User:Bc183-clp/sandbox
Introduction
[edit]
| |
| Names | |
|---|---|
| IUPAC name
N-((2S,3R,E)-1-(((2R,3R,4R,5S,6R)-5-(((2S,3R,4R,5R,6R)-5-(((2R,3R,4S,5S,6R)-4-(((2S,3R,4R,5R,6R)-3-acetamido-4-(((2R,3R,4S,5R,6R)-4,5-dihydroxy-6-(hydroxymethyl)-3-(((2S,3S,4R,5S,6S)-3,4,5-trihydroxy-6-methyltetrahydro-2H-pyran-2-yl)oxy)tetrahydro-2H-pyran-2-yl)oxy)-5-hydroxy-6-(hydroxymethyl)tetrahydro-2H-pyran-2-yl)oxy)-3,5-dihydroxy-6-(hydroxymethyl)tetrahydro-2H-pyran-2-yl)oxy)-3,4-dihydroxy-6-(hydroxymethyl)tetrahydro-2H-pyran-2-yl)oxy)-3,4-dihydroxy-6-(hydroxymethyl)tetrahydro-2H-pyran-2-yl)oxy)-3-hydroxyoctadec-4-en-2-yl)palmitamide
| |
Other names
| |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
|
Globo H (globohexaosylceramide) is a globo-series glycosphingolipid antigen that is present on the outer membrane of some cancer cells.[1][2] Globo H is not expressed in normal tissue cells, but is expressed in a number of types of cancers, including cancers of the breast, prostate, and pancreas.[1][3] Globo H’s exclusivity for cancer cells makes it a target of interest for cancer therapies.[1][2]
Structure
[edit]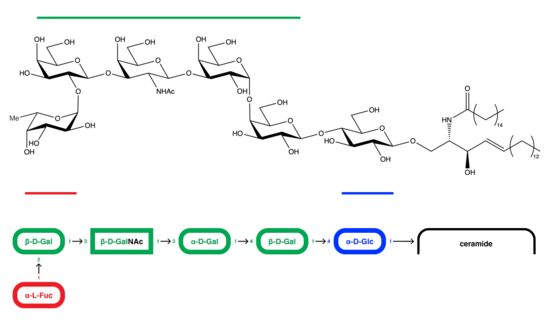
Defined by the monoclonal antibody MBr1, Globo H has been isolated from breast cancer cell line MCF-7, and its structure has been determined through several analyses, including NMR spectroscopy and methylation analysis.[5] Globo H consists of a hexasaccharide of the structure Fucα(1-2)Galβ(1-3)GalNAcβ(1-3)Galα(1-4)Galβ(1-4)Glcβ(1) with a ceramide attached to its terminal glucose ring at the 1 position in a beta linkage.[6]
Synthesis
[edit]Biosynthesis
[edit]
Globo H’s biosynthetic pathway is involved in the synthesis pathways of other globo-series glycosphingolipid antigens that are also specific to cancer cells, including stage-specific embryonic antigen-3 (SSEA3) and stage-specific embryonic antigen-4 (SSEA4).[1] The biosynthetic pathway of these antigens includes the enzyme β 1,3-galactosyltransferase V (β3GalT5).[1] β3GalT5 catalyzes the galactosylation of globoside-4 (Gb4) to SSEA3.[1] SSEA3 can then be converted to SSEA4 by sialyltransferase adding a sialic acid group to its end, or it can be converted to Globo H by fucosyltransferase adding a fucose ring to its end.[1] Playing a part in the formation of three different cancer-specific antigens, β3GalT5 is of particular interest in its relevance to cancer treatment, and it has been shown to be critical for cancer cell survival.[8]
Chemical Synthesis
[edit]In order to study its potential as a cancer therapy target, Globo H has been synthesized in the laboratory.[9] One synthesis is achieved by first building two trisaccharides from their component sugars, and then linking them.[9] The trisaccharides, with most of their functional groups protected to prevent side reactions, are linked by creating the GalNAcβ(1-3)Gal bond.[9] A thioethyl group is added to the 1 position on one of the protected galactose rings, and in the presence of methyl triflate, this reacts with the hydroxyl group on the 3 position of the other galactose to link the trisaccharides and form the hexasaccharide.[9] The ceramide is added to the 1 position of the terminal glucose ring after hexasaccharide formation.[9]
Globo H as a Therapeutic Target
[edit]As a Tumor Associated Carbohydrate Antigen (TACA), Globo-H is a promising clinical target for immunotherapy. While absent in normal tissues, the glycosphingolipid is overexpressed in a variety of epithelial cancer cell types including human pancreatic, gastric, lung, colorectal, esophageal, and breast tumors.[10][11]
Globo H Anticancer Vaccines
[edit]Globo-H’s TACA character allows for its utilization as an anticancer vaccine, inducing antibody response against the epitope. The resulting humoral immunity could enable the selective eradication of Globo H-presenting tumors.[12] The Taiwanese biopharma company OBI Pharma, Inc., was first to develop Adagloxad Simolenin (OBI-822), a Globo H hexasaccharide conjugated with the immunostimulatory carrier protein KLH.[12] The Phase III GLORIA study is underway evaluating the carbohydrate-based immunogen’s effects in high risk triple-negative breast cancer (TNBC) patients with an estimated completion date in 2027.[13]
Alternative vaccine conjugates have been developed which avoid issues associated with the protein carrier KLH by substituting it with a lipid or carbohydrate-based carrier. Examples include the use of lipid A derivatives[14] or entirely carbohydrate vaccine conjugates such as Globo H-PS A1[15].
Anti-Globo H Antibodies
[edit]Globo H-targeting antibodies are another strategy currently being evaluated in the cancer therapeutic space. OBI Pharma’s OBI-888 is a humanized IgG1 antibody that selectively binds to the Globo H antigen among other Globo series glycosphingolipids such as SSEA-3 and SSEA-4.[16] Additionally, in vivo studies of OBI-888 in various Globo H-positive (GH+) xenografts models showed promising tumor growth inhibition results.[17] OBI-888’s human Phase I/II study for the treatment of metastatic and locally advanced solid tumors is estimated to finish in December 2022.[18]
Based on OBI-888, the first-in-class antibody-drug conjugate (ADC) 0BI-999 was additionally developed, linking OBI-888 to monomethyl auristatin E, a synthetic antineoplastic agent.[19] The ADC is currently undergoing phase II trial in patients with advanced solid tumors, with an estimated completion date in Dec 2023.[20] In Dec 2019 & Jan 2020, OBI-999 was granted two Orphan Drug Designations by the FDA for the treatment of pancreatic and gastric cancer.[21]
- ^ a b c d e f g h Chuang, Po-Kai; Hsiao, Michael; Hsu, Tsui-Ling; Chang, Chuan-Fa; Wu, Chung-Yi; Chen, Bo-Rui; Huang, Han-Wen; Liao, Kuo-Shiang; Chen, Chen-Chun; Chen, Chi-Long; Yang, Shun-Min (2019-02-26). "Signaling pathway of globo-series glycosphingolipids and β1,3-galactosyltransferase V (β3GalT5) in breast cancer". Proceedings of the National Academy of Sciences. 116 (9): 3518–3523. doi:10.1073/pnas.1816946116. ISSN 0027-8424. PMID 30808745.
- ^ a b Hung, Jung-Tung; Cheng, Jing-Yan; Wu, Chia-Cheng; Yu, Alice L. (2012-04-15). "Abstract 2529: A monoclonal anti-Globo H antibody, VK9 can mediate CDC/ADCC and inhibit adhesion of Globo H+ cancer cells to extracellular matrix". Cancer Research. 72 (8 Supplement): 2529–2529. doi:10.1158/1538-7445.AM2012-2529. ISSN 0008-5472.
- ^ Yang, Ching-Yao; Lin, Mong-Wei; Chang, Yih-Leong; Wu, Chen-Tu (2017-12-12). "Globo H expression is associated with driver mutations and PD-L1 expressions in stage I non-small cell lung cancer". Cancer Biomarkers: Section A of Disease Markers. 21 (1): 211–220. doi:10.3233/CBM-170660. ISSN 1875-8592. PMID 29036791.
- ^ Park, Tae Kyo; Kim, In Jong; Hu, Shuanghua; Bilodeau, Mark T.; Randolph, John T.; Kwon, Ohyun; Danishefsky, Samuel J. (1996-11-20). "Total Synthesis and Proof of Structure of a Human Breast Tumor (Globo-H) Antigen". Journal of the American Chemical Society. 118 (46): 11488–11500. doi:10.1021/ja962048b. ISSN 0002-7863.
- ^ Bremer, E. G.; Levery, S. B.; Sonnino, S.; Ghidoni, R.; Canevari, S.; Kannagi, R.; Hakomori, S. (1984-12-10). "Characterization of a glycosphingolipid antigen defined by the monoclonal antibody MBr1 expressed in normal and neoplastic epithelial cells of human mammary gland". The Journal of Biological Chemistry. 259 (23): 14773–14777. ISSN 0021-9258. PMID 6501317.
- ^ Wang, Cheng-Chi; Huang, Yen-Lin; Ren, Chien-Tai; Lin, Chin-Wei; Hung, Jung-Tung; Yu, Jyh-Cherng; Yu, Alice L.; Wu, Chung-Yi; Wong, Chi-Huey (2008-08-19). "Glycan microarray of Globo H and related structures for quantitative analysis of breast cancer". Proceedings of the National Academy of Sciences. 105 (33): 11661–11666. doi:10.1073/pnas.0804923105. ISSN 0027-8424. PMID 18689688.
- ^ Detzner, Johanna; Pohlentz, Gottfried; Müthing, Johannes (2020-06-04). "Valid Presumption of Shiga Toxin-Mediated Damage of Developing Erythrocytes in EHEC-Associated Hemolytic Uremic Syndrome". Toxins. 12 (6): 373. doi:10.3390/toxins12060373. ISSN 2072-6651. PMC 7354503. PMID 32512916.
{{cite journal}}: CS1 maint: PMC format (link) CS1 maint: unflagged free DOI (link) - ^ Cheung, Sarah K. C.; Chuang, Po-Kai; Huang, Han-Wen; Hwang-Verslues, Wendy W.; Cho, Candy Hsin-Hua; Yang, Wen-Bin; Shen, Chia-Ning; Hsiao, Michael; Hsu, Tsui-Ling; Chang, Chuan-Fa; Wong, Chi-Huey (2015-12-17). "Stage-specific embryonic antigen-3 (SSEA-3) and β3GalT5 are cancer specific and significant markers for breast cancer stem cells". Proceedings of the National Academy of Sciences. 113 (4): 960–965. doi:10.1073/pnas.1522602113. ISSN 0027-8424.
- ^ a b c d e Bilodeau, Mark T.; Park, Tae Kyo; Hu, Shuanghua; Randolph, John T.; Danishefsky, Samuel J.; Livingston, Philip O.; Zhang, Shengli (1995-07-01). "Total Synthesis of a Human Breast Tumor Associated Antigen". Journal of the American Chemical Society. 117 (29): 7840–7841. doi:10.1021/ja00134a043. ISSN 0002-7863.
- ^ Zhang, Shengle; Cordon-Cardo, Carlos; Zhang, Helen S.; Reuter, Victor E.; Adluri, Sucharita; Hamilton, Wm. Bradley; Lloyd, Kenneth O.; Livingston, Philip O. (1997-09-26). <42::aid-ijc8>3.0.co;2-1 "Selection of tumor antigens as targets for immune attack using immunohistochemistry: I. Focus on gangliosides". International Journal of Cancer. 73 (1): 42–49. doi:10.1002/(sici)1097-0215(19970926)73:1<42::aid-ijc8>3.0.co;2-1. ISSN 0020-7136.
- ^ Chen, I-Ju; Yang, Ming-Chen; Chen, Yu-Jung (2020-08-13). "Abstract 2946: The prevalence of Globo H in different tumor types: Breast, pancreatic, lung, gastric, colorectal, liver, and esophageal cancers". Experimental and Molecular Therapeutics. American Association for Cancer Research. doi:10.1158/1538-7445.am2020-2946.
- ^ a b Danishefsky, Samuel J.; Shue, Youe-Kong; Chang, Michael N.; Wong, Chi-Huey (2015-03-17). "Development of Globo-H Cancer Vaccine". Accounts of Chemical Research. 48 (3): 643–652. doi:10.1021/ar5004187. ISSN 0001-4842.
- ^ Rugo, Hope S.; Chow, Louis W. C.; Cortes, Javier; Fasching, Peter A.; Hsu, Pei; Huang, Chiun-Sheng; Kim, Sung-Bae; Lu, Yen-Shen; Melisko, Michelle E.; Nanda, Rita; Sharma, Priyanka (2020-05-20). "Phase III, randomized, double-blind, placebo-controlled study to evaluate the efficacy and safety of adagloxad simolenin (OBI-822) and OBI-821 treatment in patients with early-stage triple-negative breast cancer (TNBC) at high risk for recurrence". Journal of Clinical Oncology. 38 (15_suppl): TPS599–TPS599. doi:10.1200/jco.2020.38.15_suppl.tps599. ISSN 0732-183X.
- ^ Zhou, Zhifang; Liao, Guochao; Mandal, Satadru S.; Suryawanshi, Sharad; Guo, Zhongwu (2015). "A fully synthetic self-adjuvanting globo H-Based vaccine elicited strong T cell-mediated antitumor immunity". Chemical Science. 6 (12): 7112–7121. doi:10.1039/c5sc01402f. ISSN 2041-6520.
- ^ Ghosh, Samir; Trabbic, Kevin R.; Shi, Mengchao; Nishat, Sharmeen; Eradi, Pradheep; Kleski, Kristopher A.; Andreana, Peter R. (2020). "Chemical synthesis and immunological evaluation of entirely carbohydrate conjugate Globo H-PS A1". Chemical Science. 11 (48): 13052–13059. doi:10.1039/d0sc04595k. ISSN 2041-6520.
- ^ Chen, Yu-Chi; Yang, Ming-Chen; Shia, Chi-Sheng; Tsao, Chun-Yen; Lai, Jiann-Shiun; Chen, I-Ju (2019-07-01). "Abstract 4814: Specificity, biodistribution, tumor targeting, and pharmacokinetics of a novel humanized anti-Globo H antibody, OBI-888, for cancer immunotherapy". Experimental and Molecular Therapeutics. American Association for Cancer Research. doi:10.1158/1538-7445.am2019-4814.
- ^ Chen, Yu-Chi; Yang, Ming-Chen; Tsai, Yi-Chien; Chang, Hui-Wen; Hsieh, Chang-Lin; Chen, Yu-Jung; Lee, Kuang-Hsiu; Lai, Jiann-Shiun; Chen, I-Ju (2019-07-01). "Abstract 544: Anti-tumor efficacy and potential mechanism of action of a novel therapeutic humanized anti-Globo H antibody, OBI-888". Clinical Research (Excluding Clinical Trials). American Association for Cancer Research. doi:10.1158/1538-7445.am2019-544.
- ^ OBI Pharma, Inc (2020-08-04). "A Phase I/II, Open-Label, Dose Escalation and Cohort Expansion Study Evaluating the Safety, Pharmacokinetics (PK), Pharmacodynamics (PD), and Therapeutic Activity of OBI-888 in Patients With Locally Advanced or Metastatic Solid Tumors".
{{cite journal}}: Cite journal requires|journal=(help) - ^ Yang, Ming-Chen; Chen, Yu-Jung; Shia, Chi-Sheng; Chang, Hui-Wen; Li, Wan-Fen; Yu, Cheng-Der Tony; Chen, I-Ju (2019-07-01). "Abstract 4815: Novel Globo H targeting antibody-drug conjugate with binding specificity and anti-tumor efficacy in multiple cancer types". Experimental and Molecular Therapeutics. American Association for Cancer Research. doi:10.1158/1538-7445.am2019-4815.
- ^ Tsimberidou, Apostolia Maria; Ajani, Jaffer A.; Hsu, Pei; Chen, I-Ju; Pearce, Tillman E. (2020-05-20). "A phase I/II, open-label, dose-escalation, and cohort-expansion study evaluating the safety, pharmacokinetics, and therapeutic activity of OBI-999 in patients with advanced solid tumors". Journal of Clinical Oncology. 38 (15_suppl): TPS3657–TPS3657. doi:10.1200/jco.2020.38.15_suppl.tps3657. ISSN 0732-183X.
- ^ Inc, OBI Pharma. "OBI Pharma Granted U.S. FDA Orphan Drug Designation for the Treatment of Gastric Cancer for Its Antibody-Drug Conjugate (ADC) Targeted Cancer Therapy, OBI-999". www.prnewswire.com. Retrieved 2021-02-27.
{{cite web}}:|last=has generic name (help)

