Indium(III) iodide
| |
| Names | |
|---|---|
| Other names
Indium triiodide
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.033.475 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| InI3 | |
| Molar mass | 495.53 g/mol |
| Appearance | Yellow solid |
| Density | 4.69 g/cm3 |
| Melting point | 210 °C (410 °F; 483 K) |
| Boiling point | 500 °C (932 °F; 773 K) |
| Related compounds | |
Other anions
|
Indium(III) bromide Indium(III) chloride |
Other cations
|
Aluminum iodide Gallium(III) iodide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Indium(III) iodide or indium triiodide is a chemical compound of indium and iodine with the formula InI3.
Preparation
[edit]Indium(III) iodide can be obtained by reacting indium with iodine vapor:[1]
- 2 In + 3I2 → 2 InI3
Indium(III) iodide can also be obtained by evaporation of a solution of indium in HI.[2]
Properties
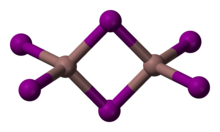
[edit]Indium(III) iodide is a pale yellow, very hygroscopic monoclinic solid (space group P21/c (space group no. 14), a = 9.837 Å, b = 6.102 Å, c = 12.195 Å, β = 107.69°),[3] which melts at 210 °C to form a dark brown liquid and is highly soluble in water. Its crystals consist of dimeric molecules.[4] The yellow β form slowly converts to the red α form.[5] In the presence of water vapor, the compound reacts with oxygen at 245 °C to form indium(III) oxide iodide.[6]
Distinct yellow and red forms are known. The red form undergoes a transition to the yellow at 57 °C. The structure of the red form has not been determined by X-ray crystallography; however, spectroscopic evidence indicates that indium may be six coordinate.[7] The yellow form consists of In2I6 with 4 coordinate indium centres.
References
[edit]- ^ Handbuch der präparativen anorganischen Chemie. 1 (3., umgearb. Aufl ed.). Stuttgart: Enke. 1975. ISBN 978-3-432-02328-1.
- ^ E. Donges (1963). "Indium(III) Iodide". In G. Brauer (ed.). Handbook of Preparative Inorganic Chemistry, 2nd Ed. Vol. 1. NY, NY: Academic Press. pp. 861–2.
- ^ Forrester, J. D.; Zalkin, Allan; Templeton, David H. (Jan 1964). "Crystal and Molecular Structure of Indium(III) Iodide (In 2 I 6 )". Inorganic Chemistry. 3 (1): 63–67. doi:10.1021/ic50011a013. ISSN 0020-1669.
- ^ Handbuch der präparativen anorganischen Chemie. 1 (3., umgearb. Aufl ed.). Stuttgart: Enke. 1975. ISBN 978-3-432-02328-1.
- ^ Downs, Anthony John (1993). Chemistry of aluminium, gallium, indium and thallium. London Glasgow New York [etc.]: Blackie. ISBN 978-0-7514-0103-5.
- ^ Hagen, A. P. (2009-09-17). Inorganic Reactions and Methods, The Formation of Bonds to Group VIB (O, S, Se, Te, Po) Elements (Part 1). John Wiley & Sons. ISBN 978-0-470-14540-1.
- ^ Taylor M. J., Kloo L. A. Journal of Raman Spectroscopy 31, 6, (2000), 465
