Dechlorane plus

| |
| Names | |
|---|---|
| IUPAC name
1,2,3,4,7,8,9,10,13,13,14,14-dodecachloro-1,4,4a,5,6,6a,7,10,10a,11,12,12a-dodecahydro-1,4,7,10-dimethanodibenzo[a,e]cyclooctene
| |
| Other names
Dechloran A
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.033.575 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C18H12Cl12 | |
| Molar mass | 653.72 g/mol |
| Appearance | white, crystalline solid |
| Density | 1.8 g/cm3 |
| Melting point | 350 °C (662 °F; 623 K) (decomposes) |
| 0.044–249 μg/L | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Dechlorane plus (abbrev. DDC-CO[1]) is a polychlorinated flame retardant produced by Oxychem.
It is produced by the Diels-Alder reaction of two equivalents of hexachlorocyclopentadiene with one equivalent of cyclooctadiene. The syn and anti isomer are formed in the approximate ratio of 1:3.[3][4]
-
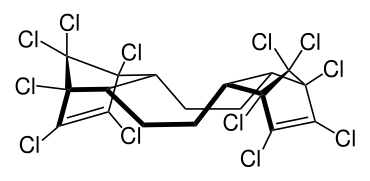
syn isomer
-
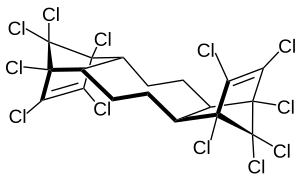
anti isomer
Environmental concerns
[edit]Dechlorane plus was first found in the environment in 2006 in air around the Great Lakes.[5] Since then, its environmental occurrence has been further documented in several studies including sediments of the Great Lakes;[6] zooplankton, fish, and mussels in Lake Winnipeg and Lake Ontario;[7] air and seawater from the Arctic to Antarctica;[8] and Chinese air.[9] In 2011, modeling data indicated that Dechlorane Plus may be persistent, bioaccumulative, and subject to long-range transport.[10] The 2023 Conference of the Parties of the United Nations Stockholm Convention on Persistent Organic Pollutants took the decision to eliminate the use of Dechlorane Plus, by listing this chemical in Annex A to the Convention.[11]
Dechlorane plus was added to the list of REACH Substances of Very High Concern on January 15, 2018.[12]
Literature
[edit]- ENVIRON Health Sciences Institute. "Dechlorane Plus High Production Volume (HPV) Chemical Challenge Program Test Plan. 201-15635" (PDF). Archived from the original (PDF) on 2012-10-23.
References
[edit]- ^ Bergman, Å.; Rydén, A.; Law, R. J.; De Boer, J.; Covaci, A.; Alaee, M.; Birnbaum, L.; Petreas, M.; Rose, M.; Sakai, S.; Van Den Eede, N.; Van Der Veen, I. (2012). "A novel abbreviation standard for organobromine, organochlorine and organophosphorus flame retardants and some characteristics of the chemicals". Environment International. 49: 57–82. doi:10.1016/j.envint.2012.08.003. PMC 3483428. PMID 22982223.
- ^ "1,6,7,8,9,14,15,16,17,17,18,18-dodecachloropent... - Registration Dossier - ECHA". echa.europa.eu. Retrieved 2020-03-18.
- ^ Sverko, E.; Tomy, G. T.; Reiner, E. J.; Li, Y. F.; McCarry, B. E.; Arnot, J. A.; Law, R. J.; Hites, R. A. (2011). "Dechlorane Plus and Related Compounds in the Environment: A Review". Environmental Science & Technology. 45 (12): 5088–5098. Bibcode:2011EnST...45.5088S. doi:10.1021/es2003028. PMID 21574656.
- ^ Garcia, J. G.; Fronczek, F. R.; McLaughlin, M. L. (1991). "Tandem reverse-electron-demand diels-alder reactions of 1,5-cyclooctadiene". Tetrahedron Letters. 32 (28): 3289–3292. doi:10.1016/S0040-4039(00)92688-1.
- ^ Hoh, Eunha; Lingyan Zhu; Ronald A. Hites (2006). "Dechlorane Plus, a chlorinated flame retardant, in the Great Lakes". Environmental Science & Technology. 40 (4): 1184–1189. Bibcode:2006EnST...40.1184H. doi:10.1021/es051911h. PMID 16572773.
- ^ Sverko, Ed; Gregg T. Tomy; Chris H. Marvin; Donna Zaruk; Eric Reiner; Paul A. Helm; Brad Hill; Brian E. McCarry (2007). "Dechlorane plus levels in sediment of the lower Great Lakes". Environmental Science & Technology. 42 (2): 361–366. doi:10.1021/es0710104. PMID 18284131.
- ^ Tomy, Gregg; Kerri Pleskach; Nargis Ismail; D. Michael Whittle; Paul A. Helm; E. D. Sverko; Donna Zaruk; Chris H. Marvin (2007). "Isomers of dechlorane plus in Lake Winnipeg and Lake Ontario food webs". Environmental Science & Technology. 41 (7): 2249–2254. Bibcode:2007EnST...41.2249T. doi:10.1021/es062781v. PMID 17438771.
- ^ Möller, Axel; Zhiyong Xie; Renate Sturm; Ralf Ebinghaus (2010). "Large-scale distribution of Dechlorane Plus in air and seawater from the Arctic to Antarctica". Environmental Science & Technology. 44 (23): 8977–8982. Bibcode:2010EnST...44.8977M. doi:10.1021/es103047n. PMID 21047104.
- ^ Ren, Nanqi; Ed Sverko; Yi-Fan Li; Zhi Zhang; Tom Harner; Degao Wang; Xinnan Wan; Brian E. McCarry (2008). "Levels and isomer profiles of Dechlorane Plus in Chinese air". Environmental Science & Technology. 42 (17): 6476–6480. Bibcode:2008EnST...42.6476R. doi:10.1021/es800479c. PMID 18800517.
- ^ Sverko, Ed; Gregg T. Tomy; Eric J. Reiner; Yi-Fan Li; Brian E. McCarry; Jon A. Arnot; Robin J. Law; Ronald A. Hites (2011). "Dechlorane plus and related compounds in the environment: a review". Environmental Science & Technology. 45 (12): 5088–5098. Bibcode:2011EnST...45.5088S. doi:10.1021/es2003028. PMID 21574656.
- ^ "Governments accelerate action and take bold decisions to address pollution from chemicals and wastes". Secretariat of the Basel, Rotterdam and Stockholm Conventions. May 15, 2023. Retrieved 6 July 2023.
- ^ "1,6,7,8,9,14,15,16,17,17,18,18-Dodecachloropentacyclo[12.2.1.16,9.02,13.05,10]octadeca-7,15-diene ("Dechlorane Plus"™) - Candidate List of substances of very high concern for Authorisation - ECHA". European Chemicals Agency. ECHA. Retrieved January 16, 2018.