Palladium
 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Palladium | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pronunciation | /pəˈleɪdiəm/ | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Appearance | silvery white | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Standard atomic weight Ar°(Pd) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Palladium in the periodic table | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic number (Z) | 46 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Group | group 10 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Period | period 5 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Block | d-block | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electron configuration | [Kr] 4d10 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electrons per shell | 2, 8, 18, 18 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical properties | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Phase at STP | solid | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Melting point | 1828.05 K (1554.9 °C, 2830.82 °F) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Boiling point | 3236 K (2963 °C, 5365 °F) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density (at 20° C) | 12.007 g/cm3[3] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| when liquid (at m.p.) | 10.38 g/cm3 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Heat of fusion | 16.74 kJ/mol | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Heat of vaporization | 358 kJ/mol | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molar heat capacity | 25.98 J/(mol·K) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Vapor pressure
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic properties | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Oxidation states | common: 0, +2, +4 +1,[4] +3,[5] +5[6] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electronegativity | Pauling scale: 2.20 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ionization energies |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic radius | empirical: 137 pm | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Covalent radius | 139±6 pm | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Van der Waals radius | 163 pm | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other properties | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Natural occurrence | primordial | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Crystal structure | face-centered cubic (fcc) (cF4) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lattice constant | a = 389.02 pm (at 20 °C)[3] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Thermal expansion | 11.77×10−6/K (at 20 °C)[3] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Thermal conductivity | 71.8 W/(m⋅K) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electrical resistivity | 105.4 nΩ⋅m (at 20 °C) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Magnetic ordering | paramagnetic[7] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molar magnetic susceptibility | +567.4×10−6 cm3/mol (288 K)[8] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Young's modulus | 121 GPa | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Shear modulus | 44 GPa | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Bulk modulus | 180 GPa | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Speed of sound thin rod | 3070 m/s (at 20 °C) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Poisson ratio | 0.39 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mohs hardness | 4.75 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Vickers hardness | 400–600 MPa | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Brinell hardness | 320–610 MPa | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS Number | 7440-05-3 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| History | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Naming | after asteroid Pallas, itself named after Pallas Athena | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Discovery and first isolation | William Hyde Wollaston (1802) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Isotopes of palladium | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Palladium is a chemical element; it has symbol Pd and atomic number 46. It is a rare and lustrous silvery-white metal discovered in 1802 by the English chemist William Hyde Wollaston. He named it after the asteroid Pallas (formally 2 Pallas), which was itself named after the epithet of the Greek goddess Athena, acquired by her when she slew Pallas. Palladium, platinum, rhodium, ruthenium, iridium and osmium form a group of elements referred to as the platinum group metals (PGMs). They have similar chemical properties, but palladium has the lowest melting point and is the least dense of them.
More than half the supply of palladium and its congener platinum is used in catalytic converters, which convert as much as 90% of the harmful gases in automobile exhaust (hydrocarbons, carbon monoxide, and nitrogen dioxide) into nontoxic substances (nitrogen, carbon dioxide and water vapor). Palladium is also used in electronics, dentistry, medicine, hydrogen purification, chemical applications, groundwater treatment, and jewelry. Palladium is a key component of fuel cells, in which hydrogen and oxygen react to produce electricity, heat, and water.
Ore deposits of palladium and other PGMs are rare. The most extensive deposits have been found in the norite belt of the Bushveld Igneous Complex covering the Transvaal Basin in South Africa; the Stillwater Complex in Montana, United States; the Sudbury Basin and Thunder Bay District of Ontario, Canada; and the Norilsk Complex in Russia. Recycling is also a source, mostly from scrapped catalytic converters. The numerous applications and limited supply sources result in considerable investment interest.
Characteristics
[edit]Palladium belongs to group 10 in the periodic table, but the configuration in the outermost electrons is in accordance with Hund's rule. Electrons that by the Madelung rule would be expected to occupy the 5s instead fill the 4d orbitals, as it is more energetically favorable to have a completely filled 4d10 shell instead of the 5s2 4d8 configuration.[clarification needed]
| Z | Element | No. of electrons/shell |
|---|---|---|
| 28 | nickel | 2, 8, 16, 2 (or 2, 8, 17, 1) |
| 46 | palladium | 2, 8, 18, 18, 0 |
| 78 | platinum | 2, 8, 18, 32, 17, 1 |
| 110 | darmstadtium | 2, 8, 18, 32, 32, 16, 2 (predicted)[10] |
This 5s0 configuration, unique in period 5, makes palladium the heaviest element having only one incomplete electron shell, with all shells above it empty.
Palladium has the appearance of a soft silver-white metal that resembles platinum. It is the least dense and has the lowest melting point of the platinum group metals. It is soft and ductile when annealed and is greatly increased in strength and hardness when cold-worked. Palladium dissolves slowly in concentrated nitric acid, in hot, concentrated sulfuric acid, and when finely ground, in hydrochloric acid.[11] It dissolves readily at room temperature in aqua regia.
Palladium does not react with oxygen at standard temperature (and thus does not tarnish in air). Palladium heated to 800 °C will produce a layer of palladium(II) oxide (PdO). It may slowly develop a slight brownish coloration over time, likely due to the formation of a surface layer of its monoxide.
Palladium films with defects produced by alpha particle bombardment at low temperature exhibit superconductivity having Tc = 3.2 K.[12]
Isotopes
[edit]Naturally occurring palladium is composed of seven isotopes, six of which are stable. The most stable radioisotopes are 107Pd with a half-life of 6.5 million years (found in nature), 103Pd with 17 days, and 100Pd with 3.63 days. Eighteen other radioisotopes have been characterized with atomic weights ranging from 90.94948(64) u (91Pd) to 122.93426(64) u (123Pd).[13] These have half-lives of less than thirty minutes, except 101Pd (half-life: 8.47 hours), 109Pd (half-life: 13.7 hours), and 112Pd (half-life: 21 hours).[14]
For isotopes with atomic mass unit values less than that of the most abundant stable isotope, 106Pd, the primary decay mode is electron capture with the primary decay product being rhodium. The primary mode of decay for those isotopes of Pd with atomic mass greater than 106 is beta decay with the primary product of this decay being silver.[14]
Radiogenic 107Ag is a decay product of 107Pd and was first discovered in 1978[15] in the Santa Clara[16] meteorite of 1976. The discoverers suggest that the coalescence and differentiation of iron-cored small planets may have occurred 10 million years after a nucleosynthetic event. 107Pd versus Ag correlations observed in bodies, which have been melted since accretion of the Solar System, must reflect the presence of short-lived nuclides in the early Solar System.[17] 107
Pd is also produced as a fission product in spontaneous or induced fission of 235
U. As it is not very mobile in the environment and has a relatively low decay energy, 107
Pd is usually considered to be among the less concerning of the long-lived fission products.
Compounds
[edit]Palladium compounds exist primarily in the 0 and +2 oxidation state. Other less common states are also recognized. Generally the compounds of palladium are more similar to those of platinum than those of any other element.
-
Structure of α-PdCl2
-
Structure of β-PdCl2
Palladium(II)
[edit]Palladium(II) chloride is the principal starting material for other palladium compounds. It arises by the reaction of palladium with chlorine. It is used to prepare heterogeneous palladium catalysts such as palladium on barium sulfate, palladium on carbon, and palladium chloride on carbon.[18] Solutions of PdCl2 in nitric acid react with acetic acid to give palladium(II) acetate, also a versatile reagent. PdCl2 reacts with ligands (L) to give square planar complexes of the type PdCl2L2. One example of such complexes is the benzonitrile derivative PdCl2(PhCN)2.[19][20]
The complex bis(triphenylphosphine)palladium(II) dichloride is a useful catalyst.[21]



Palladium(0)
[edit]Palladium forms a range of zerovalent complexes with the formula PdL4, PdL3 and PdL2. For example, reduction of a mixture of PdCl2(PPh3)2 and PPh3 gives tetrakis(triphenylphosphine)palladium(0):[22]
Another major palladium(0) complex, tris(dibenzylideneacetone)dipalladium(0) (Pd2(dba)3), is prepared by reducing sodium tetrachloropalladate in the presence of dibenzylideneacetone.[23]
Palladium(0), as well as palladium(II), are catalysts in coupling reactions, as has been recognized by the 2010 Nobel Prize in Chemistry to Richard F. Heck, Ei-ichi Negishi, and Akira Suzuki. Such reactions are widely practiced for the synthesis of fine chemicals. Prominent coupling reactions include the Heck, Suzuki, Sonogashira coupling, Stille reactions, and the Kumada coupling. Palladium(II) acetate, tetrakis(triphenylphosphine)palladium(0) (Pd(PPh3)4), and tris(dibenzylideneacetone)dipalladium(0) (Pd2(dba)3) serve either as catalysts or precatalysts.[24]
Other oxidation states
[edit]Although Pd(IV) compounds are comparatively rare, one example is sodium hexachloropalladate(IV), Na2[PdCl6]. A few compounds of palladium(III) are also known.[25] Palladium(VI) was claimed in 2002,[26][27] but subsequently disproven.[28][29]
Mixed valence palladium complexes exist, e.g. Pd4(CO)4(OAc)4Pd(acac)2 forms an infinite Pd chain structure, with alternatively interconnected Pd4(CO)4(OAc)4 and Pd(acac)2 units.[30]
When alloyed with a more electropositive element, palladium can acquire a negative charge. Such compounds are known as palladides, such as gallium palladide.[31] Palladides with the stoichiometry RPd3 exist where R is scandium, yttrium, or any of the lanthanides.[32]
Occurrence
[edit]
As overall mine production of palladium reached 210,000 kilograms in 2022, Russia was the top producer with 88,000 kilograms, followed by South Africa, Canada, the U.S., and Zimbabwe.[33] Russia's company Norilsk Nickel ranks first among the largest palladium producers globally, accounting for 39% of the world's production.[34]
Palladium can be found as a free metal alloyed with gold and other platinum-group metals in placer deposits of the Ural Mountains, Australia, Ethiopia, North and South America. For the production of palladium, these deposits play only a minor role. The most important commercial sources are nickel-copper deposits found in the Sudbury Basin, Ontario, and the Norilsk–Talnakh deposits in Siberia. The other large deposit is the Merensky Reef platinum group metals deposit within the Bushveld Igneous Complex South Africa. The Stillwater igneous complex of Montana and the Roby zone ore body of the Lac des Îles igneous complex of Ontario are the two other sources of palladium in Canada and the United States.[35][36] Palladium is found in the rare minerals cooperite[37] and polarite.[38] Many more Pd minerals are known, but all of them are very rare.[39]
Palladium is also produced in nuclear fission reactors and can be extracted from spent nuclear fuel (see synthesis of precious metals), though this source for palladium is not used. None of the existing nuclear reprocessing facilities are equipped to extract palladium from the high-level radioactive waste.[40] A complication for the recovery of palladium in spent fuel is the presence of 107
Pd, a slightly radioactive long-lived fission product. Depending on end use, the radioactivity contributed by the 107
Pd might make the recovered palladium unusable without a costly step of isotope separation.
Applications
[edit]

The largest use of palladium today is in catalytic converters.[41] Palladium is also used in jewelry, dentistry,[41][42] watch making, blood sugar test strips, aircraft spark plugs, surgical instruments, and electrical contacts.[43] Palladium is also used to make some professional transverse (concert or classical) flutes.[44] As a commodity, palladium bullion has ISO currency codes of XPD and 964. Palladium is one of only four metals to have such codes, the others being gold, silver and platinum.[45] Because it adsorbs hydrogen, palladium was a key component of the controversial cold fusion experiments of the late 1980s.[46]
Catalysis
[edit]When it is finely divided, as with palladium on carbon, palladium forms a versatile catalyst; it speeds heterogeneous catalytic processes like hydrogenation, dehydrogenation, and petroleum cracking. Palladium is also essential to the Lindlar catalyst, also called Lindlar's Palladium.[47] A large number of carbon–carbon bonding reactions in organic chemistry are facilitated by palladium compound catalysts. For example:
- Heck reaction
- Suzuki coupling
- Tsuji-Trost reactions
- Wacker process
- Negishi reaction
- Stille coupling
- Sonogashira coupling
When dispersed on conductive materials, palladium is an excellent electrocatalyst for oxidation of primary alcohols in alkaline media.[48] Palladium is also a versatile metal for homogeneous catalysis, used in combination with a broad variety of ligands for highly selective chemical transformations.
In 2010 the Nobel Prize in Chemistry was awarded "for palladium-catalyzed cross couplings in organic synthesis" to Richard F. Heck, Ei-ichi Negishi and Akira Suzuki. A 2008 study showed that palladium is an effective catalyst for carbon–fluorine bonds.[49]
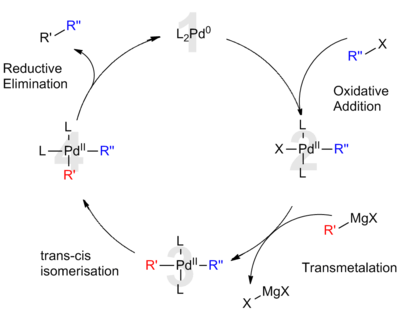
Palladium catalysis is primarily employed in organic chemistry and industrial applications, although its use is growing as a tool for synthetic biology; in 2017, effective in vivo catalytic activity of palladium nanoparticles was demonstrated in mammals to treat disease.[50]
Electronics
[edit]The primary application of palladium in electronics is in multi-layer ceramic capacitors[51] in which palladium (and palladium-silver alloy) is used for electrodes.[41] Palladium (sometimes alloyed with nickel) is or can be used for component and connector plating in consumer electronics[52][53] and in soldering materials. The electronic sector consumed 33 tonnes (1.07 million troy ounces) of palladium in 2006, according to a Johnson Matthey report.[54]
Technology
[edit]Hydrogen easily diffuses through heated palladium,[11] and membrane reactors with Pd membranes are used in the production of high purity hydrogen.[55] Palladium is used in palladium-hydrogen electrodes in electrochemical studies. Palladium(II) chloride readily catalyzes carbon monoxide gas to carbon dioxide and is useful in carbon monoxide detectors.[56]
Hydrogen storage
[edit]Palladium readily adsorbs hydrogen at room temperatures, forming palladium hydride PdHx with x less than 1.[57] While this property is common to many transition metals, palladium has a uniquely high absorption capacity and does not lose its ductility until x approaches 1.[58] This property has been investigated in designing an efficient and safe hydrogen fuel storage medium, though palladium itself is currently prohibitively expensive for this purpose.[59] The content of hydrogen in palladium can be linked to magnetic susceptibility, which decreases with the increase of hydrogen and becomes zero for PdH0.62. At any higher ratio, the solid solution becomes diamagnetic.[60]
Palladium is used for purification of hydrogen on a laboratory[61]: 183–217 but not industrial scale.[62]
Dentistry
[edit]Palladium is used in small amounts (about 0.5%) in some alloys of dental amalgam to decrease corrosion and increase the metallic lustre of the final restoration.[63][64]
Jewelry
[edit]Palladium has been used as a precious metal in jewelry since 1939 as an alternative to platinum in the alloys called "white gold", where the naturally white color of palladium does not require rhodium plating. Palladium, being much less dense than platinum, is similar to gold in that it can be beaten into leaf as thin as 100 nm (1⁄250,000 in).[11] Unlike platinum, palladium may discolor at temperatures above 400 °C (752 °F)[65] due to oxidation, making it more brittle and thus less suitable for use in jewelry; to prevent this, palladium intended for jewelry is heated under controlled conditions.[66]
Prior to 2004, the principal use of palladium in jewelry was the manufacture of white gold. Palladium is one of the three most popular alloying metals in white gold (nickel and silver can also be used).[41] Palladium-gold is more expensive than nickel-gold, but seldom causes allergic reactions (though certain cross-allergies with nickel may occur).[67]
When platinum became a strategic resource during World War II, many jewelry bands were made out of palladium. Palladium was little used in jewelry because of the technical difficulty of casting. With the casting problem resolved[68] the use of palladium in jewelry increased, originally because platinum increased in price while the price of palladium decreased.[69] In early 2004, when gold and platinum prices rose steeply, China began fabricating volumes of palladium jewelry, consuming 37 tonnes in 2005. Subsequent changes in the relative price of platinum lowered demand for palladium to 17.4 tonnes in 2009.[70][71] Demand for palladium as a catalyst has increased the price of palladium to about 50% higher than that of platinum in January 2019.[72]
In January 2010, hallmarks for palladium were introduced by assay offices in the United Kingdom, and hallmarking became mandatory for all jewelry advertising pure or alloyed palladium. Articles can be marked as 500, 950, or 999 parts of palladium per thousand of the alloy.
Fountain pen nibs made from gold are sometimes plated with palladium when a silver (rather than gold) appearance is desired. Sheaffer has used palladium plating for decades, either as an accent on otherwise gold nibs or covering the gold completely.
Palladium is also used by the luxury brand Hermès as one of the metals plating the hardware on their handbags, most famous of which being Birkin.
Photography
[edit]In the platinotype printing process, photographers make fine-art black-and-white prints using platinum or palladium salts. Often used with platinum, palladium provides an alternative to silver.[73]
Effects on health
[edit]Toxicity
[edit]| Hazards | |
|---|---|
| GHS labelling: | |

| |
| Warning | |
| H317 | |
| P261, P273, P280, P302+P352, P321, P333+P313, P363, P501[74] | |
| NFPA 704 (fire diamond) | |
Palladium is a metal with low toxicity as conventionally measured (e.g. LD50). Recent research on the mechanism of palladium toxicity suggests high toxicity if measured on a longer timeframe and at the cellular level in the liver and kidney.[75] Mitochondria appear to have a key role in palladium toxicity via mitochondrial membrane potential collapse and depletion of the cellular glutathione (GSH) level. Until that recent work, it had been thought that palladium was poorly absorbed by the human body when ingested. Plants such as the water hyacinth are killed by low levels of palladium salts, but most other plants tolerate it, although tests show that, at levels above 0.0003%, growth is affected. High doses of palladium could be poisonous; tests on rodents suggest it may be carcinogenic, though until the recent research cited above, no clear evidence indicated that the element harms humans.[76]
Precautions
[edit]Like other platinum-group metals, bulk Pd is quite inert. Although contact dermatitis has been reported, data on the effects are limited. It has been shown that people with an allergic reaction to palladium also react to nickel, making it advisable to avoid the use of dental alloys containing palladium on those so allergic.[77][78][79][80][81]
Some palladium is emitted with the exhaust gases of cars with catalytic converters. Between 4 and 108 ng/km of palladium particulate is released by such cars, while the total uptake from food is estimated to be less than 2 μg per person a day. The second possible source of palladium is dental restoration, from which the uptake of palladium is estimated to be less than 15 μg per person per day. People working with palladium or its compounds might have a considerably greater uptake. For soluble compounds such as palladium chloride, 99% is eliminated from the body within three days.[77]
The median lethal dose (LD50) of soluble palladium compounds in mice is 200 mg/kg for oral and 5 mg/kg for intravenous administration.[77]
History
[edit]

William Hyde Wollaston noted the discovery of a new noble metal in July 1802 in his lab book and named it palladium in August of the same year. He named the element after the asteroid 2 Pallas, which had been discovered two months earlier (and which was previously considered a planet).[11] Wollaston purified a quantity of the material and offered it, without naming the discoverer, in a small shop in Soho in April 1803. After harsh criticism from Richard Chenevix, who claimed that palladium was an alloy of platinum and mercury, Wollaston anonymously offered a reward of £20 for 20 grains of synthetic palladium alloy.[82] Chenevix received the Copley Medal in 1803 after he published his experiments on palladium. Wollaston published the discovery of rhodium in 1804 and mentions some of his work on palladium.[83][84] He disclosed that he was the discoverer of palladium in a publication in 1805.[82][85]
Wollaston found palladium in crude platinum ore from South America by dissolving the ore in aqua regia, neutralizing the solution with sodium hydroxide, and precipitating platinum as ammonium chloroplatinate with ammonium chloride. He added mercuric cyanide to form the compound palladium(II) cyanide, which was heated to extract palladium metal.[83]
Palladium chloride was at one time prescribed as a tuberculosis treatment at the rate of 0.065 g per day (approximately one milligram per kilogram of body weight). This treatment had many negative side-effects, and was later replaced by more effective drugs.[86]
Most palladium is used for catalytic converters in the automobile industry.[77] Catalytic converters are targets for thieves because they contain palladium and other rare metals. In the run up to year 2000, the Russian supply of palladium to the global market was repeatedly delayed and disrupted; for political reasons, the export quota was not granted on time.[87] The ensuing market panic drove the price to an all-time high of $1,340 per troy ounce ($43/g) in January 2001.[88] Around that time, the Ford Motor Company, fearing that automobile production would be disrupted by a palladium shortage, stockpiled the metal. When prices fell in early 2001, Ford lost nearly US$1 billion.[89]
World demand for palladium increased from 100 tons in 1990 to nearly 300 tons in 2000. The global production of palladium from mines was 222 tonnes in 2006 according to the United States Geological Survey.[35] Many were concerned about a steady supply of palladium in the wake of Russia's annexation of Crimea, partly as sanctions could hamper Russian palladium exports; any restrictions on Russian palladium exports could have exacerbated what was already expected to be a large palladium deficit in 2014.[90] Those concerns pushed palladium prices to their highest level since 2001.[91] In September 2014 they soared above the $900 per ounce mark. In 2016 however palladium cost around $614 per ounce as Russia managed to maintain stable supplies.[92] In January 2019 palladium futures climbed past $1,344 per ounce for the first time on record, mainly due to the strong demand from the automotive industry.[93] Palladium reached $2,024.64 per troy ounce ($65.094/g) on 6 January 2020, passing $2,000 per troy ounce the first time.[94] The price rose above $3,000 per troy ounce in May 2021 and March 2022.[95]
Palladium as investment
[edit]
Global palladium sales were 8.84 million troy ounces (275 t) in 2017,[96] of which 86% was used in the manufacturing of automotive catalytic converters, followed by industrial, jewelry, and investment usages.[97] More than 75% of global platinum and 40% of palladium are mined in South Africa. Russia's mining company, Norilsk Nickel, produces another 44% of palladium, with US and Canada-based mines producing most of the rest.
The price for palladium reached an all-time high of $2,981.40 per ounce on May 3, 2021,[98][99] driven mainly on speculation of the catalytic converter demand from the automobile industry. Palladium is traded in the spot market with the code "XPD". When settled in USD, the code is "XPDUSD". A later surplus of the metal was caused by the Russian government selling stockpiles from the Soviet era, at a rate of about 1.6 to 2 million troy ounces (50 to 62 t) a year. The amount and status of this stockpile are a state secret.
During the Russo-Ukrainian War in March 2022, prices for palladium increased 13%, since the first of March. Russia is the primary supplier to Europe and the country supplies 37% of the global production.[100]
Palladium producers
[edit]Exchange-traded products
[edit]WisdomTree Physical Palladium (LSE: PHPD) is backed by allocated palladium bullion and was the world's first palladium ETF. It is listed on the London Stock Exchange as PHPD,[101] Xetra Trading System, Euronext and Milan. ETFS Physical Palladium Shares (NYSE: PALL) is an ETF traded on the New York Stock Exchange.
Bullion coins and bars
[edit]A traditional way of investing in palladium is buying bullion coins and bars made of palladium. Available palladium coins include the Canadian Palladium Maple Leaf, the Chinese Panda, and the American Palladium Eagle. The liquidity of direct palladium bullion investment is poorer than that of gold and silver because there is low circulation of palladium coins.[102]
See also
[edit]- 2000s commodities boom
- 2020s commodities boom
- Bullion
- Bullion coin
- Inflation hedge
- Pseudo palladium
- Rare materials as an investment:
References
[edit]- ^ "Standard Atomic Weights: Palladium". CIAAW. 1979.
- ^ Prohaska, Thomas; Irrgeher, Johanna; Benefield, Jacqueline; Böhlke, John K.; Chesson, Lesley A.; Coplen, Tyler B.; Ding, Tiping; Dunn, Philip J. H.; Gröning, Manfred; Holden, Norman E.; Meijer, Harro A. J. (4 May 2022). "Standard atomic weights of the elements 2021 (IUPAC Technical Report)". Pure and Applied Chemistry. doi:10.1515/pac-2019-0603. ISSN 1365-3075.
- ^ a b c Arblaster, John W. (2018). Selected Values of the Crystallographic Properties of Elements. Materials Park, Ohio: ASM International. ISBN 978-1-62708-155-9.
- ^ Pd(I) is known in [Pd2]2+ compounds; see Christoph Fricke; Theresa Sperger; Marvin Mendel; Franziska Schoenebeck (2020). "Catalysis with Palladium(I) Dimers". Angewandte Chemie International Edition. 60 (7). doi:10.1002/anie.202011825.
- ^ Pd(III) has been observed; see Powers, D. C.; Ritter, T. (2011). "Palladium(III) in Synthesis and Catalysis" (PDF). Higher Oxidation State Organopalladium and Platinum Chemistry. Topics in Organometallic Chemistry. Vol. 35. pp. 129–156. Bibcode:2011hoso.book..129P. doi:10.1007/978-3-642-17429-2_6. ISBN 978-3-642-17428-5. PMC 3066514. PMID 21461129. Archived from the original (PDF) on 12 June 2013.
- ^ Palladium(V) has been identified in complexes with organosilicon compounds containing pentacoordinate palladium; see Shimada, Shigeru; Li, Yong-Hua; Choe, Yoong-Kee; Tanaka, Masato; Bao, Ming; Uchimaru, Tadafumi (2007). "Multinuclear palladium compounds containing palladium centers ligated by five silicon atoms". Proceedings of the National Academy of Sciences. 104 (19): 7758–7763. doi:10.1073/pnas.0700450104. PMC 1876520. PMID 17470819.
- ^ Lide, D. R., ed. (2005). "Magnetic susceptibility of the elements and inorganic compounds". CRC Handbook of Chemistry and Physics (PDF) (86th ed.). Boca Raton (FL): CRC Press. ISBN 0-8493-0486-5.
- ^ Weast, Robert (1984). CRC, Handbook of Chemistry and Physics. Boca Raton, Florida: Chemical Rubber Company Publishing. pp. E110. ISBN 0-8493-0464-4.
- ^ Kondev, F. G.; Wang, M.; Huang, W. J.; Naimi, S.; Audi, G. (2021). "The NUBASE2020 evaluation of nuclear properties" (PDF). Chinese Physics C. 45 (3): 030001. doi:10.1088/1674-1137/abddae.
- ^ Hoffman, Darleane C.; Lee, Diana M.; Pershina, Valeria (2006). "Transactinides and the future elements". In Morss; Edelstein, Norman M.; Fuger, Jean (eds.). The Chemistry of the Actinide and Transactinide Elements (3rd ed.). Dordrecht, The Netherlands: Springer Science+Business Media. p. 1722. ISBN 1-4020-3555-1.
- ^ a b c d Hammond, C. R. (2004). "The Elements". Handbook of Chemistry and Physics (81st ed.). CRC press. ISBN 978-0-8493-0485-9.
- ^ B. Strizker, Phys. Rev. Lett., 42, 1769 (1979).
- ^ "Atomic Weights and Isotopic Compositions for Palladium (NIST)". NIST. 23 August 2009. Retrieved 12 November 2009.
- ^ a b Audi, Georges; Bersillon, Olivier; Blachot, Jean; Wapstra, Aaldert Hendrik (2003), "The NUBASE evaluation of nuclear and decay properties", Nuclear Physics A, 729: 3–128, Bibcode:2003NuPhA.729....3A, doi:10.1016/j.nuclphysa.2003.11.001
- ^ Kelly, W. R.; Gounelle, G. J.; Hutchison, R. (1978). "Evidence for the existence of 107Pd in the early solar system". Philosophical Transactions of the Royal Society of London, Series A. 359 (1787): 1079–1082. Bibcode:2001RSPTA.359.1991R. doi:10.1098/rsta.2001.0893. S2CID 120355895.
- ^ "Mexico's Meteorites" (PDF). mexicogemstones.com. Archived from the original (PDF) on 6 May 2006.
- ^ Chen, J. H.; Wasserburg, G. J. (1990). "The isotopic composition of Ag in meteorites and the presence of 107Pd in protoplanets". Geochimica et Cosmochimica Acta. 54 (6): 1729–1743. Bibcode:1990GeCoA..54.1729C. doi:10.1016/0016-7037(90)90404-9.
- ^ Mozingo, Ralph (1955). "Palladium Catalysts". Organic Syntheses; Collected Volumes, vol. 3, p. 685.
- ^ Anderson, Gordon K.; Lin, Minren; Sen, Ayusman; Gretz, Efi (1990). "Bis(Benzonitrile)Dichloro Complexes of Palladium and Platinum". Inorganic Syntheses. Vol. 28. pp. 60–63. doi:10.1002/9780470132593.ch13. ISBN 978-0-470-13259-3.
- ^ Zalevskaya, O. A; Vorob'eva, E. G; Dvornikova, I. A; Kuchin, A. V (2008). "Palladium complexes based on optically active terpene derivatives of ethylenediamine". Russian Journal of Coordination Chemistry. 34 (11): 855–857. doi:10.1134/S1070328408110110. S2CID 95529734.
- ^ Miyaura, Norio & Suzuki, Akira (1993). "Palladium-catalyzed reaction of 1-alkenylboronates with vinylic halides: (1Z,3E)-1-Phenyl-1,3-octadiene". Organic Syntheses; Collected Volumes, vol. 8, p. 532.
- ^ Coulson, D. R.; Satek, L. C.; Grim, S. O. (1972). "Tetrakis(triphenylphosphine)palladium(0)". Inorganic Syntheses. 13: 121–124. doi:10.1002/9780470132449.ch23. ISBN 978-0-470-13244-9.
- ^ Takahashi, Y; Ito, Ts; Sakai, S; Ishii, Y (1970). "A novel palladium(0) complex; bis(dibenzylideneacetone)palladium(0)". Journal of the Chemical Society D: Chemical Communications (17): 1065. doi:10.1039/C29700001065.
- ^ Crabtree, Robert H. (2009). "Application to Organic Synthesis". The Organometallic Chemistry of the Transition Metals. John Wiley and Sons. p. 392. ISBN 978-0-470-25762-3.
- ^ Powers, David C; Ritter, Tobias (2011). "Palladium(III) in Synthesis and Catalysis". Higher Oxidation State Organopalladium and Platinum Chemistry. Topics in Organometallic Chemistry. Vol. 35. pp. 129–156. doi:10.1007/978-3-642-17429-2_6. ISBN 978-3-642-17428-5. PMC 3066514. PMID 21461129.
- ^ Chen, W; Shimada, S; Tanaka, M (2002). "Synthesis and Structure of Formally Hexavalent Palladium Complexes". Science. 295 (5553): 308–310. Bibcode:2002Sci...295..308C. doi:10.1126/science.1067027. PMID 11786638. S2CID 45249108.
- ^ Crabtree, R. H (2002). "CHEMISTRY: A New Oxidation State for Pd?". Science. 295 (5553): 288–289. doi:10.1126/science.1067921. PMID 11786632. S2CID 94579227.
- ^ Aullón, G; Lledós, A; Alvarez, S (2002). "Hexakis(silyl)palladium(VI) or palladium(II with eta2-disilane ligands?". Angewandte Chemie International Edition in English. 41 (11): 1956–9. doi:10.1002/1521-3773(20020603)41:11<1956::AID-ANIE1956>3.0.CO;2-#. PMID 19750645.
- ^ Sherer, E. C; Kinsinger, C. R; Kormos, B. L; Thompson, J. D; Cramer, C. J (2002). "Electronic structure and bonding in hexacoordinate silyl-palladium complexes". Angewandte Chemie International Edition in English. 41 (11): 1953–6. doi:10.1002/1521-3773(20020603)41:11<1953::AID-ANIE1953>3.0.CO;2-H. PMID 19750644.
- ^ Yin, Xi; Warren, Steven A; Pan, Yung-Tin; Tsao, Kai-Chieh; Gray, Danielle L; Bertke, Jeffery; Yang, Hong (2014). "A Motif for Infinite Metal Atom Wires". Angewandte Chemie International Edition. 53 (51): 14087–14091. doi:10.1002/anie.201408461. PMID 25319757.
- ^ Armbrüster, Marc (31 January 2020). "Intermetallic compounds in catalysis – a versatile class of materials meets interesting challenges". Science and Technology of Advanced Materials. 21 (1). Informa UK Limited: 303–322. Bibcode:2020STAdM..21..303A. doi:10.1080/14686996.2020.1758544. ISSN 1468-6996. PMC 7889166. PMID 33628119.
- ^ Wang, Qiaoming; Collins, Gary S. (2013). "Nuclear quadrupole interactions of 111In/Cd solute atoms in a series of rare-earth palladium alloys". Hyperfine Interactions. 221 (1–3): 85–98. arXiv:1209.3822. Bibcode:2013HyInt.221...85W. doi:10.1007/s10751-012-0686-4. ISSN 0304-3843. S2CID 98580013.
- ^ U. S. Geological Survey (2023). Mineral commodity summaries 2023 (Report). p. 210. doi:10.3133/mcs2023.
- ^ ""Norilsk Nickel" Group announces preliminary consolidated production results for 4 th quarter and full 2016, and production outl". Nornickel. Archived from the original on 29 June 2018. Retrieved 29 January 2018.
- ^ a b "Platinum-Group Metals" (PDF). Mineral Commodity Summaries. United States Geological Survey. January 2007.
- ^ "Platinum-Group Metals" (PDF). Mineral Yearbook 2007. United States Geological Survey. January 2007.
- ^ Verryn, Sabine M. C.; Merkle, Roland K. W. (1994). "Compositional variation of cooperite, braggite, and vysotskite from the Bushveld Complex". Mineralogical Magazine. 58 (2): 223–234. Bibcode:1994MinM...58..223V. CiteSeerX 10.1.1.610.640. doi:10.1180/minmag.1994.058.391.05. S2CID 53128786.
- ^ Genkin, A. D.; Evstigneeva, T. L. (1986). "Associations of platinum- group minerals of the Norilsk copper-nickel sulfide ores". Economic Geology. 81 (5): 1203–1212. Bibcode:1986EcGeo..81.1203G. doi:10.2113/gsecongeo.81.5.1203.
- ^ "Mindat.org - Mines, Minerals and More". www.mindat.org.
- ^ Kolarik, Zdenek; Renard, Edouard V. (2003). "Recovery of Value Fission Platinoids from Spent Nuclear Fuel. Part I PART I: General Considerations and Basic Chemistry" (PDF). Platinum Metals Review. 47 (2): 74–87. doi:10.1595/003214003X4727487.
- ^ a b c d "Palladium". United Nations Conference on Trade and Development. Archived from the original on 6 December 2006. Retrieved 5 February 2007.
- ^ Rushforth, Roy (2004). "Palladium in Restorative Dentistry: Superior Physical Properties make Palladium an Ideal Dental Metal". Platinum Metals Review. 48 (1). doi:10.1595/003214004X4813031.
- ^ Hesse, Rayner W. (2007). "palladium". Jewelry-making through history: an encyclopedia. Greenwood Publishing Group. p. 146. ISBN 978-0-313-33507-5.
- ^ Toff, Nancy (1996). The flute book: a complete guide for students and performers. Oxford University Press. p. 20. ISBN 978-0-19-510502-5.
- ^ Weithers, Timothy Martin (2006). "Precious Metals". Foreign exchange: a practical guide to the FX markets. Wiley. p. 34. ISBN 978-0-471-73203-7.
- ^ Fleischmann, M; Pons S; Hawkins M (1989). "Electrochemically induced nuclear fusion of deuterium". J. Electroanal. Chem. 261 (2): 301. doi:10.1016/0022-0728(89)80006-3.
- ^ Brown, William Henry; Foote, Christopher S; Iverson, Brent L (2009). "Catalytic reduction". Organic chemistry. Cengage Learning. p. 270. ISBN 978-0-495-38857-9.
- ^ Tsuji, Jiro (2004). Palladium reagents and catalysts: new perspectives for the 21st century. John Wiley and Sons. p. 90. ISBN 978-0-470-85032-9.
- ^ Drahl, Carmen (2008). "Palladium's Hidden Talent". Chemical & Engineering News. 86 (35): 53–56. doi:10.1021/cen-v086n035.p053.
- ^ Miller, Miles A; Askevold, Bjorn; Mikula, Hannes; Kohler, Rainer H; Pirovich, David; Weissleder, Ralph (2017). "Nano-palladium is a cellular catalyst for in vivo chemistry". Nature Communications. 8: 15906. Bibcode:2017NatCo...815906M. doi:10.1038/ncomms15906. PMC 5510178. PMID 28699627.
- ^ Zogbi, Dennis (3 February 2003). "Shifting Supply and Demand for Palladium in MLCCs". TTI, Inc.
- ^ Mroczkowski, Robert S. (1998). Electronic connector handbook: theory and applications. McGraw-Hill Professional. pp. 3–. ISBN 978-0-07-041401-3.
- ^ Harper, Charles A. (1997). Passive electronic component handbook. McGraw-Hill Professional. pp. 580–. ISBN 978-0-07-026698-8.
- ^ Jollie, David (2007). "Platinum 2007" (PDF). Johnson Matthey. Archived from the original (PDF) on 16 February 2008.
- ^ Shu, J.; Grandjean, B. P. A.; Neste, A. Van; Kaliaguine, S. (1991). "Catalytic palladium-based membrane reactors: A review". The Canadian Journal of Chemical Engineering. 69 (5): 1036. doi:10.1002/cjce.5450690503.
- ^ Allen, T. H.; Root, W. S. (1955). "An improved palladium chloride method for the determination of carbon monoxide in blood". The Journal of Biological Chemistry. 216 (1): 319–323. doi:10.1016/S0021-9258(19)52308-0. PMID 13252031.
- ^ Manchester, F. D.; San-Martin, A.; Pitre, J. M. (1994). "The H-Pd (hydrogen-palladium) System". Journal of Phase Equilibria. 15: 62–83. doi:10.1007/BF02667685. S2CID 95343702.
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. pp. 1150–151. ISBN 978-0-08-037941-8.
- ^ Grochala, Wojciech; Edwards, Peter P. (2004). "Thermal Decomposition of the Non-Interstitial Hydrides for the Storage and Production of Hydrogen". Chemical Reviews. 104 (3): 1283–316. doi:10.1021/cr030691s. PMID 15008624.
- ^ Mott, N. F. and Jones, H. (1958) The Theory of Properties of metals and alloys. Oxford University Press. ISBN 0-486-60456-X. p. 200
- ^ Angelo Basile, ed. (2013). Handbook of membrane reactors Vol. 1, Fundamental materials science, design and optimisation. Cambridge, UK: Woodhead Publishing. ISBN 978-0-85709-414-8. OCLC 870962388.
- ^ Häussinger, Peter; Lohmüller, Reiner; Watson, Allan M. (2011). "Hydrogen, 3. Purification". Ullmann's Encyclopedia of Industrial Chemistry. doi:10.1002/14356007.o13_o04. ISBN 978-3-527-30385-4.
- ^ Colon, Pierre; Pradelle-Plasse, Nelly; Galland, Jacques (2003). "Evaluation of the long-term corrosion behavior of dental amalgams: influence of palladium addition and particle morphology". Dental Materials. 19 (3): 232–9. doi:10.1016/S0109-5641(02)00035-0. PMID 12628436.
- ^ Sakaguchi, Ronald; Ferracane, Jack; Powers, John, eds. (1 January 2019), "Chapter 10 - Restorative Materials: Metals", Craig's Restorative Dental Materials (Fourteenth Edition), Philadelphia: Elsevier, pp. 171–208, doi:10.1016/B978-0-323-47821-2.00010-X, ISBN 978-0-323-47821-2, retrieved 11 February 2023
- ^ Gupta, Dinesh C.; Langer, Paul H.; ASTM Committee F-1 on Electronics (1987). Emerging semiconductor technology: a symposium. ASTM International. pp. 273–. ISBN 978-0-8031-0459-4.
- ^ Mann, Mark B. (2007). "950 Palladium: Manufacturing Methods".
- ^ Hindsen, M.; Spiren, A.; Bruze, M. (2005). "Cross-reactivity between nickel and palladium demonstrated by systemic administration of nickel". Contact Dermatitis. 53 (1): 2–8. doi:10.1111/j.0105-1873.2005.00577.x. PMID 15982224. S2CID 20927683.
- ^ Battaini, Paolo (2006). "The Working Properties for Jewelry Fabrication Using New Hard 950 Palladium Alloys". SANTA FE SYMPOSIUM PAPERS.
- ^ Holmes, E. (13 February 2007). "Palladium, Platinum's Cheaper Sister, Makes a Bid for Love". The Wall Street Journal (Eastern edition). pp. B.1.
- ^ "Platinum-Group Metals" (PDF). Mineral Yearbook 2009. United States Geological Survey. January 2007.
- ^ "Platinum-Group Metals" (PDF). Mineral Yearbook 2006. United States Geological Survey. January 2007.
- ^ "Johnson Matthey Base Prices". 2019. Retrieved 7 January 2019.
- ^ Ware, Mike (2005). "Book Review of : Photography in Platinum and Palladium". Platinum Metals Review. 49 (4): 190–195. doi:10.1595/147106705X70291.
- ^ Sigma-Aldrich Co., SDS Palladium.
- ^ Hosseini et al, Metallomics, 2016,8, 252–259; doi:10.1039/C5MT00249D
- ^ Emsley, John (2011). Nature's Building Blocks: An A-Z Guide to the Elements. Oxford University Press. pp. 384, 387. ISBN 978-0-19-960563-7.
- ^ a b c d Kielhorn, Janet; Melber, Christine; Keller, Detlef; Mangelsdorf, Inge (2002). "Palladium – A review of exposure and effects to human health". International Journal of Hygiene and Environmental Health. 205 (6): 417–32. Bibcode:2002IJHEH.205..417K. doi:10.1078/1438-4639-00180. PMID 12455264.
- ^ Zereini, Fathi; Alt, Friedrich (2006). "Health Risk Potential of Palladium". Palladium emissions in the environment: analytical methods, environmental assessment and health effects. Springer Science & Business. pp. 549–563. ISBN 978-3-540-29219-7.
- ^ Wataha, J. C.; Hanks, C. T. (1996). "Biological effects of palladium and risk of using palladium in dental casting alloys". Journal of Oral Rehabilitation. 23 (5): 309–20. doi:10.1111/j.1365-2842.1996.tb00858.x. PMID 8736443.
- ^ Aberer, Werner; Holub, Henriette; Strohal, Robert; Slavicek, Rudolf (1993). "Palladium in dental alloys – the dermatologists' responsibility to warn?". Contact Dermatitis. 28 (3): 163–5. doi:10.1111/j.1600-0536.1993.tb03379.x. PMID 8462294. S2CID 43020912.
- ^ Wataha, John C.; Shor, Kavita (2010). "Palladium alloys for biomedical devices". Expert Review of Medical Devices. 7 (4): 489–501. doi:10.1586/erd.10.25. PMID 20583886. S2CID 41325428.
- ^ a b Usselman, Melvyn (1978). "The Wollaston/Chenevix controversy over the elemental nature of palladium: A curious episode in the history of chemistry". Annals of Science. 35 (6): 551–579. doi:10.1080/00033797800200431.
- ^ a b Griffith, W. P. (2003). "Rhodium and Palladium – Events Surrounding Its Discovery". Platinum Metals Review. 47 (4): 175–183. doi:10.1595/003214003X474175183.
- ^ Wollaston, W. H. (1804). "On a New Metal, Found in Crude Platina". Philosophical Transactions of the Royal Society of London. 94: 419–430. doi:10.1098/rstl.1804.0019.
- ^ Wollaston, W. H. (1805). "On the Discovery of Palladium; With Observations on Other Substances Found with Platina". Philosophical Transactions of the Royal Society of London. 95: 316–330. doi:10.1098/rstl.1805.0024.
- ^ Garrett, Christine E.; Prasad, Kapa (2004). "The Art of Meeting Palladium Specifications in Active Pharmaceutical Ingredients Produced by Pd-Catalyzed Reactions". Advanced Synthesis & Catalysis. 346 (8): 889–900. doi:10.1002/adsc.200404071. S2CID 94929244.
- ^ Williamson, Alan. "Russian PGM Stocks" (PDF). The LBMA Precious Metals Conference 2003. The London Bullion Market Association. Archived from the original (PDF) on 21 October 2013. Retrieved 2 October 2010.
- ^ "Historical Palladium Prices and Price Chart". InvestmentMine. Retrieved 27 January 2015.
- ^ "Ford fears first loss in a decade". BBC News. 16 January 2002. Retrieved 19 September 2008.
- ^ Nat Rudarakanchana (27 March 2014). "Palladium Fund Launches in South Africa, As Russian Supply Fears Warm Prices". International Business Times.
- ^ Rosenfeld, Everett (20 August 2014). "The other commodity that's leaping on Ukraine war". CNBC. Retrieved 29 January 2018.
- ^ "Palladium Rally Is About More Than Just Autos". Bloomberg.com. 30 August 2017. Retrieved 29 January 2018.
- ^ "Don't Expect Palladium Prices To Plunge | OilPrice.com". OilPrice.com. Retrieved 29 January 2018.
- ^ "Gold soars as Middle East tensions brew perfect storm". Reuters. 6 January 2020. Retrieved 6 January 2020.
- ^ Patel, Brijesh (4 March 2022). "Palladium tops $3,000/oz as supply fears grow, gold jumps over 1%". Reuters.
- ^ "Total palladium supply worldwide 2017 | Statistic". Statista. Retrieved 15 October 2018.
- ^ "Global palladium demand distribution by application 2016 | Statistic". Statista. Retrieved 15 October 2018.
- ^ "Historical Palladium Charts and Data - London Fix".
- ^ "CPI Inflation Calculator". data.bls.gov. Retrieved 13 August 2018.
- ^ Staff, Writer (OilPrice.com) (10 March 2022). "Palladium Prices Are Soaring As Russian Sanctions Sting". Yahoo! Finance. OilPrice.com. Retrieved 13 March 2022.
- ^ "ETFS METAL PAL ETP price (PHPD)". London Stock Exchange.
- ^ "Size of the Palladium Market | Sunshine Profits". www.sunshineprofits.com. Retrieved 11 February 2023.
External links
[edit]- Palladium at The Periodic Table of Videos (University of Nottingham)
- Current and Historical Palladium Price
- . Encyclopædia Britannica. Vol. 20 (11th ed.). 1911. pp. 636–637.





