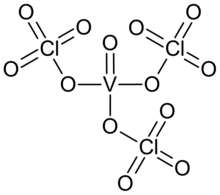
Vanadyl perchlorate
| |
| Names | |
|---|---|
| Other names
tripercloratooxovanadium
| |
| Identifiers | |
3D model (JSmol)
|
|
| |
| |
| Properties | |
| VO(ClO4)3 | |
| Molar mass | 356.29 g/mol |
| Appearance | golden yellow liquid or crystals. |
| Melting point | 21-22 °C |
| Boiling point | 33.5 °C in vacuum |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
oxidant |
| Related compounds | |
Related compounds
|
niobium perchlorate, vanadyl nitrate, chromyl perchlorate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Vanadyl perchlorate or vanadyl triperchlorate is a golden yellow coloured liquid or crystalline compound of vanadium, oxygen and perchlorate group. The substance consists of molecules covalently bound and is quite volatile; it ignites organic solvents on contact and explodes at temperatures above 80 °C.[1]
Formation
[edit]Vanadyl perchlorate can be made by reacting vanadium pentoxide with dichlorine heptoxide at 5 °C. It is purified by distillation under a vacuum and recrystallisation at 21 °C.[1]
A solution of vanadium(V) perchlorate can be made by dissolving vanadium pentoxide in perchloric acid.[2]
The reaction of vanadium pentoxide and dichlorine hexoxide could produce VO(ClO4)3:[3]
- 2 V2O5 + 12 Cl2O6 → 4 VO(ClO4)3 + 12 ClO2 + 3 O2
Properties
[edit]It can react with vanadium oxychloride to form another vanadyl perchlorate (VO2ClO4): [3]
- 4 VO(ClO4)3 + 2 VOCl3 → 6 VO2ClO4 + 6 ClO2 + 3 Cl2 + 3 O2
Related
[edit]Other perchlorates include pervanadyl perchlorate, also known as dioxovanadium perchlorate, which contains VO2+ ions,[4] vanadyl diperchlorate, oxovanadium perchlorate or vanadium(IV) perchlorate, and VO(ClO4)2, which dissolves in water.[5][6] Vanadic perchlorate, also known as vanadium(III) perchlorate solution in water, is a green-tinged blue colour, significantly different to most other V(III) solutions, which are complexed.[7]
References
[edit]- ^ a b Fedoroff, Basil T; Sheffield, Oliver E. Encyclopedia of Explosives and Related Items Vol 10 of 10- U to Z, Page V5 (PDF). Vol. 10. p. 136.
- ^ Kustin, Kenneth; Toppen, David L. (June 1973). "Reduction of vanadium(V) by L-ascorbic acid". Inorganic Chemistry. 12 (6): 1404–1407. doi:10.1021/ic50124a038.
- ^ a b Schmeisser, Martin (1955). "Die Chemie der anorganischen Acylnitrate (ein Problem des Nitrylchlorids) und Acylperchlorate (ein Problem des Dichlorhexoxyds)". Angewandte Chemie. 67 (17–18): 493–501. Bibcode:1955AngCh..67..493S. doi:10.1002/ange.19550671708. ISSN 0044-8249.
- ^ Ramsey, J. B.; Heldman, M. J. (July 1936). "Kinetics of the Trivalent Vanadium—Iodine Reaction". Journal of the American Chemical Society. 58 (7): 1153–1157. doi:10.1021/ja01298a026.
- ^ Iannuzzi, Melanie M.; Rieger, Philip H. (December 1975). "Nature of vanadium(IV) in basic aqueous solution". Inorganic Chemistry. 14 (12): 2895–2899. doi:10.1021/ic50154a006.
- ^ Wuethrich, K.; Connick, Robert E. (March 1967). "Nuclear magnetic resonance relaxation of oxygen-17 in aqueous solutions of vanadyl perchlorate and the rate of elimination of water molecules from the first coordination sphere". Inorganic Chemistry. 6 (3): 583–590. doi:10.1021/ic50049a035.
- ^ Furman, Sydney C.; Garner, Clifford S. (April 1950). "Absorption Spectra of Vanadium(III) and Vanadium(IV) Ions in Complexing and Non-complexing Media". Journal of the American Chemical Society. 72 (4): 1785–1789. doi:10.1021/ja01160a105.
