TAF4
Transcription initiation factor TFIID subunit 4 is a protein that in humans is encoded by the TAF4 gene.[5][6][7]
Function
[edit]Initiation of transcription by RNA polymerase II requires the activities of more than 70 polypeptides. The protein that coordinates these activities is transcription factor IID (TFIID), which binds to the core promoter to position the polymerase properly, serves as the scaffold for assembly of the remainder of the transcription complex, and acts as a channel for regulatory signals. TFIID is composed of the TATA-binding protein (TBP) and a group of evolutionarily conserved proteins known as TBP-associated factors or TAFs. TAFs may participate in basal transcription, serve as coactivators, function in promoter recognition or modify general transcription factors (GTFs) to facilitate complex assembly and transcription initiation. This gene encodes one of the larger subunits of TFIID that has been shown to potentiate transcriptional activation by retinoic acid, thyroid hormone and vitamin D3 receptors. In addition, this subunit interacts with the transcription factor CREB, which has a glutamine-rich activation domain, and binds to other proteins containing glutamine-rich regions. Aberrant binding to this subunit by proteins with expanded polyglutamine regions has been suggested as one of the pathogenetic mechanisms underlying a group of neurodegenerative disorders referred to as polyglutamine diseases.[7]
Interactions
[edit]TAF4 has been shown to interact with:
Protein domain
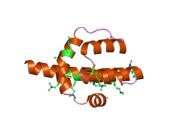
[edit]| TAF4 | |||||||||
|---|---|---|---|---|---|---|---|---|---|
 crystal structure of the human taf4-taf12 (tafii135-tafii20) complex | |||||||||
| Identifiers | |||||||||
| Symbol | TAF4 | ||||||||
| Pfam | PF05236 | ||||||||
| InterPro | IPR007900 | ||||||||
| SCOP2 | 1h3o / SCOPe / SUPFAM | ||||||||
| |||||||||
Yeast TFIID comprises the TATA binding protein and 14 TBP-associated factors (TAFIIs), nine of which contain histone-fold domains (INTERPRO). The C-terminal region of the TFIID-specific yeast TAF4 (yTAF4) containing the HFD shares strong sequence similarity with Drosophila (d)TAF4 and human TAF4. A structure/function analysis of yTAF4 demonstrates that the HFD, a short conserved C-terminal domain (CCTD), and the region separating them are all required for yTAF4 function. This region of similarity is found in Transcription initiation factor TFIID component TAF4.[12]
References
[edit]- ^ a b c GRCh38: Ensembl release 89: ENSG00000130699 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000039117 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Tanese N, Saluja D, Vassallo MF, Chen JL, Admon A (1996). "Molecular cloning and analysis of two subunits of the human TFIID complex: hTAFII130 and hTAFII100". Proc. Natl. Acad. Sci. U.S.A. 93 (24): 13611–6. Bibcode:1996PNAS...9313611T. doi:10.1073/pnas.93.24.13611. PMC 19367. PMID 8942982.
- ^ Mengus G, May M, Carré L, Chambon P, Davidson I (July 1997). "Human TAF(II)135 potentiates transcriptional activation by the AF-2s of the retinoic acid, vitamin D3, and thyroid hormone receptors in mammalian cells". Genes Dev. 11 (11): 1381–95. doi:10.1101/gad.11.11.1381. PMID 9192867.
- ^ a b "Entrez Gene: TAF4 TAF4 RNA polymerase II, TATA box binding protein (TBP)-associated factor, 135kDa".
- ^ Vassallo MF, Tanese N (April 2002). "Isoform-specific interaction of HP1 with human TAFII130". Proc. Natl. Acad. Sci. U.S.A. 99 (9): 5919–24. Bibcode:2002PNAS...99.5919V. doi:10.1073/pnas.092025499. PMC 122877. PMID 11959914.
- ^ Pointud JC, Mengus G, Brancorsini S, Monaco L, Parvinen M, Sassone-Corsi P, Davidson I (May 2003). "The intracellular localisation of TAF7L, a paralogue of transcription factor TFIID subunit TAF7, is developmentally regulated during male germ-cell differentiation". J. Cell Sci. 116 (Pt 9): 1847–58. doi:10.1242/jcs.00391. PMID 12665565.
- ^ Bellorini M, Lee DK, Dantonel JC, Zemzoumi K, Roeder RG, Tora L, Mantovani R (June 1997). "CCAAT binding NF-Y-TBP interactions: NF-YB and NF-YC require short domains adjacent to their histone fold motifs for association with TBP basic residues". Nucleic Acids Res. 25 (11): 2174–81. doi:10.1093/nar/25.11.2174. PMC 146709. PMID 9153318.
- ^ Brand M, Moggs JG, Oulad-Abdelghani M, Lejeune F, Dilworth FJ, Stevenin J, Almouzni G, Tora L (June 2001). "UV-damaged DNA-binding protein in the TFTC complex links DNA damage recognition to nucleosome acetylation". EMBO J. 20 (12): 3187–96. doi:10.1093/emboj/20.12.3187. PMC 150203. PMID 11406595.
- ^ Thuault S, Gangloff YG, Kirchner J, Sanders S, Werten S, Romier C, Weil PA, Davidson I (November 2002). "Functional analysis of the TFIID-specific yeast TAF4 (yTAF(II)48) reveals an unexpected organization of its histone-fold domain". J. Biol. Chem. 277 (47): 45510–7. doi:10.1074/jbc.M206556200. PMID 12237303.
Further reading
[edit]- Zhou Q, Sharp PA (1995). "Novel mechanism and factor for regulation by HIV-1 Tat". EMBO J. 14 (2): 321–8. doi:10.1002/j.1460-2075.1995.tb07006.x. PMC 398086. PMID 7835343.
- Parada CA, Yoon JB, Roeder RG (1995). "A novel LBP-1-mediated restriction of HIV-1 transcription at the level of elongation in vitro". J. Biol. Chem. 270 (5): 2274–83. doi:10.1074/jbc.270.5.2274. PMID 7836461.
- Ou SH, Garcia-Martínez LF, Paulssen EJ, Gaynor RB (1994). "Role of flanking E box motifs in human immunodeficiency virus type 1 TATA element function". J. Virol. 68 (11): 7188–99. doi:10.1128/JVI.68.11.7188-7199.1994. PMC 237158. PMID 7933101.
- Kashanchi F, Piras G, Radonovich MF, Duvall JF, Fattaey A, Chiang CM, Roeder RG, Brady JN (1994). "Direct interaction of human TFIID with the HIV-1 transactivator tat". Nature. 367 (6460): 295–9. Bibcode:1994Natur.367..295K. doi:10.1038/367295a0. PMID 8121496. S2CID 4362048.
- Wang Z, Morris GF, Rice AP, Xiong W, Morris CB (1996). "Wild-type and transactivation-defective mutants of human immunodeficiency virus type 1 Tat protein bind human TATA-binding protein in vitro". J. Acquir. Immune Defic. Syndr. Hum. Retrovirol. 12 (2): 128–38. doi:10.1097/00042560-199606010-00005. PMID 8680883.
- Pendergrast PS, Morrison D, Tansey WP, Hernandez N (1996). "Mutations in the carboxy-terminal domain of TBP affect the synthesis of human immunodeficiency virus type 1 full-length and short transcripts similarly". J. Virol. 70 (8): 5025–34. doi:10.1128/JVI.70.8.5025-5034.1996. PMC 190456. PMID 8764009.
- Kashanchi F, Khleif SN, Duvall JF, Sadaie MR, Radonovich MF, Cho M, Martin MA, Chen SY, Weinmann R, Brady JN (1996). "Interaction of human immunodeficiency virus type 1 Tat with a unique site of TFIID inhibits negative cofactor Dr1 and stabilizes the TFIID-TFIIA complex". J. Virol. 70 (8): 5503–10. doi:10.1128/JVI.70.8.5503-5510.1996. PMC 190508. PMID 8764062.
- Zhou Q, Sharp PA (1996). "Tat-SF1: cofactor for stimulation of transcriptional elongation by HIV-1 Tat". Science. 274 (5287): 605–10. Bibcode:1996Sci...274..605Z. doi:10.1126/science.274.5287.605. PMID 8849451. S2CID 13266489.
- García-Martínez LF, Ivanov D, Gaynor RB (1997). "Association of Tat with purified HIV-1 and HIV-2 transcription preinitiation complexes". J. Biol. Chem. 272 (11): 6951–8. doi:10.1074/jbc.272.11.6951. PMID 9054383.
- Saluja D, Vassallo MF, Tanese N (1998). "Distinct subdomains of human TAFII130 are required for interactions with glutamine-rich transcriptional activators". Mol. Cell. Biol. 18 (10): 5734–43. doi:10.1128/mcb.18.10.5734. PMC 109159. PMID 9742090.
- Brand M, Yamamoto K, Staub A, Tora L (1999). "Identification of TATA-binding protein-free TAFII-containing complex subunits suggests a role in nucleosome acetylation and signal transduction". J. Biol. Chem. 274 (26): 18285–9. doi:10.1074/jbc.274.26.18285. PMID 10373431.
- Inada A, Someya Y, Yamada Y, Ihara Y, Kubota A, Ban N, Watanabe R, Tsuda K, Seino Y (1999). "The cyclic AMP response element modulator family regulates the insulin gene transcription by interacting with transcription factor IID". J. Biol. Chem. 274 (30): 21095–103. doi:10.1074/jbc.274.30.21095. PMID 10409662.
- Gangloff YG, Werten S, Romier C, Carré L, Poch O, Moras D, Davidson I (2000). "The human TFIID components TAF(II)135 and TAF(II)20 and the yeast SAGA components ADA1 and TAF(II)68 heterodimerize to form histone-like pairs". Mol. Cell. Biol. 20 (1): 340–51. doi:10.1128/MCB.20.1.340-351.2000. PMC 85089. PMID 10594036.
- Martinez E, Palhan VB, Tjernberg A, Lymar ES, Gamper AM, Kundu TK, Chait BT, Roeder RG (2001). "Human STAGA complex is a chromatin-acetylating transcription coactivator that interacts with pre-mRNA splicing and DNA damage-binding factors in vivo". Mol. Cell. Biol. 21 (20): 6782–95. doi:10.1128/MCB.21.20.6782-6795.2001. PMC 99856. PMID 11564863.
- Guermah M, Tao Y, Roeder RG (2001). "Positive and negative TAF(II) functions that suggest a dynamic TFIID structure and elicit synergy with traps in activator-induced transcription". Mol. Cell. Biol. 21 (20): 6882–94. doi:10.1128/MCB.21.20.6882-6894.2001. PMC 99865. PMID 11564872.
- Felinski EA, Quinn PG (2001). "The coactivator dTAF(II)110/hTAF(II)135 is sufficient to recruit a polymerase complex and activate basal transcription mediated by CREB". Proc. Natl. Acad. Sci. U.S.A. 98 (23): 13078–83. doi:10.1073/pnas.241337698. PMC 60827. PMID 11687654.