Tröger's base

| |
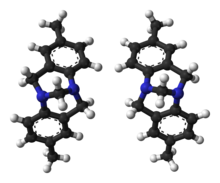 Ball-and-stick models of the two enantiomers of Tröger's base
| |
| Names | |
|---|---|
| Preferred IUPAC name
2,8-Dimethyl-6H,12H-5,11-methanodibenzo[b,f][1,5]diazocine | |
| Other names
Troeger's base
| |
| Identifiers | |
| |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.150.499 |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C17H18N2 | |
| Molar mass | 250.345 g·mol−1 |
| Melting point | 135–6 °C (275–43 °F; 408–279 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Tröger's base[1] is a white solid tetracyclic organic compound.[2] Its chemical formula is (CH
3C
6H
3NCH
2)
2CH
2. Tröger's base and its analogs are soluble in various organic solvents and strong acidic aqueous solutions due to their protonation. It is named after Julius Tröger, who first synthesized it in 1887.
History
[edit]Tröger's original research in 1887[1] failed to elaborate the exact structure of his new product, leading Johannes Wislicenus, the departmental director of the time, to assign a mediocre grade for Tröger's thesis. Though various possible structures had been drawn for Tröger's product, its correct structure remained as a mystery for 48 years, until the final elucidation in 1935 by Spielman.[3]
Structure and chirality
[edit]
The nitrogen inversion normally leads to a rapid equilibrium between the enantiomers of chiral amines, that prevents them showing any optical activity. The inversion can be stopped by conformational strain as Tröger's base has demonstrated that nitrogen is capable of forming a stereogenic center in organic molecules. In Tröger's base, this inversion is not possible, and the nitrogen atoms are defined stereogenic centers. The separation of the enantiomers of Tröger's base was first accomplished by Vladimir Prelog in 1944.[4] Prelog performed column chromatography using a chiral stationary phase as a relatively new method that later on gained popularity and became a standard procedure. Tröger's base and its analogs can be resolved by various methods including chiral HPLC[5][6] or be made as a single enantiomer.[7][8]
Almost 30 years after Tröger's initial report, Hünlich described another mysterious product obtained from the condensation of formaldehyde and 2,4-diaminotoluene.[9][10] After almost a century the structure of Hünlich's product was elucidated by X-ray crystallography as a C
2-symmetric amine-carrying analogue of Tröger's base.[11] Tröger's base is a diamine, which exceptionally exhibits chirality due to the prevented inversion of configuration of two bridgehead stereogenic tertiary amine groups. Tröger's base and its analogs racemize under acidic conditions through the formation of iminium intermediates,[12] which can be prevented by the replacement of methano-bridge with an ethano-bridge.[6]
The molecule can be considered a molecular tweezer as the skeleton forces the molecule in a rigid locked conformation with the aromatic rings in 90 degree proximity.[13][11]
Reactions
[edit]
Tröger's base and its analogs have attracted much academic interest.[14] They function as ligands and ligand precursors in coordination chemistry.[15] When the methyl groups are replaced by carboxylic acids[16] or pyridine amide groups a host–guest chemistry interaction can take place between the Tröger's base and other molecules including glycosaminoglycans.[17] It is found that the cavity dimensions are optimal for inclusion of suberic acid but that with a longer acid sebacic acid or a shorter acid adipic acid the interaction is less favorable.

Chromophore-carrying analogs of the Tröger's base[10][12] have displayed NLO properties[18] and can be used as molecular switches[11] and liquid crystal dopants.[6]
Spirocyclic analogues of Tröger's base (spiroTB) have been described.[19]
Synthesis
[edit]
Tröger's base is of historic interest as was first synthesised in 1887[1] from p-toluidine and formaldehyde in an acidic solution by Julius Tröger.[1] It can also be prepared with hydrochloric acid and dimethyl sulfoxide (DMSO)[20] or hexamethylene tetraamine (HMTA) as formaldehyde replacement.[21]
The reaction mechanism with DMSO as methylene donor for this reaction is similar to that of the Pummerer rearrangement. The interaction of DMSO and hydrochloric acid yields an electrophilic sulfenium ion that reacts with the aromatic amine in an electrophilic addition. Methanethiol is eliminated and the resulting imine reacts with a second amine. Sulfenium ion addition and elimination is repeated with the second amino group and the imine group reacts in an intramolecular electrophilic aromatic substitution reaction. Imine generation is repeated a third time and the reaction concludes with a second electrophilic substitution to the other aromat. Stereoselective, enantiospecific methods have also been introduced for the direct synthesis[22] of optically active analogs of Tröger's base.[6]
References
[edit]- ^ a b c d Julius Tröger (1887). "Ueber einige mittelst nascirenden Formaldehydes entstehende Basen". Journal für Praktische Chemie. 36 (1): 225–245. doi:10.1002/prac.18870360123.
- ^ Ostami (2017). "Facile preparation of Ʌ-shaped building blocks: Hünlich-base derivatization". Synlett. 28 (13): 1641–1645. doi:10.1055/s-0036-1588180. S2CID 99294625.
- ^ Spielman, M. A. (1935). "The Structure of Troeger's Base". J. Am. Chem. Soc. 57 (3): 583–585. doi:10.1021/ja01306a060.
- ^ Prelog, V.; Wieland, P. (1944). "Über die Spaltung der Tröger'schen Base in optische Antipoden, ein Beitrag zur Stereochemie des dreiwertigen Stickstoffs". Helvetica Chimica Acta. 27 (1): 1127–1134. doi:10.1002/hlca.194402701143.
- ^ Sergeyev Sergey, Diederich François (2006). "Semipreparative enantioseparation of Tröger base derivatives by HPLC". Chirality. 18 (9): 707–712. doi:10.1002/chir.20318. PMID 16845673.
- ^ a b c d e Kaztami (2019). "Optically active and photoswitchable Tröger's base analogs". New Journal of Chemistry. 43 (20): 7751–7755. doi:10.1039/C9NJ01372E. S2CID 164362391.
- ^ Takuya; et al. (2018). "Modular Synthesis of Optically Active Tröger's Base Analogues". ChemPlusChem. 78 (12): 1510–1516. doi:10.1002/cplu.201300295. PMID 31986657.
- ^ Lacour; et al. (2017). "Stereoselective and Enantiospecific Mono and Bis C-H azidation of Tröger Bases. Insight on Bridgehead Iminium Intermediates and Application to Anion-Binding Catalysis". Chemistry – A European Journal. 23 (36): 8678–8684. doi:10.1002/chem.201700845. PMID 28406541.
- ^ Stephan Rigol; Lothar Beyer; Lothar Hennig; Joachim Sieler; Athanassios Giannis (2013). "Hünlich Base: (Re)Discovery, Synthesis, and Structure Elucidation after a Century". Organic Letters. 15 (6): 1418–1420. doi:10.1021/ol400357t. PMID 23470133.
- ^ a b Kazem-Rostami, M. (2017). "Design and synthesis of Ʌ-shaped photoswitchable compounds employing Tröger's base scaffold". Synthesis. 49 (6): 1214–1222. doi:10.1055/s-0036-1588913. S2CID 99913657.
- ^ a b c d Novruz G. Akhmedov; et al. (2019). "Molecular lambda shape light-driven dual switches: spectroscopic and computational studies of the photoisomerization of bisazo Tröger base analogs". Journal of Molecular Structure. 1178: 538–543. Bibcode:2019JMoSt1178..538K. doi:10.1016/j.molstruc.2018.10.071. S2CID 105312344.
- ^ a b K. R. Masoud and A. Moghanian (2017). "Hunlich base derivatives as photo-responsive Ʌ-shaped hinges". Organic Chemistry Frontiers. 4 (2): 224–228. doi:10.1039/C6QO00653A.
- ^ Pardo, C; Sesmilo, E; Gutiérrez-Puebla, E; Monge, A; Elguero, J; Fruchier, A (2001). "New Chiral Molecular Tweezers with a Bis-Tröger's Base Skeleton". Journal of Organic Chemistry. 66 (5): 1607–1611. doi:10.1021/jo0010882. PMID 11262103.
- ^ Zdeněk Kejíka; Tomáš Bříza; Martin Havlík; Bohumil Dolenský; Robert Kaplánek; Jarmila Králová; Ivan Mikula; Pavel Martásek; Vladimír Král (2016). "Specific ligands based on Tröger's base derivatives for the recognition of glycosaminoglycans". Dyes and Pigments. 134: 212–218. doi:10.1016/j.dyepig.2016.07.002.
- ^ Shejwalkar, Pushkar; Sedinkin, Sergey L.; Bauer, Eike B. (2011). "New amino-dithiaphospholanes and phosphoramidodithioites and their rhodium and iridium complexes". Inorganica Chimica Acta. 366 (1): 209–218. doi:10.1016/j.ica.2010.11.006.
- ^ Adrian, J. C.; C. S. Wilcox (1989). "Chemistry of synthetic receptors and functional group arrays. 10. Orderly functional group dyads. Recognition of biotin and adenine derivatives by a new synthetic host". Journal of the American Chemical Society. 111 (20): 8055–8057. doi:10.1021/ja00202a078.
- ^ Goswami, S; Ghosh, K; Dasgupta, S (2000). "Troger's Base Molecular Scaffolds in Dicarboxylic Acid Recognition". Journal of Organic Chemistry. 65 (7): 1907–1914. doi:10.1021/jo9909204. PMID 10774008.
- ^ Sergey Sergeyev; Delphine Didier; Vitaly Boitsov; Ayele Teshome; Inge Asselberghs; Koen Clays; Christophe M. L. Vande Velde; Aurélie Plaquet; Benoît Champagne (2010). "Symmetrical and Nonsymmetrical Chromophores with Tröger's Base Skeleton: Chiroptical, Linear, and Quadratic Nonlinear Optical Properties—A Joint Theoretical and Experimental Study". Chemistry – A European Journal. 16 (27): 8181–8190. doi:10.1002/chem.201000216. PMID 20533454.
- ^ Navrátilová, Tereza; Tatar, Ameneh; Havlík, Martin; Hajduch, Jan; Drozdová, Michaela; Gurung, Kshitij; Palatinus, Lukáš; Čejka, Jan; Sedláček, Jakub; Anzenbacher, Pavel; Dolenský, Bohumil (2022-11-18). "Preparation and Characterization of Metalloporphyrin Tröger's and Spiro-Tröger's Base Derivatives". The Journal of Organic Chemistry. 87 (22): 15178–15186. doi:10.1021/acs.joc.2c01716. ISSN 0022-3263.
- ^ Li, Zhong; Xu, Xiaoyong; Peng, Yanqing; Jiang, Zhaoxing; Ding, Chuanyong; Qian, Xuhong (2005). "An Unusual Synthesis of Tröger's Bases Using DMSO/HCl as Formaldehyde Equivalent". Synthesis (8): 1228–30. doi:10.1055/s-2005-861868.
- ^ Masa, Thierry; Pardo, Carmen; Elguero, José (2004). "A shorter synthesis of symmetrical 2,11-dimethyl-bis-Tröger's bases. A new molecular tweezer". Arkivoc. (EM-973K).
- ^ Bosmani, Alessandro; Pujari, Sandip; Guenee, Laure; Besnard, Céline; Poblador Bahamonde, Amalia Isabel; Lacour, Jerome (2017). "Stereoselective and Enantiospecific Mono and Bis C-H azidation of Tröger Bases. Insight on Bridgehead Iminium Intermediates and Application to Anion-Binding Catalysis". Chemistry – A European Journal. 23 (36): 8678–8684. doi:10.1002/chem.201700845. PMID 28406541.
