Zucapsaicin
 | |
| Clinical data | |
|---|---|
| Trade names | Civanex |
| Other names | Civamide; (Z)-Capsaicin; cis-Capsaicin |
| Routes of administration | Topical |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.164.527 |
| Chemical and physical data | |
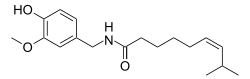
| Formula | C18H27NO3 |
| Molar mass | 305.418 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Zucapsaicin (Civanex) is a medication used to treat osteoarthritis of the knee and other neuropathic pain. Zucapsaicin is a member of phenols and a member of methoxybenzenes.[2] It is a modulator of transient receptor potential cation channel subfamily V member 1 (TRPV-1), also known as the vanilloid or capsaicin receptor 1 that reduces pain, and improves articular functions.[3][unreliable medical source?][4] It is the cis-isomer of capsaicin. Civamide, manufactured by Winston Pharmaceuticals, is produced in formulations for oral, nasal, and topical use (patch and cream).[5][6]
Zucapsaicin has been tested for treatment of a variety of conditions associated with ongoing nerve pain. This includes herpes simplex infections; cluster headaches and migraine; and knee osteoarthritis.[7][dead link] It was approved by the Health Canada in 2010 as topical cream sold under the brand name Zuacta.[3] It has a melting point of 71.5–74.5 °C.[3]
Pharmacology
[edit]Zucapsaicin mediates an antinociceptive action via acting as an agonist at TRPV1. TRPV1 play an important physiological role of transducing chemical, mechanical and thermal stimuli as well as pain transduction, and participate in pain modulation and perception. They are mainly distributed in C sensory nerve fibers as well as Aẟ fibers to transmit sensory information involving inflammatory and neuropathic pain, and activation of these channels releases somatostatin, calcitonin gene-related peptide (CGRP) and other neuropeptides (neurokinin A, kassinin), leading to neurogenic inflammation [A19720]. Zucapsaicin is also reported to affect the peptidergic afferent neurons via a desensitization mechanism to decrease the levels of dorsal root ganglia and sciatic calcitonin gene-related peptide (CGRP) and substance P (SP) [L877].[3]
Pharmacodynamics
[edit]Zucapsaicin mediates an antinociceptive action via acting as an agonist at TRPV1. TRPV1 play an important physiological role of transducing chemical, mechanical and thermal stimuli as well as pain transduction, and participate in pain modulation and perception. They are mainly distributed in C sensory nerve fibers as well as Aẟ fibers to transmit sensory information involving inflammatory and neuropathic pain, and activation of these channels releases somatostatin, calcitonin gene-related peptide (CGRP) and other neuropeptides (neurokinin A, kassinin), leading to neurogenic inflammation.[6] Zucapsaicin is also reported to affect the peptidergic afferent neurons via a desensitization mechanism to decrease the levels of dorsal root ganglia and sciatic calcitonin gene-related peptide (CGRP) and substance P (SP).[3]
Mechanism of action
[edit]Zucapsaicin excites and desensitizes C-fibers via agonist at TRPV1 on nociceptive neurons. It binds to intracellular sites and initially stimulates the channels, causing burning sensation.[4] The mechanism of pharmacological action of zucapsaicin has not been fully understood yet. It is suggested that this compound, similarly to its trans isomer, is an agonist of the vanilloid receptor VR1 (TRPV1) and a neuronal calcium channel blocker.[8][9] Capsaicin is able to excite and desensitize C-fibers. As such, it is not only able to cause pain, but also exhibit analgesic properties. Initially, it stimulates TRPV1, which is responsible for a burning sensation. This effect is followed by a longlasting refractory state – ‘desensitization’ – during which the previously excited sensory neurons become unresponsive to capsaicin and other stimuli. It was shown that desensitization and tachyphylaxis of TRPV1 channels contribute to capsaicin-induced pain relief.[10] Desensitization of TRPV1 represents the main mechanism of its inhibitory function.
Three distinct pathways of capsaicin-induced desensitization have been described: i) activation of calcineurin, which results in dephosphorylation of TRPV1; ii) activation of phospholipase C with the subsequent phosphatidylinositol 4,5-biphosphate hydrolysis (rather controversial) and iii) activation of calcium-dependent protein kinase C isoforms and subsequent channel phosphorylation.[11][12] Desensitization involves both tachyphylaxis (short-term desensitization) and long-term, persistent, desensitization.[13][14][15] It is suggested that the downregulation of proalgesic substances (such as SP) and upregulation of analgesic peptides are implicated in desensitization.[16] The exhaustion of SP reserves renders neurons desensitized and refractory. These mechanisms of desensitization are not fully understood. It is thought that the short-term desensitization is related to capsaicin's ability to block the intra-axonal transport of NGF, SP and somatostatin.[17]
The desensitization is a reversible phenomenon. It begins a few hours after capsaicin application and may last even several weeks.[16] The reversible desensitization was found useful in the treatment of pain, whereas the site-specific ablation of sensory nerves transmitting pain stimuli is a promising approach (‘molecular scalpel’) to achieve a permanent pain relief in patients suffering from bone cancer pain or HIV-induced neuropathies.[13][14] Desensitization and depletion of pronociceptive neurotransmitters induce chemical denervation with a loss of function, which is clinically used in osteoarthritis, diabetic neuropathy, psoriasis and others.[18][19][20] In dorsal root ganglia and the sciatic nerve, zucapsaicin decreases levels of SP and CGRP, indicating that it influences peptidergic afferent neurons via a desensitization mechanism[21][41]. When administered topically, the intended targets for zucapsaicin are the neurons that innervate the local area of application. These neurons transmit pain toward the CNS.
Pharmacokinetics
[edit]Absorption
[edit]Zucapsaicin displays low systemic absorption and localizes at the area of application. In animal studies, systemic absorption is 0.075%.[22][unreliable medical source?][23][24]
Metabolism
[edit]In vitro studies demonstrates weak to moderate inhibitory effects on various cytochrome P450 enzymes, although not clinically significant due to low systemic absorption.[23]
Route of elimination
[edit]In rat studies, zucapsaicin and its metabolites are slowly excreted into urine and feces (up to 2/3), with minimal elimination via exhalation following dermal administration.[22][23]
Half life
[edit]In rats, the elimination half life of zucapsaicin and its metabolites is approximately 7 to 11 hours.[22][23]
Toxicity
[edit]Most common adverse effects involved application site reactions such as transient burning and warm sensation. Other adverse effects observed in clinical trials are eye irritation, arthralgia, aggravated osteoarthritis, burning sensation, headache, cough and sneezing. Oral LD50 in mouse is >87.5 mg/kg in male and <60 mg/kg in females. Oral LD50 in rats is >90 mg/kg in males and >60 mg/kg in females.[22]
Chemical and Physical Properties
[edit]Computed properties
[edit]| Property Name | Property Value[25] |
|---|---|
| Molecular Weight | 305.418 g/mol |
| XLogP3-AA | 3.6 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 9 |
| Exact Mass | 305.199 g/mol |
| Monoisotopic Mass | 305.199 g/mol |
| Topological Polar Surface Area | 58.6 A^2 |
| Heavy Atom Count | 22 |
| Formal Charge | 0 |
| Complexity | 341 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 1 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently-Bonded Unit Count | 1 |
| Compound Is Canonicalized | Yes |
References
[edit]- ^ "Zuacta PI". Health Canada. 17 July 2020. Retrieved 15 October 2024.
- ^ "zucapsaicin (CHEBI:135952)". Chemical Entities of Biological Interest (ChEBI). EMBL-EBI. Retrieved 25 June 2019.
- ^ a b c d e "Zucapsaicin". DrugBank. Retrieved 25 June 2019.
- ^ a b Studer M, McNaughton PA (October 2010). "Modulation of single-channel properties of TRPV1 by phosphorylation". The Journal of Physiology. 588 (Pt 19): 3743–56. doi:10.1113/jphysiol.2010.190611. PMC 2998224. PMID 20693293.
- ^ "Civamide". Winston Pharmaceuticals. Archived from the original on 25 April 2012. Retrieved 16 November 2011.
- ^ a b Sałat K, Jakubowska A, Kulig K (October 2014). "Zucapsaicin for the treatment of neuropathic pain". Expert Opinion on Investigational Drugs. 23 (10): 1433–40. doi:10.1517/13543784.2014.956079. PMID 25171227. S2CID 2969900.
- ^ Zucapsaicin information from the National Library of Medicine http://druginfo.nlm.nih.gov/drugportal
- ^ Bevan SJ, Docherty RJ (1993). "Cellular mechanisms of the action of capsaicin.". In Wood J (ed.). Capsaicin in the study of pain. London, England: Academic Press. pp. 27–44.
- ^ Anand P, Bley K (October 2011). "Topical capsaicin for pain management: therapeutic potential and mechanisms of action of the new high-concentration capsaicin 8% patch". British Journal of Anaesthesia. 107 (4): 490–502. doi:10.1093/bja/aer260. PMC 3169333. PMID 21852280.
- ^ St Pierre M, Reeh PW, Zimmermann K (June 2009). "Differential effects of TRPV channel block on polymodal activation of rat cutaneous nociceptors in vitro". Experimental Brain Research. 196 (1): 31–44. doi:10.1007/s00221-009-1808-3. PMID 19404626. S2CID 25823889.
- ^ Stucky CL, Dubin AE, Jeske NA, et al. Roles of transient receptor potential channels in pain. Brain Res Rev 2009;60(1):2-23
- ^ Nilius B, Owsianik G (2013). "Transient Receptor Potential Family of Ion Channels". Encyclopedia of Pain. Vol. 12. Springer Berlin Heidelberg. p. 4037. doi:10.1007/978-3-642-28753-4_202324. ISBN 978-3-642-28752-7. PMC 3129667. PMID 21401968.
{{cite encyclopedia}}:|journal=ignored (help) - ^ a b Szallasi A, Sheta M (September 2012). "Targeting TRPV1 for pain relief: limits, losers and laurels". Expert Opinion on Investigational Drugs. 21 (9): 1351–69. doi:10.1517/13543784.2012.704021. PMID 22780443. S2CID 32934502.
- ^ a b Trevisani M (26 July 2010). "Targeting TRPV1: Challenges and Issues in Pain Management" (PDF). The Open Drug Discovery Journal. 2 (3): 37–49. doi:10.2174/1877381801002030037 (inactive 1 November 2024). ISSN 1877-3818.
{{cite journal}}: CS1 maint: DOI inactive as of November 2024 (link) - ^ Khairatkar-Joshi N, Szallasi A (January 2009). "TRPV1 antagonists: the challenges for therapeutic targeting". Trends in Molecular Medicine. 15 (1): 14–22. doi:10.1016/j.molmed.2008.11.004. PMID 19097938.
- ^ a b Brederson JD, Kym PR, Szallasi A. Targeting TRP channels for pain relief. Eur J Pharmacol 2013;716:61-76
- ^ Papoiu AD, Yosipovitch G (June 2010). "Topical capsaicin. The fire of a 'hot' medicine is reignited". Expert Opinion on Pharmacotherapy. 11 (8): 1359–71. doi:10.1517/14656566.2010.481670. PMID 20446852.
- ^ Palazzo E, Luongo L, de Novellis V, Berrino L, Rossi F, Maione S (October 2010). "Moving towards supraspinal TRPV1 receptors for chronic pain relief". Molecular Pain. 6: 1744-8069–6-66. doi:10.1186/1744-8069-6-66. PMC 2959024. PMID 20937102.
- ^ Lambert DG (February 2009). "Capsaicin receptor antagonists: a promising new addition to the pain clinic". British Journal of Anaesthesia. 102 (2): 153–5. doi:10.1093/bja/aen354. PMID 19151045.
- ^ Lambert GA, Davis JB, Appleby JM, Chizh BA, Hoskin KL, Zagami AS (October 2009). "The effects of the TRPV1 receptor antagonist SB-705498 on trigeminovascular sensitisation and neurotransmission". Naunyn-Schmiedeberg's Archives of Pharmacology. 380 (4): 311–25. doi:10.1007/s00210-009-0437-5. PMID 19690836. S2CID 11113986.
- ^ Holzer P (March 1988). "Local effector functions of capsaicin-sensitive sensory nerve endings: involvement of tachykinins, calcitonin gene-related peptide and other neuropeptides". Neuroscience. 24 (3): 739–68. doi:10.1016/0306-4522(88)90064-4. PMID 3288903. S2CID 35325223.
- ^ a b c d "Zuacta (zucapsaicin cream) product monograph" (PDF). Sanofi Canada.
- ^ a b c d WO 2011100668, Bernstein JE, "Method and compositions of civamide to treat disease of the intestines.", published 2011
- ^ Schnitzer TJ, Pelletier JP, Haselwood DM, Ellison WT, Ervin JE, Gordon RD, et al. (March 2012). "Civamide cream 0.075% in patients with osteoarthritis of the knee: a 12-week randomized controlled clinical trial with a longterm extension". The Journal of Rheumatology. 39 (3): 610–20. doi:10.3899/jrheum.110192. PMID 22089461.
- ^ "Zucapsaicin. Computed Properties". PubChem. U.S. National Library of Medicine. Retrieved 23 August 2019.
