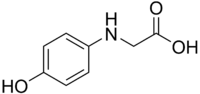
Glycin
| |
| Names | |
|---|---|
| IUPAC name
N-(4-Hydroxyphenyl)glycine
| |
| Systematic IUPAC name
(4-Hydroxyanilino)acetic acid | |
| Other names
p-hydroxyanilinoacetic acid
photoglycine | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.004.165 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C8H9NO3 | |
| Molar mass | 167.16 g/mol |
| Appearance | brown powder |
| Density | 1.411 g/mL |
| Melting point | 244 °C (471 °F; 517 K) |
| Boiling point | 446.3 °C (835.3 °F; 719.5 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Glycin, or N-(4-hydroxyphenyl)glycine, is N-substituted p-aminophenol. It is a photographic developing agent used in classic black-and-white developer solutions.[2] It is not identical to, but derived from glycine, the proteinogenic amino acid. It is typically characterized as thin plates of white or silvery powder, although aged samples appear brown. It is sparingly soluble in water and most organic solvents; it is readily soluble in alkalies and acids.[3]
Glycin is structurally related to 4-aminophenol and metol. Decarboxylation of glycin gives metol. Glycin has a milder reduction potential than metol. The two developers have markedly different character. Glycin is slower-acting, but much longer-lasting in solution. Glycin is rarely used as a developing agent, primarily because it is expensive. In its dry form, it also has limited shelf life compared to metol and phenidone.
Glycin can be synthesized by treating p-aminophenol with chloracetic acid in a solvent.
Glycin is employed in some procedures of analytical chemistry.
References
[edit]- ^ Merck Index, 11th Edition, 4771.
- ^ Photographic Chemical Descriptions
- ^ Mitchell, Stephen C.; Waring, Rosemary H. (2000). "Aminophenols". Ullmann's Encyclopedia of Industrial Chemistry. Wiley-VCH. doi:10.1002/14356007.a02_099. ISBN 978-3527306732.
