User:Hakkinen2013/sandbox
Assignment 1
[edit]I just got wait listed for medical school and in other news...
cAMP is an amazing second messenger necessary for myelination in Mouse Schwann cells [1]

Assignment 2
[edit]"The proteasome most exclusively used in mammals is the cytosolic 26S proteasome, which is about 2000 kilodaltons (kDa) in molecular mass containing one 20S protein subunit and two 19S regulatory cap subunits." I would like to edit this to be...
The proteasome most exclusively used in mammals and found "exclusively in Eukaryotes" [2] is the cytosolic 26S proteasome, which is about 2000 kilodaltons (kDa) in molecular mass containing one 20S protein subunit and two 19S regulatory cap subunits.
- The talk page as had a section added about the S26 proteasome Talk:Proteasome
- Links to my three page edits and comments Adhesion-GPCRs , Talk:ErbB , Talk:EGR1
here is a link to my edit for Adhesion-GPCRs [1] and ErbB 2 and finally EGR1 3
- Link to my added info for Wikipedia talk:WikiProject Cell Signaling
- Link to my added info for User talk:Biolprof
Assignment 3
[edit]I will be mock adding to two headings in the Adhesion-GPCRs page. The two headings are ligand and signaling.
- Ligand A majority of the Adhesion GPCRs are orphan receptors and work is currently underway to de-orphanize as many of these receptors as possible.[3] Adhesion GPCRs get their name from their N-terminal domains that have adhesion-like domains, such as EGF.[3] While ligands for many receptors are still not known, researchers are utilizing drug libraries to investigate compounds that can activate GPCRs and using these data for future ligand research.
- Signaling Adhesion GPCRs follow standard GPCR signaling and signal through Gαs, Gαq, Gαi, and Gα12/13. As of today, many of the 800 Adhesion GPCRs are still orphan receptors and their signalling pathways are also not identified. Research groups are working to elucidate the downstream signaling molecules utilizing several methods, including chemical screens and analysis of second messenger levels in over-expressed cells. Adding drugs in vitro, while the cells are over-expressing an Adhesion GPCR has allowed the identification of which molecules are activating the GPCR and the second messenger it is utilizing.
I am trying to figure out a way to cite a source only once and then reuse the citation number over instead of it becoming a new citation each time.
- I believe you just reuse the ref tags from the previous citation (so that they're exactly the same) and it will show up under the same number. Maybe also ask Grace, as she is our group's Wikipedia point person.
- got it thanks!
- All comment changes considered and changed. Thanks for all the suggestions.
- got it thanks!
Assignment 4
[edit]Evolution of Adhesion GPCRs: Adhesion GPCRs are found in Fungi.[4] It is believed that Adhesion GPCRs evolved from the cAMP receptor family and arose approximately 1275 million years ago, before the split of Unikots from a common ancestor. Several Fungi have novel adhesion GPCRs that have both short, 2-66 amino acid residues, and long, 312-4202 amino acid residues. Analysis of Fungi showed that there were no secretin receptor family GPCRs, which suggests that they evolved from Adhesion GPCRs in a later organism.
Genome analysis of the TeleostFugu has revealed that it has only two adhesion GPCRs that showed homology to Ig-hepta/GPR116.[5] While the Fugu genome is relatively compact and limited with the number of adhesion GPCRs, the Tetraodon nigroviridis another species of puffer fish has considerably more, 29 adhesion GPCRs.
Ligands Adhesion GPCRs have numerous orphan receptors and many groups as of 2013 are working to de-orphanize them. One GPCR, GPR56, currently has a known ligand, Collagen III, which is involved in neural migration inhibition.[6] GPR56 has been shown to be the cause of polymicrogyria in humans and may play a role in cancer metastasis. The binding of Collagen III to GPR56 occurs on the N-terminus and has been narrowed down to a short stretch of aminoacids. The N-terminus of GPR56 is naturally glycosylated, but this glycosylation is not necessary for Collagen III binding. Collagen III, results in GPR56 to signal through Gα12/13 activating RhoA.
Role in disease: BFPP Mutations in GPR56 result in bilateral frontoparietal polymicrogyria in humans, characterized by abnormal neuronal migration and surface ectopias.[7]
Signaling Adhesion GPCRs follow standard GPCR signaling and signal through Gαs, Gαq, Gαi, and Gα12/13. As of today, many of the 800 Adhesion GPCRs are still orphan receptors and their signalling pathways are also not identified. Research groups are working to elucidate the downstream signaling molecules utilizing several methods, including chemical screens and analysis of second messenger levels in over-expressed cells. Adding drugs in vitro, while the cells are over-expressing an Adhesion GPCR has allowed the identification of which molecules are activating the GPCR and the second messenger it is utilizing.
GPR133 signals through Gαs to activate adenylyl cyclase.[8] It has been shown that overexpressing GPCRs in vitro leads can result in receptor activation in absence of ligand or agonist. By over expressing GPR133 in vitro and an elevation in reporter genes and cAMP was observed. Signaling of the overexpressed GPR133 did not require an N-terminus, nor was GPS cleavage. Missense mutations in the 7tm resulted in loss of signalling.
Lat-1 was shown in C. elegans to require a GPS for signaling, but cleavage at the GPS site was not.[9] Having a shortened 7 transmembrane domain, but an intact GPS domain also resulted in a loss of signaling, suggesting that the two are involved in signaling together and that the GPS site could act as an endogenous ligand.
Bone marrow and hematapoietic stem cells
[edit]GPR56 knockout mice have a decreased number of hematopoietic stem cell in the bone marrow, while higher levels were found in the spleen and peripheral blood.[10] This suggest that GPR56 plays a role in interactions between bone marrow and hematapoietic stem cells.
Neuronal development
[edit]GPR126 is necessary for Schwann cell myelination. Knockouts of this GPCR in both Danio rerio and Mus musculus result in an arrest at the promyelinating stage.[11][12] Schwann cells arise from the neural crest that migrate to peripheral nerves to form either myelinating and non myelinating cells. In GPR126 knockouts, these precursor cells develop to the promyelinating stage, where they have wrapped aproximatley 1.5 times. Myelination is arrested at the promyelinating stage and in fish no myelin basic protein can be detected. In fish this can be rescued by adding Forskolin during development, which rescues myelin basic protein expression.
Miscellaneous
[edit]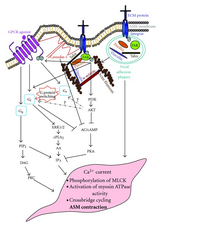
Cross Talk
[edit]GPCRs downstream signals have been shown to interact with integrin signals, such a FAK. [13] (in GPCR page)
Diffs
[edit]GPCRs: cross talk [2]
Adhesion-GPCRs: [3]
future papers
[edit]
Assignment 7
[edit]VlgR1:
The adhesion GPCR Very Large GPCR receptor 1 (Vlg1R1) is the largest GPCR known, with a size of 6300 amino acids and consisting of 90 exons.[14] There are 8 splice variants of VlgR1, named VlgR1a-1e and Mass1.1-1.3. The N-terminus consists of 5800 amino acids containing 35 Calx-beta domains, one pentraxin domain, and one epilepsy associated repeat. Mutations of VlgR1 have been shown to result in Usher's syndrome. Knockouts of Vlgr1 in mice have been shown to phenocopy Usher's syndrome and lead to audigoenic seizures.
The Sea Urchin genome has a homolog of Vlgr1 in it.[15]
BaI3:
The adhesion GPCR BaI3 is an orphan receptor that has a long N-terminus consisting of one cub domain, five BaI Thrombospondin type 1 repeats, and one hormone binding domain.[16] BaI3 is expressed in neural tissues of the central nervous system. BaI3 has been shown to have a high affinity for C1q proteins. C1q added to hippocampal neurons expressing BaI3 resulted in a decrease in the number of synapses.
CD97:
CD97 is an adhesion GPCR that is normally found on tissue of hematapoetic origin and in smooth muscle.[17] It signals through Gα12/13 and activates RHO. It has been found to be upregulated in Cancers, including prostate, breast, and thyroid. In prostate cancer. it has been shown that CD97 is hetoerodimerizing with another GPCR, LPAR. The interaction is believed to further enhance the RHO signaling between both receptors.[18]
GPR56 expanded:
GPR56 has been shown to be cleaved at the GPS site and then remain associated with the 7TM domain.[19] In a study where the N-terminus was removed up to N342, the start of the GPS, the receptor became constitutively active and an up regulation of Gα12/13 was seen. When receptors are active they are ubiquitinated and GPR56 lacking an N-terminus was highly ubiquitinated.
EMR2:
EGF-like module-containing mucin-like hormone receptor-like 2 (EMR2) is an adhesion GPCR that undergoes GPS autoproteolysis before being trafficked to the plasma membrane.[20] Mutations in the GPS have shown that EMR2 does not need to undergo autoproteolysis to be trafficked, but loses function. EMR2 has been shown to be necessary for in vitro cell migration.
Upon cleavage the N-terminus has been shown to associate with the 7TM, but to also dissociate, giving two possible functions. When the N-terminus dissociates it can be found in lipid rafts. Additionally, the cleaved EMR2 protein 7TM has been found to associate with EMR4 N-terminus.
GPR64:
The adhesion GPCR, GPR64, is an orphan receptor characterized by a long N-terminus with that has been suggested to be highly glycosylated.[21] GPR64's N-terminus has been reported to be cleaved at the GPS domain to allow for trafficking to the plasma membrane. After cleavage the N-terminus is believed to remain non-covalently associated with the 7TM. Male mice lacking GPR64 are infertile.
GPR64 expression has been mostly reported in the male reproductive organs, but more recently has been shown to be expressed in the central nervous system.[22]
Reorganization of the Adhesion GPCRs:
There are 33 human adhesion GPCRs that can be broken down in the 8 groups and 2 independent receptors. Group I consists of Lec1,Lec2,Lec3, and ETL. Group II consists of CD97, EMR1, EMR2, EMR3, and EMR4. Group III consists of GPR123, GPR124, and GPR125. Group IV consists of CELSR1, CELSR2, and CELSR3. Group V consists of GPR133 and GPR144. Group VI consists of GPR110, GPR111, GPR113, GPR115, and GPR116. Group VII consists of BAI1, BAI2, and BAI3. Group VIII consists of GPR56, GPR97, GPR112, GPR114, GPR126, and GPR64. There are two other adhesion GPCRs that are not in these groups, VLGR1 and GPR128.[23]
1. The first thing I noticed was the structure of the article didn't seem to be the best. There is a template about cell signaling pathways that begins with the history/discovery, then the mechanism, then the role, then the human diseases, associated with that structure or pathway, and then the research that is currently being performed on the structure and mechanism. Hopefully this will be a good place to begin. It worked very well for my topic.
I will consider changing the format with headings, but most likely not until the final set of revisions.
2.Also that list of human adhesion proteins seems like it is sort of in the way and doesn't provide much to the overall article. Maybe putting it towards the end of the article would be good, as a source where people can go and check on one of the many adhesion proteins.
Moved.
3.There were many places that I felt citations should be made. I was originally going to go through sentence by sentence where I felt there should be citations, but I feel like you know where they should be made. The entire section regarding cleavage had only a couple citations that I felt were not necessary. I completely redid that paragraph as will become apparent later.
I have the mind set that one citation for relevant chunk of information should be fine, but I will add more where there are strong assertions. I did not write the cleavage section, but thank you for editing it.
4.I also moved some citations from the beginning of paragraphs to the end of them, as the one citation was referring to the entire paragraph and not just the first sentence.
sure that works just as well.
5. For the first paragraph under signaling, you indicate a technique used for the identification of molecules interacting with the GPCR. However, I feel like citing this technique with a paper that used the technique would be beneficial for people to see how it was done.
I will see what I can find.
6. I added citations towards the ends of the paragraphs about the two adhesion proteins listed in the third and fourth paragraphs. I also have an idea that would dramatically change the structure that I will talk about a little bit further on.
Nice and thank you.
7. The cleavage section needed a whole overhaul of the language that was used. I felt it was much to confusing and complex for many reasons. I have tried to make it so that a causal viewer can come upon the page and read it with good understanding. If a more advanced person wants knowledge, I feel that that is when they should go to the citations. Also, there were only three citations in this entire section and they all referred to other organisms where the GAIN domain is found. I thought this sentence to be pretty useless and have taken it out, among many other words that I felt were unnecessary. If you don't like the change, feel free and put it back the way it was. I feel that this is more of the style that Wikipedia is going after, however. I didn't want to go in and change everything about the article, but I wanted to provide this as an example for how it should be done I feel. The page is yours, however, so feel free to make the changes you feel should be made. I am just providing that as an example.
That section was again not written by me but I will follow your lead and put some more focus on adding clarity to it.
8.The section on the immune system I found to be very hard to read. As someone with some background knowledge regarding biology at this level, I still found it very difficult to read through.
Again not written by me, but I will work on that one too.
9. The sentence regarding the BAI1 adhesion protein, I redid as well. I thought that by stating what the protein was thought to be involved in and then stating what it actually is involved in was too similar to the review article this came from. It also, at least for me, adds nothing to my knowledge and current understanding about the protein if it is listed about what the protein was thought to have been involved in.
10.I liked the way the GPCR adhesion protein GPR126 was described in the Neuronal Development section. I liked that it had multiple citations, and the way the information on this page was written was not anywhere similar to the way that it was presented in the articles that cited it. I thought that was very well done.
Why thank you.
11.I didn't like the way that GPR56 was addressed in the entire article. Little pieces of it are found everywhere. A paragraph is devoted to it in the ligand section. There is a sentence about it in the immune system section, a sentence about it in the bone marrow section, and a sentence about it in the bilateral frontoparietal polymicrogyria section. This brings me to a main point that I had regarding the article as a whole. I feel that a much better structure for this article were to describe each GPCR adhesion protein as if it were its own perhaps mini article, where the ligands, signaling, functional roles, and roles in disease were all put into one section that was all under the heading of the GPR56. Perhaps a better method would be to even put most of this information in the GPR56 page of wikipedia. I also noticed with the GPR12 protein, there was more information on the GPCR adhesion protein in this article than there was on the actual GPR12 article itself. It might be hard to figure out where the information should go, especially if you are trying to fix the main article, regarding ahesion GPCRs. I went searching on Pubmed for general review articles about adhesion GPCRs and found several that seemed to be very good places to begin, as a sort of overview of the GPCR adhesion proteins in general. It seemed like many of the articles that you have cited are primary articles, that could be very dense and hard to get through, while coming to a conclusion that yields the same knowledge that one sentence of a review article could provide. Those primary articles also seem to focus in on one mechanism or structure of one GPCR adhesion protein without addressing what you want to be addressing, which is the overall view of GPCR adhesion proteins.
I will mull this over for a few days and then maybe make the moves as you suggest.
12. This leads me to another very important point, which is that the way you cited the articles, one on Wikipedia is not simply able to follow the link to a website such as Pubmed. I have included where you can use citations for the Pubmed journal articles Wikipedia template filling tool. I will do a couple for you. When doing this, you need to make sure you press the "Add ref tag" button below. I think it is very important to make them so you are able to follow the link to the article online. I feel that this is very important for someone who is doing some research of their own and have gone to Wikipedia as a starting place. In the past, I have gone to a Wikipedia page to mainly look for articles that were cited at the bottom of the page and not pay heed to the information that was presented at the top. Here are a few more things about the citations, upon which I clicked on and delved into the article a bit. In the second paragraph of signaling, regarding the GPR133 protein, the wording you used and the wording that was used in the research article is very similar, except where you use "via" they use the word "through."
13. For the the citation regarding the Lat-1 protein, I felt that the sentence summed up the article very well without directly copying any of their specific word usage. I thought that this was very well done. I thought that the paragraph was a little wordy as well. I feel like you could simply state that the GPS domain and the 7 transmembrane domain are required for the Lat-1 signaling. In addition, I was not convinced after going into the article that the GPS site was necessary as part of an endogenous ligand. But you have probably read it more clearly than I have. In addition, when you link to the GPS, it gives you the Global Positioning System page, and not the GPS you are talking about.
14. For the citation and the overall entry about the bone marrow GPCR adhesion protein, I felt that taking one sentence from the primary literature on one GPCR adhesion protein and using that as part of a section on the Wikipedia entry of all of the GPCR adhesion proteins may not be the best way to style the article. I also made the sentence more concise.
I need to add more to it.
Diffs
[edit]as of 4/16/2013 Adhesion GPCR
Assignment 8
[edit]part 1 peer review
[edit]These are responses to comments from Biosthmors as I did not find Grace's suggestions.
1. I will see what I can do with the rewording of the intro paragraph: In the lead paragraph, I am deleting the first paragraph because it is repeated in a later section. This should help shorten the lead. Other changes may follow. I feel the lead offers a good "abstract" style for the article and although it may be long is informative for those that would like to skim and get a general idea of what Adhesion GPCRs are.
2. I added adhesion g protein coupled receptor to the beginning.
3. The not so recent work that recently says "recently" is not mine, but that recent work will soon become recently changed. The current work of mine that currently says currently in two places is currently about to be changed concurrently of one another.
4. Super- means very large combined with family meaning all those persons considered as descendants of a common progenitor. Redirecting to protein family should be enough.
5. Not my work, but I will correct it. I think metazoans is fine as people should learn what it means... I will link it as well.
6.I would agree a table may be better, but I think for now it is fine.
7. I left it in for a quick reference at the bottom, but will delete it.
8. yes? The proposed part was removed seemed a little strange.
10. Thank you!
11. Possibly in future revisions
12. I think Adhesion-GPCR is good for now as a tittle, but that is something that I think people should know what GPCR stands for and can easily find out.
Here are the Difs
part two peer review of other articles
[edit]All suggestions are made on the peer review area of each article.
-Matt's plant article is very well done and I do like his use of bullet points. I have no comments as of now to add to the plant article section.
-Caitlin's SR protein article: -linked metazoans. -I think the last line of the lead could be put else where.
-Max's small vesicles is pretty good covers a broad range of topics and gives insights into not just signaling and mechanisms of release, but into disease implications as well. No comments or changes as of yet.
-Julia's GST article: -"GSTs can constitute up to 10% of cytosolic protein in some mammalian organs" would it be possible to say what organs?
-Grace's WNT is good.
-Bobby's mTor: -There are some differences with the bold font for titles of sections.
References
[edit]- ^ http://www.ucl.ac.uk/jessenmirsky/wp-content/uploads/2009/08/Glia-2011-Arthur-Farraj.pdf
- ^ The proteasome: molecular machinery and pathophysiological roles Tannaka K et al
- ^ a b amila Gupte, Gayathri Swaminath, Jay Danao, Hui Tian, Yang Li, Xinle Wu Signaling property study of adhesion G-protein-coupled receptors FEBS Letters, Volume 586, Issue 8, 24 April 2012, Pages 1214–1219
- ^ Krishnan A., Almén M. S., Fredriksson R., Schiöth H. B. (2012). The origin of GPCRs: identification of mammalian like Rhodopsin, Adhesion, Glutamate and Frizzled GPCRs in fungi. PLoS ONE 7:e29817. doi: 10.1371/journal.pone.0029817
- ^ Sarkar A.,Kumar S., and Sundar D.(2011). The G protein-coupled receptors in the pufferfish Takifugu rubripes. BMC Bioinformatics 12(S-1):S3
- ^ Luo R, Jin Z, Deng Y, Strokes N, Piao X (2012) Disease-Associated Mutations Prevent GPR56-Collagen III Interaction. PLoS ONE 7(1): e29818. doi:10.1371/journal.pone.0029818
- ^ Singer K., Luo R., Jeong S., Piao X. (2012) GPR56 and the Developing Cerebral Cortex: Cells, Matrix, and Neuronal Migration. Springer Science+Business Media, LLC 2012 10.1007/s12035-012-8343-0
- ^ Jens Bohnekamp and Torsten Schöneberg. Cell Adhesion Receptor GPR133 Couples to Gs Protein J. Biol. Chem. 2011 286: 41912-41916. First Published on October 24, 2011, doi:10.1074/jbc.C111.265934
- ^ Prömel S, Frickenhaus M, Hughes S, Mestek L, Staunton D, Woollard A, Vakonakis I, Schöneberg T, Schnabel R, Russ AP, Langenhan T. The GPS motif is a molecular switch for bimodal activities of adhesion class G protein-coupled receptors.Cell Rep. 2012 Aug 30;2(2):321-31. doi: 10.1016/j.celrep.2012.06.015. Epub 2012 Aug 2. Erratum in: Cell Rep. 2012 Sep 27;2(3):705.
- ^ Y Saito, K Kaneda, A Suekane, E Ichihara, S Nakahata, N Yamakawa, K Nagai, N Mizuno, K Kogawa, I Miura, H Itoh, K Morishita, Maintenance of the hematopoietic stem cell pool in bone marrow niches by EVI1-regulated GPR56,Leukemia accepted article preview 12 March 2013; doi: 10.1038/leu.2013.75.
- ^ Monk KR, Oshima K, Jörs S, Heller S, Talbot WS. Gpr126 is essential for peripheral nerve development and myelination in mammals" Development 2011 Jul;138(13) 2673-80. Epub 2011 May 25.
- ^ Monk KR, Naylor SG, Glenn TD, Mercurio S, Perlin JR, Dominguez C, Moens CB, Talbot WS. A G protein-coupled receptor is essential for Schwann cells to initiate myelination. (2009) Science. 325: 1402-5.
- ^ Chun Ming Teoh, John Kit Chung Tam, Thai Tran. Integrin and GPCR Crosstalk in the Regulation of ASM Contraction Signaling in Asthma. J Allergy (Cairo). 2012; 2012: 341282. Published online 2012 September 29. doi: 10.1155/2012/341282
- ^ Sun JP, Li R, Ren HZ, Xu AT, Yu X, Xu ZG. The very large g protein coupled receptor (vlgr1) in hair cells.J Mol Neurosci. 2013 May;50(1):204-14. doi: 10.1007/s12031-012-9911-5. Epub 2012 Nov 20.
- ^ Charles A. Whittakera, Karl-Frederik Bergerone, James Whittlec, Bruce P. Brandhorste, Robert D. Burked, Richard O. Hynes. The echinoderm adhesome. Developmental Biology. Volume 300, Issue 1, 1 December 2006, Pages 252–266.
- ^ Marc F. Bolliger, David C. Martinelli, and Thomas C. Südhof. The cell-adhesion G protein-coupled receptor BAI3 is a high-affinity receptor for C1q-like proteins. PNAS 2011 ; published ahead of print January 24, 2011, doi:10.1073/pnas.1019577108
- ^ Ward, Y., R. Lake, J. J. Yin, C. D. Heger, M. Raffeld, P. K. Goldsmith, M. Merino, K. Kelly. 2011. LPA receptor heterodimerizes with CD97 to amplify LPA-initiated RHO-dependent signaling and invasion in prostate cancer cells. Cancer Res. 71: 7301–7311.
- ^ Ward Y, Lake R, Martin P L, Killian K, Salerno P, Wang T, Meltzer P, Merino M, Cheng S-Y, Santoro M, Garcia-Rostan G, Kelly K,CD97 amplifies LPA receptor signaling and promotes thyroid cancer progression in a mouse model. Oncogene. 2012/07/16/online
- ^ Paavola KJ, Stephenson JR, Ritter SL, Alter SP, Hall RA (2011) The N terminus of the adhesion G protein-coupled receptor GPR56 controls receptor signaling activity. J Biol Chem 286:28914–28921.
- ^ Huang, Y. S., N. Y. Chiang, C. H. Hu, C. C. Hsiao, K. F. Cheng, W. P. Tsai, S. Yona, M. Stacey, S. Gordon, G. W. Chang, H. H. Lin. 2012. Activation of myeloid cell-specific adhesion class G protein-coupled receptor EMR2 via ligation-induced translocation and interaction of receptor subunits in lipid raft microdomains. Mol. Cell. Biol. 32: 1408–1420.
- ^ Davies B, Baumann C, Kirchhoff C, Ivell R, Nubbemeyer R, Habenicht UF, Theuring F, Gottwald U. Targeted deletion of the epididymal receptor HE6 results in fluid dysregulation and male infertility. Mol Cell Biol 2004; 24: 8642 8648
- ^ Haitina T, Olsson F, Stephansson O, Alsiö J, Roman E, Ebendal T, Schiöth HB, Fredriksson R. Expression profile of the entire family of Adhesion G protein-coupled receptors in mouse and rat. BMC Neurosci. 2008;9:43–43. doi: 10.1186/1471-2202-9-43.
- ^ Schiöth HB, Nordström KJ, Fredriksson R.The adhesion GPCRs; gene repertoire, phylogeny and evolution. Adv Exp Med Biol. 2010;706:1-13.
Arthur-Farraj, P., Wanek, K., Hantke, J., Davis, C. M., Jayakar, A., Parkinson, D. B., Mirsky, R. and Jessen, K. R. (2011), Mouse schwann cells need both NRG1 and cyclic AMP to myelinate. Glia, 59: 720–733. doi: 10.1002/glia.21144
