Palladium(II) iodide
| |

| |
| Identifiers | |
|---|---|
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.029.276 |
| EC Number |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| I2Pd | |
| Molar mass | 360.229 g/mol |
| Appearance | Black crystals |
| Density | 6,003 g/cm3 |
| Melting point | 350 °C (decomposes) |
| Insoluble in water | |
| Hazards | |
| GHS labelling:[1] | |

| |
| Warning | |
| H315, H319, H335 | |
| Related compounds | |
Other anions
|
Palladium(II) fluoride Palladium(II) chloride Palladium(II) bromide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Palladium(II) iodide is an inorganic compound of palladium and iodine. It is commercially available, though less common than palladium(II) chloride, the usual entry point to palladium chemistry. Three polymorphs are known.[2]
Preparation
[edit]Palladium(II) iodide can be obtained by treating a dilute solution of palladium in nitric acid with sodium iodide at 80 °C.[2]
The high-temperature polymorph α-palladium(II) iodide can be produced by reaction of the elements at temperature above 600 °C. The γ-modification is produced as an almost amorphous powder by addition of iodide salts to aqueous H2PdCl4 solution . When heated in dilute hydrogen iodide solution, this polymorph transforms into the β phase at around 140 °C.[3]
Reactions and uses
[edit]Palladium(II) iodide is insoluble in water. It reacts with iodide giving PdI42− anion:
- PdI2 + 2I− → PdI2−4
It finds use as a catalyst.[4]
Historically, the quantity of palladium in a solution may be determined gravimetrically by precipitation as palladium(II) iodide.[5]
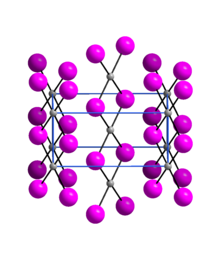
Crystallography
[edit]Palladium(II) iodide is an almost X-ray amorphous black powder. The α-modification has an orthorhombic crystal structure with the space group Pnmn(space group no. 58, position 5).[6]
References
[edit]- ^ "C&L Inventory". echa.europa.eu. Retrieved 13 December 2021.
- ^ a b Handbuch der präparativen anorganischen Chemie. 3 (3., umgearb. Aufl ed.). Stuttgart: Enke. 1981. ISBN 978-3-432-87823-2.
- ^ Brendel, Kristin; Thiele, Gerhard (2001). "Binäre und Ternäre Verbindungen der Platinmetalle Palladium und Rhodium mit Tellur und Halogenen. Präparationen und strukturelle Charakterisierung".
{{cite journal}}: Cite journal requires|journal=(help) - ^ Gabriele, Bartolo; Salerno, Giuseppe (2006), "Palladium(II) Iodide", Encyclopedia of Reagents for Organic Synthesis, American Cancer Society, doi:10.1002/047084289x.rn00658, ISBN 978-0-470-84289-8, retrieved 2021-03-26
- ^ Beamish, F. E.; Dale, J. (1938). "Determination of Palladium by Means of Potassium Iodide". Industrial & Engineering Chemistry Analytical Edition. 10 (12): 697. doi:10.1021/ac50128a015.
- ^ Ans, Jean d'; Lax, Ellen (1998). Taschenbuch für Chemiker und Physiker (in German). Springer. ISBN 978-3-540-60035-0.
