Edwards, Gavin D.; Phillips, Gavin; Barjat, Hannah; Kent, Joss; Dewey, Ken; Schmitgen, Sandra; Kley, Dieter (2002). "Potential for photochemical ozone formation in the troposphere over the North Atlantic as derived from aircraft observationsduring ACSOE". Journal of Geophysical Research. 107 (D23): 4707. Bibcode:2002JGRD..107.4707R. doi:10.1029/2002JD002415.
- ^ Ozone and other photochemical oxidants. National Academies. 1977. p. 23. ISBN 978-0-309-02531-7.
- ^ a b Penney, David; Benignus, Vernon; Kephalopoulos, Stylianos; Kotzias, Dimitrios; Kleinman, Michael; Verrier, Agnes (2010), "Carbon monoxide", WHO Guidelines for Indoor Air Quality: Selected Pollutants, World Health Organization, ISBN 978-92-890-0213-4, OCLC 696099951, archived from the original on March 8, 2021, retrieved March 18, 2024
- ^ "MSHA - Occupational Illness and Injury Prevention Program - Health Topics - Carbon Monoxide". arlweb.msha.gov. Archived from the original on 2017-12-31. Retrieved 2017-12-31.
- ^ a b c d e f g h i Hopper, Christopher P.; Zambrana, Paige N.; Goebel, Ulrich; Wollborn, Jakob (2021-06-01). "A brief history of carbon monoxide and its therapeutic origins". Nitric Oxide. 111–112: 45–63. doi:10.1016/j.niox.2021.04.001. ISSN 1089-8603. PMID 33838343. S2CID 233205099.
- ^ Peng, Jin-Bao; Geng, Hui-Qing; Wu, Xiao-Feng (2019-03-14). "The Chemistry of CO: Carbonylation". Chem. 5 (3): 526–552. Bibcode:2019Chem....5..526P. doi:10.1016/j.chempr.2018.11.006. ISSN 2451-9294.
- ^ a b Koch, H.; Haaf, W. (1964). "1-Adamantanecarboxylic Acid". Organic Syntheses. 44: 1. doi:10.15227/orgsyn.044.0001.
- ^ a b Coleman, G. H.; Craig, David (1932). "p-Tolualdehyde". Organic Syntheses. 12: 80. doi:10.15227/orgsyn.012.0080.
- ^ Chatani, N.; Murai, S. "Carbon Monoxide" in Encyclopedia of Reagents for Organic Synthesis (Ed: L. Paquette) 2004, J. Wiley & Sons, New York. doi:10.1002/047084289X
- ^ Büchner, W.; Weiss, E. (1964). "Zur Kenntnis der sogenannten "Alkalicarbonyle" IV[1] Über die Reaktion von geschmolzenem Kalium mit Kohlenmonoxid". Helvetica Chimica Acta. 47 (6): 1415–1423. doi:10.1002/hlca.19640470604.
- ^ Fownes, George (1869). A Manual of elementary chemistry. H.C. Lea. p. 678.
- ^ Katz, Allen I.; Schiferl, David; Mills, Robert L. (1984). "New phases and chemical reactions in solid carbon monoxide under pressure". The Journal of Physical Chemistry. 88 (15): 3176–3179. doi:10.1021/j150659a007.
- ^ Evans, W. J.; Lipp, M. J.; Yoo, C.-S.; Cynn, H.; Herberg, J. L.; Maxwell, R. S.; Nicol, M. F. (2006). "Pressure-Induced Polymerization of Carbon Monoxide: Disproportionation and Synthesis of an Energetic Lactonic Polymer". Chemistry of Materials. 18 (10): 2520–2531. doi:10.1021/cm0524446.
- ^ Brauer, Georg (1963). Handbook of Preparative Inorganic Chemistry Vol. 1, 2nd Ed. New York: Academic Press. p. 646. ISBN 978-0121266011.
- ^ Holleman, A. F.; Wiberg, E. "Inorganic Chemistry" Academic Press: San Diego, 200. ISBN 0-12-352651-5.
- ^ "Carbon Monoxide". Retrieved 21 May 2021.
- ^ Higman, C; van der Burgt, M (2003). Gasification. Gulf Professional Publishing. p. 12. ISBN 978-0-7506-7707-3.
- ^ Zheng, Yun; Wang, Jianchen; Yu, Bo; Zhang, Wenqiang; Chen, Jing; Qiao, Jinli; Zhang, Jiujun (2017). "A review of high temperature co-electrolysis of H O and CO to produce sustainable fuels using solid oxide electrolysis cells (SOECs): advanced materials and technology". Chem. Soc. Rev. 46 (5): 1427–1463. doi:10.1039/C6CS00403B. PMID 28165079.
- ^ "New route to carbon-neutral fuels from carbon dioxide discovered by Stanford-DTU team - DTU". dtu.dk.
- ^ Skafte, Theis L.; Guan, Zixuan; Machala, Michael L.; Gopal, Chirranjeevi B.; Monti, Matteo; Martinez, Lev; Stamate, Eugen; Sanna, Simone; Garrido Torres, Jose A.; Crumlin, Ethan J.; García-Melchor, Max; Bajdich, Michal; Chueh, William C.; Graves, Christopher (October 8, 2019). "Selective high-temperature CO 2 electrolysis enabled by oxidized carbon intermediates". Nature Energy. 4 (10): 846–855. Bibcode:2019NatEn...4..846S. doi:10.1038/s41560-019-0457-4. hdl:2262/93685. S2CID 202640892 – via www.nature.com.
- ^ Elschenbroich, C.; Salzer, A. (2006). Organometallics: A Concise Introduction (2nd ed.). Weinheim: Wiley-VCH. ISBN 978-3-527-28165-7.
- ^ Wolfgang Schneider; Werner Diller. "Phosgene". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a19_411. ISBN 978-3527306732.
- ^ Landis (2001). "Mars Rocket Vehicle Using In Situ Propellants". Journal of Spacecraft and Rockets. 38 (5): 730–735. Bibcode:2001JSpRo..38..730L. doi:10.2514/2.3739.
- ^ "Sample Return from the Surface of Venus". NASA. 2024-01-04. Retrieved 2024-05-29.
- ^ a b c d Hopper, Christopher P.; De La Cruz, Ladie Kimberly; Lyles, Kristin V.; Wareham, Lauren K.; Gilbert, Jack A.; Eichenbaum, Zehava; Magierowski, Marcin; Poole, Robert K.; Wollborn, Jakob; Wang, Binghe (2020-12-23). "Role of Carbon Monoxide in Host–Gut Microbiome Communication". Chemical Reviews. 120 (24): 13273–13311. doi:10.1021/acs.chemrev.0c00586. ISSN 0009-2665. PMID 33089988. S2CID 224824871.
- ^ Ryter, Stefan W.; Alam, Jawed; Choi, Augustine M. K. (2006-04-01). "Heme Oxygenase-1/Carbon Monoxide: From Basic Science to Therapeutic Applications". Physiological Reviews. 86 (2): 583–650. doi:10.1152/physrev.00011.2005. ISSN 0031-9333. PMID 16601269.
- ^ Wu, L; Wang, R (December 2005). "Carbon Monoxide: Endogenous Production, Physiological Functions, and Pharmacological Applications". Pharmacol Rev. 57 (4): 585–630. doi:10.1124/pr.57.4.3. PMID 16382109. S2CID 17538129.
- ^ Campbell, Nicole K.; Fitzgerald, Hannah K.; Dunne, Aisling (2021-01-29). "Regulation of inflammation by the antioxidant haem oxygenase 1". Nature Reviews Immunology. 21 (7): 411–425. doi:10.1038/s41577-020-00491-x. ISSN 1474-1741. PMID 33514947. S2CID 231762031.
- ^ a b Motterlini, Roberto; Otterbein, Leo E. (2010). "The therapeutic potential of carbon monoxide". Nature Reviews Drug Discovery. 9 (9): 728–743. doi:10.1038/nrd3228. ISSN 1474-1784. PMID 20811383. S2CID 205477130.
- ^ Motterlini, Roberto; Foresti, Roberta (2017-01-11). "Biological signaling by carbon monoxide and carbon monoxide-releasing molecules". American Journal of Physiology. Cell Physiology. 312 (3): C302–C313. doi:10.1152/ajpcell.00360.2016. ISSN 0363-6143. PMID 28077358.
- ^ Hopper, Christopher P.; Meinel, Lorenz; Steiger, Christoph; Otterbein, Leo E. (2018-10-11). "Where is the Clinical Breakthrough of Heme Oxygenase-1 / Carbon Monoxide Therapeutics?". Current Pharmaceutical Design. 24 (20): 2264–2282. doi:10.2174/1381612824666180723161811. PMID 30039755. S2CID 51712930.
- ^ Wareham, Lauren K.; Southam, Hannah M.; Poole, Robert K. (2018-09-06). "Do nitric oxide, carbon monoxide and hydrogen sulfide really qualify as 'gasotransmitters' in bacteria?". Biochemical Society Transactions. 46 (5): 1107–1118. doi:10.1042/BST20170311. ISSN 0300-5127. PMC 6195638. PMID 30190328.
- ^ Roberts, G. P.; Youn, H.; Kerby, R. L. (2004). "CO-Sensing Mechanisms". Microbiology and Molecular Biology Reviews. 68 (3): 453–473. doi:10.1128/MMBR.68.3.453-473.2004. PMC 515253. PMID 15353565.
- ^ Shimizu, Toru; Lengalova, Alzbeta; Martínek, Václav; Martínková, Markéta (2019-12-09). "Heme: emergent roles of heme in signal transduction, functional regulation and as catalytic centres". Chemical Society Reviews. 48 (24): 5624–5657. doi:10.1039/C9CS00268E. ISSN 1460-4744. PMID 31748766. S2CID 208217502.
- ^ Shimizu, Toru; Huang, Dongyang; Yan, Fang; Stranava, Martin; Bartosova, Martina; Fojtíková, Veronika; Martínková, Markéta (2015-07-08). "Gaseous O2, NO, and CO in Signal Transduction: Structure and Function Relationships of Heme-Based Gas Sensors and Heme-Redox Sensors". Chemical Reviews. 115 (13): 6491–6533. doi:10.1021/acs.chemrev.5b00018. ISSN 0009-2665. PMID 26021768.
- ^ Jaouen, G., ed. (2006). Bioorganometallics: Biomolecules, Labeling, Medicine. Weinheim: Wiley-VCH. ISBN 978-3-527-30990-0.
- ^ Thauer, R. K. (1998). "Biochemistry of methanogenesis: a tribute to Marjory Stephenson. 1998 Marjory Stephenson Prize Lecture" (Free). Microbiology. 144 (9): 2377–2406. doi:10.1099/00221287-144-9-2377. PMID 9782487.
- ^ a b Van Rooyen, Lauren Anne; Allen, Paul; O'Connor, David I. (October 2017). "The application of carbon monoxide in meat packaging needs to be re-evaluated within the EU: An overview". Meat Science. 132: 179–188. doi:10.1016/j.meatsci.2017.03.016. PMID 28465017.
- ^ Eilert EJ (2005). "New packaging technologies for the 21st century". Journal of Meat Science. 71 (1): 122–127. doi:10.1016/j.meatsci.2005.04.003. PMID 22064057.
- ^ "Proof in the Pink? Meat Treated to Give It Fresh Look". ABC News. November 14, 2007. Retrieved May 5, 2009.
- ^ Carbon Monoxide in Meat Packaging: Myths and Facts. American Meat Institute. 2008. Archived from the original on 2011-07-14. Retrieved May 5, 2009.
- ^ "CO in packaged meat". Carbon Monoxide Kills Campaign. Archived from the original on September 26, 2010. Retrieved May 5, 2009.
- ^ Kitchen, Martin (2006). A history of modern Germany, 1800–2000. Wiley-Blackwell. p. 323. ISBN 978-1-4051-0041-0.
- ^ Stromeyer, Friedrich (1808). Grundriß der theoretischen Chemie: zum Behuf seiner Vorlesungen entworfen (in German). Röwer. pp. 1–18.
- ^ Kelkar, A.A. (2016). "Carbonylations and Hydroformylations for Fine Chemicals". Industrial Catalytic Processes for Fine and Specialty Chemicals. pp. 663–692. doi:10.1016/B978-0-12-801457-8.00014-8. ISBN 9780128014578.
External links
[edit]- Global map of carbon monoxide distribution
- Explanation of the structure
- International Chemical Safety Card 0023
- CDC NIOSH Pocket Guide to Chemical Hazards: Carbon monoxide—National Institute for Occupational Safety and Health (NIOSH), US Centers for Disease Control and Prevention (CDC)
- External MSDS data sheet
- Carbon Monoxide Detector Placement
- Microscale Gas Chemistry Experiments with Carbon Monoxide
- "Instant insight: Don't blame the messenger". Chemical Biology (11: Research News). 18 October 2007. Archived from the original on 28 October 2007. Retrieved 27 October 2019.
Outlining the physiology of carbon monoxide from the Royal Society of Chemistry.




