Germylene

Germylenes are a class of germanium(II) compounds with the general formula :GeR2. They are heavier carbene analogs.[1] However, unlike carbenes, whose ground state can be either singlet or triplet depending on the substituents, germylenes have exclusively a singlet ground state.[2][3] Unprotected carbene analogs, including germylenes, has a dimerization nature.[4] Free germylenes can be isolated under the stabilization of steric hindrance or electron donation.[5] The synthesis of first stable free dialkyl germylene was reported by Jutzi, et al in 1991.[6]
Structures and bonding
[edit]Bonding situation for germylene is distinctively different from that for its light analog carbene. The carbon atom from carbene is sp2 hybridized. Although germylenes still have some sp2 hybridization character, the larger energy gap between s and p-orbitals for germanium permits the retainment of 4s24p2 electron configuration to some degree. The bond angle for H2Ge and Me2Ge was found to be: H-Ge-H 93° and C-Ge-C: 98°, which is smaller than 120°, the ideal bond angle for sp2 hybridized structure and thus proves the 4s24p2 valence electron configuration nature of germylene.[1][7][8] The lone pair of germylene tends to stay in the high-s-character orbital which is relatively inert, making germylene exclusively singlet.[3]
Dimerization of germylenes lead to the formation of germylene dimers (R2Ge=GeR2).[9] Digermylene dimers (as well as the higher-order digermynes) have a trans-bent structure quasi-tetrahedral at each germanium atom. Consequently the dimerization is believed to proceed through two donor-acceptor adducts instead of the triplet double-pairing found during carbene dimerization.[3]

Synthesis
[edit]Stabilization
[edit]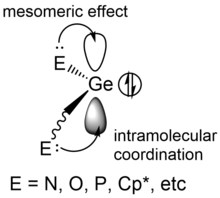
Dimerization of free germylenes does not have a noticeable energy barrier, which means that the dimerization reaction is almost spontaneous and diffusion limited, so the free germylene monomers without stabilization could dimerize or further polymerize once they form.[4] Free germylenes have to be stabilized kinetically or thermodynamically due to their high reactivity originating from the vacant p-orbital. Thermodynamical stabilization of this p-orbital is usually realized by coordination of a pentamethylcyclopentadiene (Cp*) ligand or of nitrogen (N), oxygen (O) or phosphorus (P) containing ligands, which are able to donate electrons and thus deactivate the vacant p-orbitals.[1] At the same time, stabilization can be accomplished by steric protection of bulky R groups like mesityl groups (Mes) to prevent nucleophiles from getting close to the germanium center.[10]
Synthesis of carbon substituted germylenes
[edit]Carbon substituents is different from other heteroatom N, O, P substituents which have lone pairs in that they provide less electronic perturbations.[11] As a result, a stronger steric and electronic stabilization is required to guarantee a monomeric product. Carbon substituted germylenes can be synthesized using various methods: (1) reduction of dibromogermanes with reducing agents like lithium naphthalene (LiNp) or potassium graphite (KC8), etc., (2) photolysis of strained cyclogermanes or Ge(IV) species, (3) substitution of a dihalo Ge(II) precursor species with nucleophiles like organometallic ligands (e.g. RLi/RMgBr).[1]

Synthesis of n-heterocyclic germylene and cyclic(alkyl)(amino)germylene
[edit]The introduction of heteroatom in the ligand backbone enhances the stability of reactive Ge(II) center by electron donation from N lone pair to vacant p-orbitals of germanium center. Typically, the strategy for synthesizing five-membered N-heterocyclic tetrylene involves the reaction between N-substituted 1,4-diaza-1,3-butadiene, the alkali metal based reducing agents and group 14 halides.[12] In the case of n-heterocyclic germylene (NHGe) synthesis, the method involves an initial reduction of N-substituted 1,4-diaza-1,3-butadiene by lithium. The following cyclization of the dianion with the corresponding Ge(II) halides gives the final product.[12]

The cyclic(alkyl)(amino)carbenes (CAACs) has already been known as both a better donor and better acceptor than n-heterocyclic carbenes (NHCs) due to its higher highest occupied molecular orbital (HOMO) and lower lowest unoccupied molecular orbital (LUMO).[13]
The synthetic strategy of CAAGe involves the synthesis of a α-β-unsaturated imine from a ketone and an amine via condensation followed by the treatment with GeCl2·dioxane. The resulting product is then reduced with KC8 to give CAAGe.[14] Analogous to CAAC, the electrophilicity of the germanium center can be obviously enhanced by the substitution of a π-donating and σ-withdrawing amino group along with σ-donating trimethylsilyl groups.[11]

Synthesis of a unique homoconjugation stabilized germylene
[edit]In 2016, Muller et al reported the synthesis of a unique homoconjugation stabilized germylene in a relatively high yield by the reaction between hafnocene dichloride and dipotassium germacyclopentadienediide in THF at -80 °C.[15][16] The product is stabilized by a remote interaction between a C=C double bond and vacant p-orbital of Ge center through homoconjugation.[17] This stabilization strategy results in a special structural which possesses unusual reactivity.[11]

Synthesis of PGeP pincer compounds
[edit]The pincer based germylene is of great importance not only for their ability to stabilize transition metal species via chelation effects in homogeneous catalysis, but also for its serving as a good luminescence source.[18][19] A PNHNHP ligand was used to synthesize the PGeP pincer stabilized germylene by treatment with two equivalents of potassium hexamethyldisilazide (KHMDS) and GeCl2·dioxane, which finally leads to the formation of the PGeP pincer compound.[20]

Reactivity
[edit]Oligomerization and polymerization
[edit]Dimerization of carbon substituted germylenes gives R2Ge=GeR2 dimers which could further polymerize to form polygermanes (R2Ge)n compounds.[5] The dimer could show a certain stability if prepared in an independent way.[21] Bulkier substituents are able to reduce the polymerization rate by steric effect.[22] More steric hindrance could even stop the polymerization or dimerization reactions and renders a germylene thermodynamically stable.[23][24][25]

Insertion into σ bond
[edit]R2Ge insertion into C-C bonds has not been reported so far.[5] However, going down the group 14, C-E (E = Si, Ge, Sn, Pb) bonds become more accessible for R2Ge insertion.[5] The strained C-Ge bonds allow insertion of germylene to 7,7-dialkyl-7-germanorbornadienes in the melt, forming digermabicy-clooctadienes.[26]

C-H bonds are generally unreactive toward germylene insertion.[27] However, strain release may help to overcome the activation energy barrier.[25]

Insertion to carbon-halide bonds is common for germylene. The mechanism for insertion of free Me2Ge into the C-Br bond of benzyl bromide was reported to be a two-step, radical abstraction-recombination process under thermal and photolytical conditions.[28][29] An identical mechanism through a caged singlet radical pair was proposed for C-Cl bond insertion.[29] However, the interaction between halogen electrons and empty p-orbital of the germylene may result in the formation of a donor-acceptor complex before occurrence of any of the insertion mechanisms.[5]

The insertion into the C-Hal bond in alkyne compounds go by a one-step mechanism under thermal or photolytical conditions.[30]

For C-O, the R2Ge insertion product could only remain stable at a very low temperature.[31]

Addition to unsaturated systems
[edit]Addition reaction of Me2Ge to unsaturated systems is well studied. As mentioned above, dimerization and polymerization of Me2Ge does not have a noticeable activation energy barrier and is only controlled by diffusion. As a result, addition reactions should be rapid enough complete before getting polygermanes as products.
There is no reaction between simple alkenes and free germylenes.[5] However, styrenes and α-substituted styrenes are able to react with Me2Ge. The resulting product is a 1:1 mixture of the syn and anti-isomers of 3,4-diphenyl-3,4-R-1,1-dimethyl-1-germacyclopentane.[32]

A variety of 1,2-substituted-vinylgermyl compounds can be synthesized in both high yield and high regioselectivity by addition of germylene to alkynes.[33]

1,4-Cycloaddition of conjugated (hetero-)dienes by free germylenes gives the corresponding 5-membered ring.[34][35][36]

Germylenes reacts only with one of the strained double bonds in cumulated systems like allenes (C=C=C).[5] Germylenes prefer to react with more electron-deficient allenes.[5]

Complexation by donors
[edit]During complexation with donors, the germylenes stay in the singlet ground state, where the lone pair is placed in the high-s-character orbital, while the heteroatom-containing donors like R2O, ROH, R2S, R3P, R3N and RCl interact with the vacant p-orbital at germanium center, which could stabilize the singlet germylene and prevent further polymerization. Most of the complexes are stable in room temperature.[37] The absorption bands of adducts commonly exhibits shorter wavelengths in comparison to those of the free germylenes due to substituent-influenced n-p transitions at the Ge center.[31]
Germylene catalyzed reaction
[edit]Germylenes could also act as catalysts as transition metals do.[11] Oxidative addition and reductive elimination, along with the related Mn+/M(n+2)+ redox couples are of great significance to the transition metal catalysis.[38] Due to the electronic structure and chemical properties of germylenes, including the empty p-orbital, germylenes are able to activated small molecules and give the corresponding Ge(IV) complexes, which raised researchers' interests in germylenes' acting as spectator ligands in certain catalytic cycles.[39] However, subsequent regeneration of Ge(II) compound through reductive elimination is not thermodynamically favored for germylenes.[11] The key of germylene catalysis chemistry is to maintain a balance between oxidative addition and reductive elimination.[40] One example of germylene catalyzed reaction is hydroboration of CO2, where a preliminary hydrogermylation of CO2 step is followed by the formation of methanol derivatives with 3 equivalent of catecholborane to regenerate the germylene compound.[41]

See also
[edit]References
[edit]- ^ a b c d Mizuhata, Yoshiyuki; Sasamori, Takahiro; Tokitoh, Norihiro (2009). "Stable Heavier Carbene Analogues". Chemical Reviews. 109 (8): 3479–3511. doi:10.1021/cr900093s. ISSN 0009-2665. PMID 19630390.
- ^ Sasamori, T.; Tokitoh, N. (2005). "Encyclopedia of Inorganic Chemistry. 2nd ed. Edited by R. Bruce King". Angewandte Chemie International Edition. 45 (36): 1698–1740. doi:10.1002/anie.200585394. ISSN 1433-7851.
- ^ a b c Marschner, Christoph (2015). "Silylated Group 14 Ylenes: An Emerging Class of Reactive Compounds". European Journal of Inorganic Chemistry. 2015 (23): 3805–3820. doi:10.1002/ejic.201500495. ISSN 1434-1948.
- ^ a b Gaspar, P. P. (1985). Moss, R. A.; Jones, M. (eds.). "Reactive Intermediates III". Journal of Natural Products. 67 (7). New York: J. Wiley: 1199–1200. doi:10.1021/np030742g.
- ^ a b c d e f g h Neumann, Wilhelm P. (1991). "Germylenes and stannylenes". Chemical Reviews. 91 (3): 311–334. doi:10.1021/cr00003a002. ISSN 0009-2665.
- ^ Jutzi, P.; Becker, A.; Stammler, H. G.; Neumann, B. (1991). "Synthesis and solid-state structure of (Me3Si)3CGeCH(SiMe3)2, a monomeric dialkylgermylene". Organometallics. 10 (6): 1647–1648. doi:10.1021/om00052a002. ISSN 0276-7333.
- ^ Barthelat, Jean Claude; Roch, Bruno Saint; Trinquier, Georges; Satge, Jacques (1980). "Structure and singlet-triplet separation in simple germylenes GeH2, GeF2, and Ge(CH3)2". Journal of the American Chemical Society. 102 (12): 4080–4085. doi:10.1021/ja00532a017. ISSN 0002-7863.
- ^ Olbrich, Gottfried (1980). "Restricted hartree—fock calculations on GeH2 and SnH2". Chemical Physics Letters. 73 (1): 110–113. Bibcode:1980CPL....73..110O. doi:10.1016/0009-2614(80)85214-6. ISSN 0009-2614.
- ^ Collins, Scott; Murakami, Shu; Snow, James T.; Masamune, Satoru (1985). "Generation and reactivity of bis(2,6-diethylphenyl)germanium(II)". Tetrahedron Letters. 26 (10): 1281–1284. doi:10.1016/s0040-4039(00)94870-6. ISSN 0040-4039.
- ^ Fischer, E. O.; Grubert, H. Z. (1956). "Über Aromatenkomplexe von Metallen. IV. Di-cyclopentadienyl-blei". Naturforsch. 91 (5–6): 237–242. doi:10.1002/zaac.19562860507.
- ^ a b c d Dasgupta, Rajarshi; Khan, Shabana (2020), "N-heterocyclic germylenes and stannylenes: Synthesis, reactivity and catalytic application in a nutshell", Advances in Organometallic Chemistry, vol. 74, Elsevier, pp. 105–152, doi:10.1016/bs.adomc.2020.04.001, ISBN 978-0-12-820692-8, S2CID 226476659
- ^ a b Raut, Ravindra; Amin, Sheikh; Sahoo, Padmini; Kumar, Vikas; Majumdar, Moumita (2018). "One-Pot Synthesis of Heavier Group 14 N-Heterocyclic Carbene Using Organosilicon Reductant". Inorganics. 6 (3): 69. doi:10.3390/inorganics6030069. ISSN 2304-6740.
- ^ Lavallo, Vincent; Canac, Yves; Präsang, Carsten; Donnadieu, Bruno; Bertrand, Guy (2005). "Stable Cyclic (Alkyl)(Amino)Carbenes as Rigid or Flexible, Bulky, Electron-Rich Ligands for Transition-Metal Catalysts: A Quaternary Carbon Atom Makes the Difference". Angewandte Chemie International Edition. 44 (35): 5705–5709. doi:10.1002/anie.200501841. ISSN 1433-7851. PMC 2427276. PMID 16059961.
- ^ Wang, Liliang; Lim, Yi; Li, Yongxin; Ganguly, Rakesh; Kinjo, Rei (2016). "Isolation of a Cyclic (Alkyl)(amino)germylene". Molecules. 21 (8): 990. doi:10.3390/molecules21080990. ISSN 1420-3049. PMC 6273211. PMID 27483223.
- ^ Del Rio, Natalia; Baceiredo, Antoine; Saffon-Merceron, Nathalie; Hashizume, Daisuke; Lutters, Dennis; Müller, Thomas; Kato, Tsuyoshi (2016). "A Stable Heterocyclic Amino(phosphanylidene-σ4-phosphorane) Germylene". Angewandte Chemie International Edition. 55 (15): 4753–4758. doi:10.1002/anie.201511956. ISSN 1433-7851. PMID 26954722.
- ^ Rit, Arnab; Tirfoin, Rémi; Aldridge, Simon (2016). "Exploiting Electrostatics To Generate Unsaturation: Oxidative GeE Bond Formation Using a Non π-Donor Stabilized [R(L)Ge:] + Cation". Angewandte Chemie International Edition. 55 (1): 378–382. doi:10.1002/anie.201508940. PMID 26545498.
- ^ Dong, Zhaowen; Reinhold, Crispin R. W.; Schmidtmann, Marc; Müller, Thomas (2016). "A Germylene Stabilized by Homoconjugation". Angewandte Chemie International Edition. 55 (51): 15899–15904. doi:10.1002/anie.201609576. PMID 27860046.
- ^ van der Boom, Milko E.; Milstein, David (2003). "Cyclometalated Phosphine-Based Pincer Complexes: Mechanistic Insight in Catalysis, Coordination, and Bond Activation". Chemical Reviews. 103 (5): 1759–1792. doi:10.1021/cr960118r. ISSN 0009-2665. PMID 12744693.
- ^ Kiefer, Claude; Bestgen, Sebastian; Gamer, Michael T.; Kühn, Michael; Lebedkin, Sergei; Weigend, Florian; Kappes, Manfred M.; Roesky, Peter W. (2017). "Coinage Metal Complexes of Bis-Alkynyl-Functionalized N-Heterocyclic Carbenes: Reactivity, Photophysical Properties, and Quantum Chemical Investigations". Chemistry - A European Journal. 23 (7): 1591–1603. doi:10.1002/chem.201604292. PMID 27780290.
- ^ Pan, Baofei; Evers-McGregor, Deirdra A.; Bezpalko, Mark W.; Foxman, Bruce M.; Thomas, Christine M. (2013). "Multimetallic Complexes Featuring a Bridging N -heterocyclic Phosphido/Phosphenium Ligand: Synthesis, Structure, and Theoretical Investigation". Inorganic Chemistry. 52 (16): 9583–9589. doi:10.1021/ic4012873. ISSN 0020-1669. PMID 23923983.
- ^ Marchand, Annette; Gerval, Pierre; Duboudin, Françoise; Gaufryau, M.-H.; Joanny, Marguerite; Mazerolles, Pierre (1984). "Mise en evidence de la formation de R2MMR2 a partir de disila-1,2 et digerma-1,2 cyclohexenes-4". Journal of Organometallic Chemistry. 267 (1): 93–106. doi:10.1016/0022-328x(84)80162-x. ISSN 0022-328X.
- ^ Collins, Scott; Murakami, Shu; Snow, James T.; Masamune, Satoru (1985). "Generation and reactivity of bis(2,6-diethylphenyl)germanium(II)". Tetrahedron Letters. 26 (10): 1281–1284. doi:10.1016/s0040-4039(00)94870-6. ISSN 0040-4039.
- ^ Fjeldberg, Torgny; Haaland, Arne; Schilling, Birgitte E. R.; Lappert, Michael F.; Thorne, Andrew J. (1986). "Subvalent Group 4B metal alkyls and amides. Part 8. Germanium and tin carbene analogues MR2[M = Ge or Sn, R = CH(SiMe3)2]: syntheses and structures in the gas phase (electron diffraction); molecular-orbital calculations for MH2 and GeMe2". Journal of the Chemical Society, Dalton Transactions (8): 1551. doi:10.1039/dt9860001551. ISSN 0300-9246.
- ^ VEITH, M. (1987). "ChemInform Abstract: Unsaturated Molecules Containing Main Group Metals". ChemInform. 18 (18): 1. doi:10.1002/chin.198718388. ISSN 0931-7597.
- ^ a b Lange, Lutz; Meyer, Bernd; du Mont, Wolf-Walther (1987). "Bis(2,4,6-tri-t-butylphenyl)germylen und Bis(2,4,6-tri-t-butylphenyl)germathion: Isomerisierung durch spontane C,H-Insertion". Journal of Organometallic Chemistry. 329 (2): C17–C20. doi:10.1016/s0022-328x(00)99800-0. ISSN 0022-328X.
- ^ Bleckmann, Paul; Minkwitz, Rolf; Neumann, Wilhelm P.; Schriewer, Michael; Thibud, Manfred; Watta, Bärbel (1984). "Dimethyl germylene insertion into a strained C-Ge bond and matrix isolation of tetramethyl digermene Me2Ge=GeMe2". Tetrahedron Letters. 25 (23): 2467–2470. doi:10.1016/s0040-4039(01)81206-5. ISSN 0040-4039.
- ^ Nefedov, O. M.; Skell, P. S. (1981). "THE GENERATION AND REACTIONS OF DIMETHYLSILYLENE AND DIMETHYLGERMYLENE IN VAPOR-PHASE". Doklady Akademii Nauk SSSR. 259 (2): 377–379.
- ^ Koecher, Juergen; Lehnig, Manfred (1984). "Insertion reactions of dimethylgermylene, Me2Ge, and their mechanisms as studied by CIDNP". Organometallics. 3 (6): 937–939. doi:10.1021/om00084a022. ISSN 0276-7333.
- ^ a b Koecher, Juergen.; Lehnig, Manfred.; Neumann, Wilhelm P. (1988). "Chemistry of heavy carbene analogs R2M (M = Si, Ge, Sn). 12. Concerted and nonconcerted insertion reactions of dimethylgermylene into the carbon-halogen bond". Organometallics. 7 (5): 1201–1207. doi:10.1021/om00095a029. ISSN 0276-7333.
- ^ Billeb, G.; Brauer, H.; Maslov, S.; Neumann, W.P. (1989). "Chemie der schweren Carben-Analogen R2M, M = Si, Ge, Sn". Journal of Organometallic Chemistry (in German). 373 (1): 11–19. doi:10.1016/0022-328X(89)85019-3.
- ^ a b Ando, Wataru.; Itoh, Hiroyuki.; Tsumuraya, Takeshi. (1989). "Electronic absorption spectra of diorganogermylenes in matrixes: formation of diorganogermylene complexes with heteroatom-containing substrates". Organometallics. 8 (12): 2759–2766. doi:10.1021/om00114a004. ISSN 0276-7333.
- ^ Koecher, Jurgen; Neumann, Wilhelm P. (1985). "Germacyclopentanes via cycloadditions of free dimethylgermylene to styrenes". Organometallics. 4 (2): 400–402. doi:10.1021/om00121a038. ISSN 0276-7333.
- ^ Billeb, Gilbert; Brauer, Hartmut; Neumann, Wilhelm P. (1990). "Regiospecific Synthesis of Vinylgermyl Compounds from Alkynes or Allenes and Free Singlet Dimethylgermylene". Synlett. 1990 (3): 113–114. doi:10.1055/s-1990-21006. ISSN 0936-5214. S2CID 98481510.
- ^ Schriewer, Michael; Neumann, Wilhelm P. (1983). "Chemistry of heavy carbene analogs R2M (M = Si, Ge, Sn). 8. Germylenes: singlets or triplets? [2 + 4]Cheletropic cycloadditions of dimethylgermylene and diiodogermylene to conjugated dienes". Journal of the American Chemical Society. 105 (4): 897–901. doi:10.1021/ja00342a043. ISSN 0002-7863.
- ^ Michels, Erhard; Neumann, Wilhelm P. (1986). "Additions of free dimethylgermylene to vinyl ketones and α-diketones". Tetrahedron Letters. 27 (22): 2455–2458. doi:10.1016/s0040-4039(00)84554-2. ISSN 0040-4039.
- ^ Bootz, Konrad; Neumann, Wilhelm P. (1989). "The first synthesis of aza-germacyclopentenes: Cycloadditions of 1-aza- and 1,4-diazabutadienes with free singlet dimethyl germylene". Tetrahedron Letters. 30 (48): 6669–6672. doi:10.1016/s0040-4039(00)70646-0. ISSN 0040-4039.
- ^ Nefedov, O. M.; Kolesnikov, S. P.; Rogozhin, I. S. (1980). "Molecular complexes of germylene with n-donor ligands". Proceedings of the Academy of Sciences of the USSR Series Chemical: 170–173.
- ^ Dunn, Nicole L.; Ha, Minji; Radosevich, Alexander T. (2012). "Main Group Redox Catalysis: Reversible P III /P V Redox Cycling at a Phosphorus Platform". Journal of the American Chemical Society. 134 (28): 11330–11333. doi:10.1021/ja302963p. ISSN 0002-7863. PMID 22746974.
- ^ Herrmann, W. A. (2002). "N-heterocyclic carbene: a new concept in organometallic catalysis". Angew Chem Int Ed. 41 (8): 1290–1309. doi:10.1002/1521-3773(20020415)41:8<1290::AID-ANIE1290>3.0.CO;2-Y. PMID 19750753.
- ^ Peng, Yang; Ellis, Bobby D.; Wang, Xinping; Power, Philip P. (2008). "Diarylstannylene Activation of Hydrogen or Ammonia with Arene Elimination". Journal of the American Chemical Society. 130 (37): 12268–12269. doi:10.1021/ja805358u. ISSN 0002-7863. PMID 18714994.
- ^ Hadlington, Terrance J.; Kefalidis, Christos E.; Maron, Laurent; Jones, Cameron (2017). "Efficient Reduction of Carbon Dioxide to Methanol Equivalents Catalyzed by Two-Coordinate Amido–Germanium(II) and −Tin(II) Hydride Complexes". ACS Catalysis. 7 (3): 1853–1859. doi:10.1021/acscatal.6b03306. ISSN 2155-5435.
