Corey–Seebach reaction
| Corey–Seebach reaction | |
|---|---|
| Named after | Elias James Corey Dieter Seebach |
| Reaction type | Coupling reaction |
| Identifiers | |
| Organic Chemistry Portal | corey-seebach-reaction |
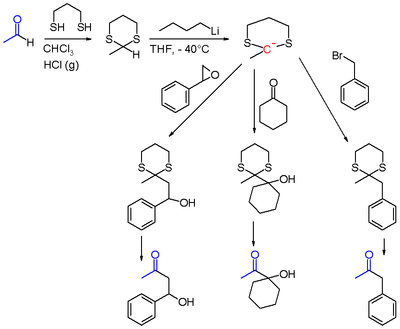
The Corey–Seebach reaction, or Seebach Umpolung is a name reaction of organic chemistry that allows for acylation by converting aldehydes into lithiated 1,3-dithianes. The lithiated 1,3-dithianes serves as an acyl anion equivalent, undergoing alkylation with electrophiles.[1] The reaction is named in honor of its discoverers, Elias J. Corey and Dieter Seebach.
Implementation
[edit]The aldehyde is first converted into a dithiane, usually with 1,3-propanedithiol. The resulting 1,3-dithiane is then lithiated with the use of butyllithium. The 2-lithio-1,3-dithiane reacts with electrophiles to give a 2-alkyl-1,3-dithiane.
Finally, the 2-alkyl-1,3-dithiane can be converted to a carbonyl by hydrolysis, usually with the use of mercury(II) oxide. Alternatively the 2-alkyl-1,3-dithiane can be reduced to an alkane.[2]
Scope
[edit]As a strategy for protecting aldehydes and ketones, dithiane formation is cumbersome because deprotection is inefficient. Typically ketones and aldehydes are protected as their dioxolanes instead of dithianes. The Corey–Seebach reaction is of interest as an acyl anion equivalent, allowing aldehydes to be converted to ketones.
The lithiated 1,3-dithiane can be alkylated with alkyl halides, epoxides,[3][4] ketones, acyl halides, and iminium salts, which after hydrolysis of dithioacetals can yield ketones, β-hydroxyketones, α-hydroxyketones, 1,2-diketones[5] and α-aminoketones. Notably, α-hydroxyketones and 1,2-diketones can not be generated through typical reactions of aldehydes such as the aldol reaction.[6] Other possible electrophiles include aldehydes,[7] amides, and esters.[6][8]
The reaction between lithiated 1,3-dithianes and arenesulfonates offers a similar path to that of alkyl halides, being able to form dithioacetals which can be converted to ketones.[9]
Historic references
[edit]- Corey, Elias James; Seebach, Dieter (1 December 1965). "Synthesis of 1,n-Dicarbonyl Derivates Using Carbanions from 1,3-Dithianes". Angewandte Chemie International Edition. 4 (12): 1077–1078. doi:10.1002/anie.196510771.
- Corey, Elias James; Seebach, Dieter (1 December 1966). "Phenylthiomethyllithium and Bis(phenylthio)methyllithium". The Journal of Organic Chemistry. 31 (12): 4097–4099. doi:10.1021/jo01350a052.
- Seebach, Dieter; Corey, Elias James (1 January 1975). "Generation and synthetic applications of 2-lithio-1,3-dithianes". The Journal of Organic Chemistry. 40 (2): 231–237. doi:10.1021/jo00890a018.
References
[edit]- ^ Smith, Amos B.; Adams, Christopher M. (6 May 2004). "Evolution of Dithiane-Based Strategies for the Construction of Architecturally Complex Natural Products". Accounts of Chemical Research. 37 (6): 365–377. doi:10.1021/ar030245r. PMID 15196046.
- ^ Yus, Miguel; Nájera, Carmen; Foubelo, Francisco (11 August 2003). "The role of 1,3-dithianes in natural product synthesis". Tetrahedron. 59 (13): 6147–6212. doi:10.1016/S0040-4020(03)00955-4.
- ^ Williams, David R.; Sit, Sing Yuen (1 May 1984). "Total synthesis of (+)-phyllanthocin". Journal of the American Chemical Society. 106 (10): 2949–2954. doi:10.1021/ja00322a035.
- ^ Smith, Amos B.; Xian, Ming (11 January 2006). "Anion Relay Chemistry: An Effective Tactic for Diversity Oriented Synthesis". Journal of the American Chemical Society. 128 (1): 66–67. doi:10.1021/ja057059w. PMID 16390124.
- ^ Corey, Elias James; Seebach, Dieter (1 December 1965). "Synthesis of 1,n-Dicarbonyl Derivates Using Carbanions from 1,3-Dithianes". Angewandte Chemie International Edition. 4 (12): 1077–1078. doi:10.1002/anie.196510771.
- ^ a b Page, Philip C. Bulman; van Niel, Monique B.; Pordger, Jeremy C. (29 November 1988). "Synthetic uses of the 1,3-dithiane grouping from 1977 to 1988". Tetrahedron. 45 (24): 7643–7677. doi:10.1016/S0040-4020(01)85784-7.
- ^ Ley, Steven V.; Abad-Somovilla, Antonio; Anderson, James C.; Ayats, Carles; Bänteli, Rolf; Beckmann, Edith; Boyer, Alistair; Brasca, Maria G.; Brice, Abigail; Broughton, Howard B.; Burke, Brenda J.; Cleator, Ed; Craig, Donald; Denholm, Alastair A.; Denton, Ross M.; Durand-Reville, Thomas; Gobbi, Luca B.; Göbel, Michael; Gray, Brian Lawrence; Grossmann, Robert B.; Gutteridge, Clair E.; Hahn, Norbert; Harding, Sarah L.; Jennens, David C.; Jennens, Lynn; Lovell, Peter J.; Lovell, Helen J.; de la Puente, Mary L.; Kolb, Martmuth C.; Koot, Win-Jan; Maslen, Sarah L.; McCusker, Catherine; Mattes, Amos; Pape, Andrew R.; Pinto, Andrea; Santafianos, Dinos; Scott, James S.; Smith, Stephen C.; Somers, Andrew Q.; Spilling, Christopher D.; Stelzer, Frank; Toogood, Peter L.; Turner, Richard M; Veitch, Gemma E.; Wood, Anthony; Zumbrunn, Cornelia (1 January 2018). "The synthesis of azadirachtin: a potent insect antifeedant". Chemistry: A European Journal. 14 (34): 10683–10704. doi:10.1002/chem.200801103. PMID 18821532.
- ^ Chen, Yue-Lei; Leguijt, Robin; Redlich, Hartmut; Fröhlich, Roland (30 April 2006). "Trimethylene Dithioacetals of Carbohydrates, Part 6: C-C Coupling Reactions of Dilithiated N-Acetyl-D-glucosamine Trimethylene Dithioacetal Derivatives". Synthesis. 2006 (24): 4212–4218. doi:10.1055/s-2006-950359.
- ^ Seebach, Dieter; Wilka, Eva-Maria (1976). "Alkylation of 2-Lithio-1,3-dithianes with Arenesulfonates of Primary Alcohols". Synthesis. 1976 (7): 476–477. doi:10.1055/s-1976-24092.