User:Hansonrstolaf/Sandbox/Nicolaou
'The Nicolaou Taxol total synthesis', published by K. C. Nicolaou and his group in 1994 concerns the total synthesis of Taxol. This organic synthesis is considered a classic in organic chemistry. [1] Taxol is an important drug in the treatment of cancer but also expensive because the compound is harvested from a scarce resource, namely the pacific yew.
This synthetic route to Taxol is one of several; other groups have presented their own solutions, notably the group of Holton with a linear synthesis starting from borneol, the Danishefsky group starting from the Wieland-Miescher ketone and the Wender group from pinene.
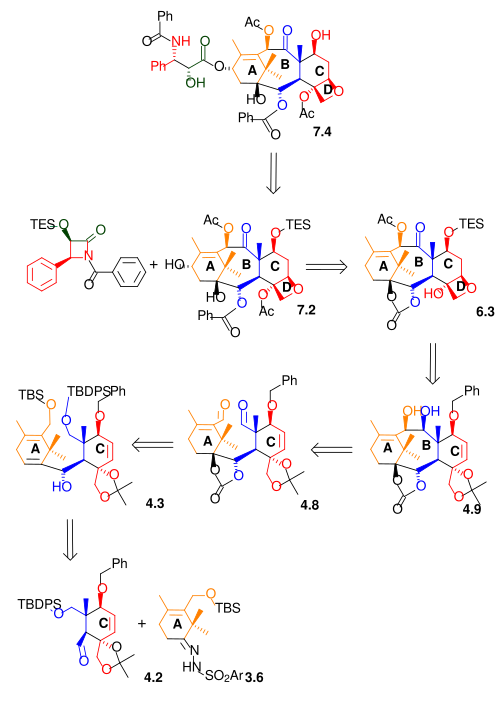
The Nicolaou synthesis is a good example of convergent synthesis because the molecule is assembled from 3 pre-assembled synthons.Two major parts are cyclohexene rings A and C that are connected by two short bridges creating an 8 membered ring in the middle (ring B). The third pre-assembled part is an amide tail. Ring D is an oxetane ring fused to ring C [2] [3] [4][5] [6] [7] [8] .
Two key chemical transformations are the Shapiro reaction and the pinacol coupling reaction.
Retrosynthesis
[edit]
 |
| Scheme 1 |
|---|
 |
| Scheme 2 |
|---|
C Ring synthesis
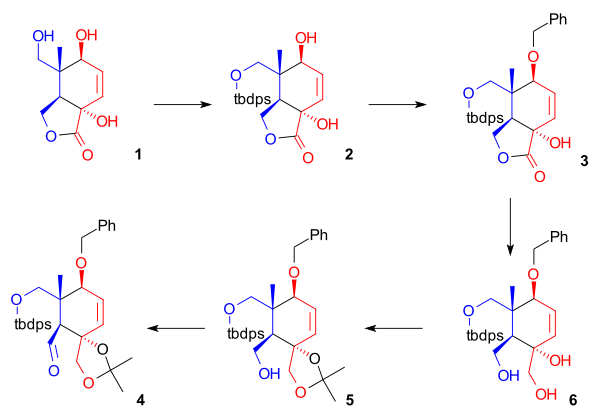
[edit]As shown in Scheme 1, the ring synthesis of ring C starts with a condensation reaction of phenylboronic acid 1.2 with the diene 3-hydroxy-2-pyrone 1.3 and dienophile 1.2 to the boronic ester 1.4. Boron serves as a template (or molecular tether) and aligns both diene and dienophile for an endo Diels-Alder cycloaddition to a bicyclic[2.2.2]lactone (1.5). The boronate ester is cleaved again in neopentyl glycol to the diol 1.6. A lactone rearrangement reaction takes place to a Bicyclo[4.2.0]lactone 1.7 with the formation of a 5-membered lactone and ring-opening of 6-membered lactone. With t-butyldimethylsilyltriflate and DMAP a silylation to 1.9 takes place preceded by the formation of an acetal protecting group in 1.8. All sensitive groups now protected for an ester reduction with lithium aluminium hydride to 1.10. Finally the protecting groups are removed by camphorsulfonic acid to 1.11.
 |
| Scheme 1 |
|---|
In the next series of steps (Scheme 2), four hydroxyl group are protected leaving one remaining hydroxyl group exposed for oxidation to an aldehyde. In preparation to a lactone reduction, the protection of the primary alcohol 2.1 with TPSCl or t-butyldiphenylsilyl chloride with imidazole as a base is performed to a TBDPS silyl ether 2.2 followed by protection of the secondary alcohol group by benzylbromide with potassium hydride as a base and tetra(n-butyl)ammonium iodine as a phase transfer catalyst to a benzyl protecting group 2.3. Reduction of the lactone takes place with lithium aluminium hydride, liberating two additional hydroxyl groups. The compound 2.4 now contains 5 hydroxyl groups two of which protected as a silyl ether. The vicinal diol group is protected by transacetalization with 2,2-dimethoxypropane to 2.5. The final remaining primary alcohol group is selectively oxidized to the aldehyde by TPAP and N-methylmorpholine N-oxide. This aldehyde (2.6) is the terminus for docking with the vinyllithium group in ring part A.
 |
| Scheme 2 |
|---|
A Ring synthesis
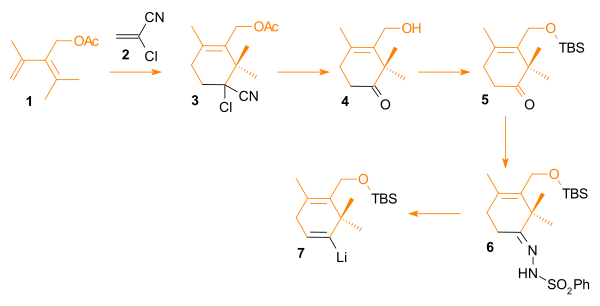
[edit]The A ring synthesis (Scheme 3) starts with a Diels-Alder reaction of the diene 3.1 with the commercially available dienophile 2-chloroacrylonitrile 3.2 to the DA product 3.3 with complete regioselectivity. Gem halide hydrolysis of the gem cyanochloro group to a ketone and simultaneous hydrolysis of the acetate group to the alcohol leads to the hydroxy ketone 3.4. The hydroxyl group is protected by silylation with tert-butyldimethylsilylchloride (TBSCl) to the silyl ether 3.5. A Shapiro reaction of the ketone group with p-toluenesulfonylhydrazide and n-butyllithium leads through the hydrazone 3.6 to the viyllithium compound 3.7. This nucleophile reacts with the aldehyde group present in ring C in Scheme 4.
 |
| Scheme 3 |
|---|
B Ring synthesis
[edit]
The coupling of ring A and ring C creates the 8 membered B ring. One connection is made via a nucleophilic addition of a vinyllithium compound to an aldehyde and the other connection through a pinacol coupling reaction of two aldehydes (scheme 4).
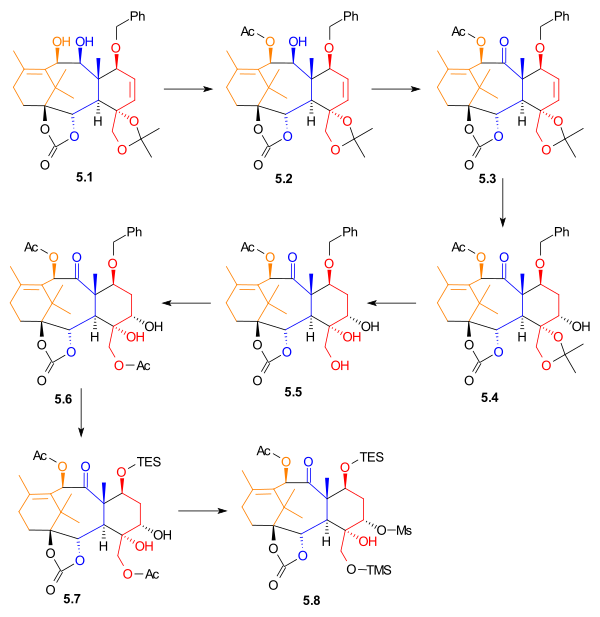
The nucleophilic addition of the vinyllithium compound 4.1 to aldehyde 4.2 is the first part in the ring closure (Scheme 4). The control of stereochemistry in 4.3 is assured because the lithium atom coordinates with the two oxygen atoms in the dioxolane ring and the nucleophile has a hindered Si face approach due to the proximity of the axial methyl group. A peroxidation with vanadyl(acetylacetate) converts the alkene bond into the epoxide 4.4 which is in turn reduced to the vicinal diol 4.5 with lithium aluminium hydride. This diol is then protected as the carbonate ester 4.6 by reaction with phosgene and potassium hydride. The carbonate group also serves to create rigidity in the ring structure for the imminent pinacol coupling reaction. The two silyl ether groups are removed by the fluoride source tetra-n-butylammonium fluoride and the diol 4.7 is formed. The two free hydroxyl groups (out of the total of 7 hydroxyl groups) are now oxidized by the TPAP / NMO combination to the dialdehyde 4.8 and the final step is the pinacol coupling reaction in a McMurry fashion with Titanium(III) chloride and a zinc / copper alloy to the diol 4.9.
 |
| Scheme 4 |
|---|
Resolution
[edit]At this point in the synthesis of Taxol, the material was a racemic mixture. To obtain the desired enantiomer, allylic alcohol 4.9 was acelated with (1S)-(-)-camphanic chloride and dimethylaminopyridine, giving two diastereomers. These were then separated using standard column chromatography. The desired enantiomer was then isolated when one of the separated disatereomers was treated with potassium bicarbonate in methanol.
 |
| Enantiomeric resolution of 4.9. |
D Ring synthesis
[edit]The desired enantiomer from resolution, allylic alcohol 5.1 (Scheme 5) was acetylated with acetic anhydride and 4-(dimethylamino)pyridine in methylene chloride to yield monoacetate 5.2. It is noteworthy that this reaction was exclusive for the allylic alcohol, and the adjacent alcohol group was not acetylated. Alcohol 5.2 was oxidized with tetrapropylammonium perruthenate and N-methylmorpholine N-oxide to give ketone 5.3. alkene 5.3 underwent hydroboration in tetrahydrofuran. Oxidation with basic hydrogen peroxide and sodium bicarbonate gave alcohol 5.4 in 35% yield, with 15% yield of a regioisomer. The acetonide was removed, giving triol 5.5. This alcohol was monoacetylated, to give acetate 5.6. The benzyl group was removed, and replaced with a triethylsilyl group. Diol 5.7 was selectively activated using methanesulfonyl chloride and 4-(dimethylamino)pyridine to give mesylate 5.8, in 78% yield.
 |
| Scheme 5 |
|---|
Tail addition
[edit]As shown in Scheme 7, the Ojima lactam 7.1 reacts with alcohol 7.2 with sodium bis(trimethylsilyl)amide as a base. This alcohol (10-deacetylbaccatinIII) is a naturally occurring compound found in Taxus baccata also known as the European Yew in concentrations of 1 gram per kilogram leaves. Therefore this reaction step is also a semi-synthesis. The final step in this reaction sequence is the hydrolysis of the remaining silyl ether with hydrofluoric acid and pyridine. The Taxol molecule is often displayed with an additional benzoyl group in the amide tail. This modification can be included by reaction of 7.4 with benzoyl chloride in the Schotten-Baumann reaction.
 |
| Scheme 7 |
|---|
Precursor synthesis
[edit]Synthesis of the diene precursor in ring C: The ethyl ester of propionic acid 1 is brominated (2) and then converted to the Wittig reagent 3 with triphenylphosphine. This compound reacts with the aldehyde 6 in a Wittig reaction. This aldehyde is obtained from allyl alcohol 4, with the alcohol group protected as a silyl ether 5 with tert-butyldiphenylsiliyl chloride and the allyl group oxidized by ozonolysis (6). The Wittig reaction product 7 is deprotected to the allyl alcohol 8

Synthesis of the diene precursor in ring A: acetone 1 and acetylacetonate 2 react in an aldol condensation to the β-keto-ester 3. The ketone group is reacted with methylmagnesium bromide derived from methyl bromide in a Grignard reaction to the alcohol 4. The final three steps are an acid catalyzed elimination reaction to the diene 5 , ester reduction to 6 and acylation to 7

See also
[edit]- Paclitaxel total synthesis
- Danishefsky Taxol total synthesis
- Holton Taxol total synthesis
- Kuwajima Taxol total synthesis
- Mukaiyama Taxol total synthesis
- Wender Taxol total synthesis
External links
[edit]References
[edit]- ^ Classics in Total Synthesis: Targets, Strategies, Methods K. C. Nicolaou, E. J. Sorensen ISBN 3-527-29231-4
- ^ Total synthesis of taxol K. C. Nicolaou, Z. Yang, J. J. Liu, H. Ueno, P. G. Nantermet, R. K. Guy, C. F. Claiborne, J. Renaud, E. A. Couladouros, K. Paulvannan & E. J. Sorensen Nature 367, 630 - 634 (17 February 1994) Abstract Pubmed
- ^ Synthesis of C-2 Taxol Analogues Kyriacos Costa Nicolaou, Elias A. Couladouros, Phillipe G. Nantermet, Joanne Renaud, Rodney Kiplin Guy, Wolfgang Wrasidlo Angewandte Chemie International Edition Volume 33, Issue 15-16 , Pages 1581 - 1583 1994 Abstract
- ^ Synthesis of a fully functionalized CD ring system of taxol K. C. Nicolaou, J. J. Liu, C.-K. Hwang, W.-M. Dai and R. K. Guy Journal of the Chemical Society, Chemical Communications, 1992, (16), 1118 - 1120
- ^ A convergent strategy towards taxol. A facile enantioselective entry into a fully functionalized ring A system K. C. Nicolaou, C.-K. Hwang, E. J. Soresen and C. F. Clairborne Journal of the Chemical Society, Chemical Communications, 1992, (16), 1117 - 1118
- ^ Novel chemistry of taxol. Retrosynthetic and synthetic studies K. C. Nicolaou, P. G. Nantermet, H. Ueno and R. K. Guy Journal of the Chemical Society, Chemical Communications, 1994, (3), 295 - 296
- ^ Synthesis of ABCtaxoid ring systems via a convergent strategy K. C. Nicolaou, Zhen Yang, Erik J. Sorensen and Masahisa Nakada Journal of the Chemical Society, Chemical Communications, 1993, (12), 1024 - 1026
- ^ Synthesis of Novel Taxoids K. C. Nicolaou, Christopher F. Claiborne, Philippe G. Nantermet, Elias A. Couladouros, and Erik J. Sorensen J. Am. Chem. Soc.; 1994; 116(4) pp 1591 - 1592; doi:10.1021/ja00083a063
