Cardiovascular agents
This article has an unclear citation style. (May 2024) |
Cardiovascular agents are drugs used to treat diseases associated with the heart or blood vessels. These medications are available for purchase only with a physician’s prescription. They include, but are not limited to, drugs that target hypertension (antihypertensives), hyperlipidemia (antihyperlipidemics) and blood clotting (blood-thinners) to reduce the risk of cardiovascular diseases.
Antihypertensive agents are classified according to their mechanism of actions. The most common classes prescribed are diuretics, angiotensin-converting enzyme inhibitors (ACEIs), angiotensin II receptor blockers (ARBs), calcium channel blockers (CCBs) and beta-blockers.
Antihyperlipidemic agents most often prescribed are statins, ezetimibe and fibrates. They either lower low-density lipoprotein cholesterol (LDL-C) or triglyceride (TG) levels in blood to manage hypercholesterolaemia.
Blood-thinning agents, particularly antiplatelets and anticoagulants, maintain smooth blood flow by preventing blood clot formation in blood vessels. Two main categories of antiplatelets are COX-1 inhibitors and ADP receptor inhibitors, while anticoagulants include vitamin K antagonists, direct oral anticoagulants (DOACs) and indirect thrombin inhibitors.
Since cardiovascular agents have narrow therapeutic windows, a slight rise in dose may result in severe toxicity. Hence, monitoring at baseline and during therapy is needed. For drug overdose, stabilisation and antidotes help lower drug concentrations.
Terminology
[edit]Cardiovascular agents are drugs that affect the rate and intensity of cardiac contraction, blood vessel diameters, blood volume, blood clotting and blood cholesterol levels.[1] They are indicated to treat diseases related to the heart or the vascular system (blood vessels), such as hypertension, hyperlipidemia, coagulation disorders, heart failure and coronary artery disease.[1] These drugs are prescription-only medicines, meaning that they should be administered strictly under a doctor’s instruction and can only be obtained by means of a doctor’s prescription.
Drug classes
[edit]Antihypertensive agents
[edit]Antihypertensive agents comprise multiple classes of compounds that are intended to manage hypertension (high blood pressure). Antihypertensive therapy aims to maintain a blood pressure goal of <140/90 mmHg in all patients, as well as to prevent the progression or recurrence of cardiovascular diseases (CVD) in hypertensive patients with established CVD.[2] An optimal blood pressure control is essential to prevent target-organ damage associated with complications of hypertension such as heart failure, ischemic heart diseases, stroke, and renal failure, ultimately reducing the risk of premature mortality.[2] Antihypertensives are classified by different mechanisms or sites of action. Some of the most commonly used drugs to treat hypertension include diuretics, angiotensin-converting enzyme inhibitors (ACEI), angiotensin II receptor blockers (ARBs), calcium channel blockers (CCBs), and beta-blockers.
Diuretics
[edit]
Diuretics act primarily by reducing the reabsorption of sodium at different sites of the renal tubular system and consequently promoting the elimination of sodium and water with increased urine output.[3]
- Loop diuretics: furosemide, bumetanide, torsemide, ethacrynic acid
- Thiazide diuretics: chlorothiazide, hydrochlorothiazide
- Thiazide-like diuretics: metolazone, indapamide, chlorthalidone
- Potassium-sparing diuretics: amiloride, triamterene, spironolactone, eplerenone
For loop diuretics, thiazide diuretics and thiazide-like diuretics, their common side effects include hypokalemia, hyponatremia, metabolic alkalosis and hyperglycaemia.[4] For potassium-sparing diuretics, its common side effects include hyponatremia, hyperkalemia, metabolic acidosis and sexual dysfunction specifically for spironolactone.[4][5]
The use of diuretics should be avoided in patients with severe dehydration, anuria (absence of urine production).[4] Diuretics are contraindicated in cases of severe electrolyte abnormalities and should not be administered until an electrolyte balance is restored. Special attention should be given to the use of thiazide and loop diuretics as they may exacerbate diabetes and gout.[4]
Angiotensin-converting-enzyme inhibitors (ACEI)
[edit]Angiotensin-converting-enzyme inhibitors (ACEI) block the conversion of angiotensin I to angiotensin II by inhibiting the action of angiotensin-converting-enzyme, causing the reduction of blood volume and peripheral vascular resistance.[6]
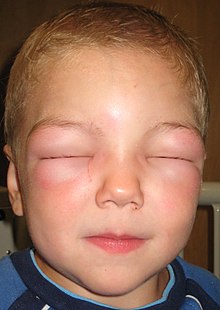
Some side effects of ACEI include hypotension, renal insufficiency, and hyperkalemia.[7] Dry cough is also a common side effect believed to be associated with decreased bradykinin breakdown. Angioedema is another possible but rare complication due to elevated levels of bradykinin.[6]
ACEI should not be used in combinations with angiotensin II receptor blockers (ARBs) or direct renin inhibitors and is contraindicated in people with a history of angioedema and pregnancy.[6][8] “Triple whammy”, the concurrent use of an ACEI with diuretics and non-steroidal anti-inflammatory drugs (NSAIDs), is also contraindicated as this combination has been correlated with an increased risk of acute kidney injury.[8][7] ACEI should be used with caution in patients with renal impairment, and renal failure risk in severe bilateral renal stenosis.[8]
Angiotensin II receptor blockers (ARBs)
[edit]Angiotensin II receptor blockers (ARBs) work by inhibiting the action of angiotensin II on, specifically AT1 receptors to prevent the vasoconstrictor effects of this receptor and block the peripheral sympathetic activity.[9]
ARBs are generally well-tolerated, in which they are less likely to cause cough or angioedema compared to ACEI. Common side effects include hypotension, renal insufficiency, and hyperkalemia.[7]
The contraindications of ARBs are similar to those of ACEI, including the contraindicated combinations with ACEI or direct renin inhibitors, "triple whammy" (the concurrent use of an ARB with diuretics and NSAIDs) and in patients with a history of angioedema and pregnancy.[6][7][8] In addition, ARB should be used with caution in patients with renal impairment and renal failure risk in severe bilateral renal stenosis[8]
Calcium channel blockers (CCBs)
[edit]Calcium channel blockers (CCBs) preferentially block the L-type voltage-gated calcium channels to prevent the flow of calcium influx in the blood vessels and the heart, thereby reducing peripheral vascular resistance and cardiac output respectively.[10]
- Dihydropyridines (DHP): amlodipine, nifedipine, felodipine, nicardipine
- Non-dihydropyridines (non-DHP): diltiazem, verapamil
In general, the side effects of CCBs include peripheral edema and gingival hyperplasia when CCBs are used chronically.[11][12] To add on, DHP may cause reflex tachycardia and peripheral edema, while non-DHP may cause bradycardia and worsening of cardiac function due to reduced cardiac contractility and cardiac conduction.[12][13]
Non-dihydropyridines are contraindicated in patients with heart failure with reduced ejection fraction (HFrEF), and second- or third-degree atrioventricular block.[13] Special attention should be given to the coadministration of non-DHP with beta-blockers or ivabradine due to the increased risk of bradycardia.[13] Since both DHP and non-DHP are metabolized through the CYP3A4 system, grapefruit juice containing furanocoumarins (the potent inhibitors of the CYP3A4 enzyme) should be avoided.[13]
Beta-blockers
[edit]Beta-blockers act as competitive antagonists that block the effects of catecholamines at beta-adrenergic receptor sites, resulting in reduced rate and force of contraction of the heart, as well as reduced peripheral vascular resistance.[14]
- Non-selective beta-blockers: propranolol, nadolol, timolol
- Beta-1-selective beta-blockers: atenolol, bisoprolol, metoprolol, esmolol
- Beta-blockers with vasodilating effect: labetalol, carvedilol, nebivolol
- Beta-blockers with intrinsic sympathomimetic activity: acebutolol, pindolol
Some common side effects include increased airway resistance for non-selective beta-blockers, exacerbation of peripheral vascular diseases, and hypotension[15]
Beta-blockers are contraindicated in patients with second- or third-degree atrioventricular block. In particular, beta-blockers with intrinsic sympathomimetic activity are contraindicated in patients with myocardial infarction, heart failure or severe bradycardia.[15][16] Beta-blockers should be used with caution in patients with asthma or chronic obstructive pulmonary disease (COPD) due to bronchoconstriction, and in patients with diabetes mellitus (DM) due to masking of hypoglycaemia.[16]
Antihyperlipidemic agents
[edit]
Antihyperlipidemic agents are drugs used for the treatment of dyslipidemia, a condition of abnormal lipid levels in the body. It is characterised by elevations of low-density lipoprotein cholesterol (LDL-C) and triglycerides (TGs) in the blood.[17] Hypercholesterolaemia induces the formation of plaques due to the buildup of excess cholesterol within the arterial wall. This increases the risk of, or aggravate, atherosclerotic cardiovascular disease (ASCVD). Therefore, antihyperlipidemic drugs are introduced for primary and secondary coronary heart disease prevention, as well as for reduction in mortality from acute coronary outcomes.[18] These drugs include statins, ezetimibe and fibrates.
Statins
[edit]Statins, also known as beta-hydroxy-beta-methylglutaryl-Coenzyme A (HMG-CoA) reductase inhibitors, are the first-line drugs for hypercholesterolaemia.[19] Examples of this drug class are atorvastatin, rosuvastatin, fluvastatin, simvastatin, pravastatin and lovastatin.
Most efficacious in lowering LDL-C levels, statins block the action of HMG-CoA reductase through competitive inhibition.[18] HMG-CoA reductase, an enzyme found in hepatocytes, is responsible for the conversion of HMG-CoA to mevalonic acid for cholesterol biosynthesis. Inhibition of this enzyme reduces the synthesis and thus, availability of endogenous cholesterol. This reduction in intracellular cholesterol, in turn, causes an increase in the number of LDL receptors on hepatic cells. The elevation of LDL receptor expression decreases the plasma LDL-C level by promoting hepatic uptake of LDL from circulation.

While statins are generally well-tolerated, severe adverse effects such as hepatotoxicity and myotoxicity may occur in rarity. Statin-induced hepatotoxicity can cause autoimmune hepatitis and an elevation in serum levels of hepatic enzymes such as alanine aminotransferase, impairing liver function.[20] Myotoxicity is commonly presented with statin-associated muscle symptoms (SAMS), which include myalgia and myositis.[21] In rare cases, they may progress into rhabdomyolysis, a condition manifested by muscle necrosis and myoglobinuria due to heightened creatine kinase levels.[22][23] Another consequence of taking statins is the risk of developing new-onset diabetes, which is more prominent in individuals with high TG levels and body mass index (BMI).[19] However, the risk is far outweighed by the benefits from statin therapy for the reduction in cardiovascular outcomes.[20]
Given the potential of statins to exacerbate liver and muscle abnormalities, contraindications of statins include decompensated liver cirrhosis, acute liver failure, unexplained and persistent elevations of serum transaminases, and myopathy. Moreover, statins are not recommended in pregnancy as they may cause foetal harm because of their mechanism of action.[19]
Metabolised by the Cytochrome P450 (CYP450) enzyme, a major metabolic enzyme, simvastatin and lovastatin may accumulate in blood when administered with CYP450 inhibitors.[18][24] Some of these inhibitors are azole antifungals, macrolides, CCBs, ticagrelor (antiplatelet) and grapefruit juice. Concomitant use of statins with CYP450 inhibitors, along with gemfibrozil (fibrate), increases the risk of myopathy.[20][23][25] This is especially significant in patients under polypharmacy.
Ezetimibe
[edit]Ezetimibe is a selective cholesterol absorption inhibitor that inhibits the intestinal absorption of cholesterol by binding to the Niemann-Pick C1-Like 1 (NPC1L1) protein on the gastrointestinal epithelium.[23] This reduces the delivery of cholesterol to the liver, which then induces the upregulation of LDL receptor expression, lowering hepatic cholesterol stores and enhancing clearance of circulating LDL. More often prescribed as second-line therapy for dyslipidemia, ezetimibe is used in individuals with statin intolerance or those who failed to achieve the target LDL-C level on statin monotherapy.[26][27] In particular, ezetimibe and statin dual therapy have shown a 15% greater LDL-C decrease compared with same-dose statins alone, favouring recovery from acute coronary syndrome.[23]
Whilst ezetimibe intolerance is uncommon, some reports have been made regarding gastrointestinal and musculoskeletal effects.[28] Common adverse reactions of ezetimibe are nausea, abdominal pain, headache, fatigue, arthralgia, myalgia and hypersensitivity reactions.[29] On rare occasions, ezetimibe may cause cholecystitis, pancreatitis, elevation of serum transaminase level and rhabdomyolysis.
As hepatic impairment hinders the rate of ezetimibe metabolism by the liver, ezetimibe is not recommended in individuals with moderate or severe hepatic insufficiency due to prolonged systemic exposure to the drug.[19] In addition, similar to combined statin and fibrate intake, individuals should avoid the concurrent use of ezetimibe with gemfibrozil as it would increase ezetimibe concentration in the body.[25]
Fibrates
[edit]Fibrates, known as derivatives of fibric acids, are peroxisome proliferator-activated receptor alpha (PPAR-α) agonists primarily used for lowering TG levels and management of atherogenic dyslipidemia.[30][31][32] Typical drugs of the class include fenofibrate, bezafibrate, ciprofibrate and gemfibrozil.
Through activation of PPAR-α receptors, fibrates decreases circulating TG via upregulation of lipoprotein lipase (LPL) expression and downregulation of apolipoprotein C-III (ApoC-III) gene expression.[23][29] LPL is a hepatic enzyme involved in lipolysis, whereas ApoC-III is an inhibitor of LPL.
Comparable to statins and ezetimibe, fibrates are usually well-tolerated with mild adverse reactions, with the exception of gemfibrozil. Some of the more prevalent side effects are minor gastrointestinal disturbances such as abdominal pain and cholithiasis on account of the increased excretion of biliary cholesterol.[23] Among atypical adverse effects, myositis can occur in patients under gemfibrozil therapy, especially in those with renal insufficiency or under co-treatment with statins. In contrast, the risk of myopathy is much lower if fenofibrate is used in replacement of gemfibrozil owing to its different pharmacokinetic pathway. Other rare effects are increased serum transaminase levels, agranulocytosis and anaemia.[31]
As fenofibrate possess the same binding mechanism as warfarin in the blood, this combination should be addressed with caution as fenofibrate may potentiate the anticoagulant effect of warfarin.[33] Furthermore, fibrates are contraindicated in patients with active liver disease, severe renal impairment or pre-existing gallbladder disease, as well as in lactating mothers.[31]
Blood-thinning agents
[edit]
Blood-thinning agents are divided into two groups, antiplatelet drugs and anticoagulants. They are indicated to facilitate smooth blood flow within blood vessels by preventing the formation of blood clots and retarding their growth.[34] Blood clots are formed to prevent an injured blood vessel from excessive bleeding by a mechanism called hemostasis. The body has intrinsic mechanisms to dissolve the blood clot as the injury heals. However, it can be dangerous when clots do not dissolve naturally and develop within vessels, also known as thrombosis. Hence, blood-thinning medications can be prescribed to reduce the risk of cardiovascular diseases led by blood clots, such as myocardial infarction (heart attack), ischemic stroke, and venous thromboembolism.[35] Haemorrhage (internal bleeding) is the most prominent side effect of blood-thinning therapy.[36] Concomitant use of drugs that increase the risk of bleeding is not recommended. Meanwhile, patients should receive education about proper management of cuts, bruises and nosebleeds. The agents can be classified according to different mechanisms of action.
Antiplatelet drugs
[edit]Antiplatelet drugs inhibit blood cells called platelets from aggregating to form a clot.
COX-1 inhibitors
[edit]Potent antiplatelet medications that irreversibly inhibit the activity of cyclooxygenase (COX), an enzyme involved in the synthesis of thromboxane A2 (TXA2) which is responsible for platelet activation and aggregation. The major member of this class is aspirin. Some common adverse effects associated with this class of medications include bronchospasm and gastrointestinal disturbances such as dyspepsia and nausea. Therefore, this class of drugs should be used with caution in patients with a history of peptic ulcer disease.[34][37]
ADP receptor inhibitors
[edit]Also known as P2Y12 receptor antagonists, they work by reversibly interacting with the P2Y12 receptor to inhibit the adenosine diphosphate (ADP) receptors on platelets, thus preventing the linkage of platelets by fibrinogen.[34][35] ADP-induced platelet aggregation and activation are hence hindered. Examples include clopidogrel, prasugrel and ticagrelor. Clopidogrel has a common drug interaction with CYP2C19 inhibitors, particularly omeprazole and esomeprazole which are indicated for treatment of peptic ulcer and gastro-oesophageal reflux disease (GERD). As the activation of clopidogrel requires an enzyme called CYP2C19, its inhibitors should be avoided.[38]
Anticoagulants
[edit]Anticoagulants are considered more aggressive than antiplatelet drugs.[34] Anticoagulants work by interfering with various clotting factors to lengthen the time for coagulation. This can be achieved by either reducing the formation of bioactive clotting factors or accelerating the inactivation of clotting factors.

Drugs that inhibit the formation of the vitamin K-dependent clotting factors and hence thrombin, an endogenous protein involved in the coagulation cascade. Vitamin K is an essential cofactor for the synthesis of clotting factors. By inhibiting vitamin K epoxide reductase, an enzyme for activating the vitamin K available in the body, the formation of bioactive clotting factors can be reduced. Although warfarin is commonly prescribed, it exhibits a delayed onset of action, which takes approximately 5 to 7 days to reach its full therapeutic effect.[39] Apart from haemorrhage, jaundice (yellowing of the skin and eyes) is also a common side effect caused by this class of drug.[34]
Direct oral anticoagulants (DOACs)
[edit]DOACs are agents that inhibit the formation of thrombin which is the central effector of coagulation derived from factor Xa. They are categorised into direct thrombin inhibitors and direct factor Xa inhibitors. Direct thrombin inhibitors bind to the active sites of free or clot-bound thrombin to inhibit its effects. Dabigatran etexilate is a common example which has a rapid onset of action.[40] Whereas direct factor Xa inhibitors including apixaban and rivaroxaban directly bind to clotting factor Xa to block its activity, thus inhibiting thrombin formation.[34] DOACs are advantageous over warfarin because of a wider therapeutic window, which indicates safer and more effective use with minimal adverse effects. DOACs also have more stable and predictable anticoagulation effects. Therefore, routine coagulation monitoring is not required.[41]
Indirect thrombin inhibitors
[edit]Indirect thrombin inhibitors bind to antithrombin to enhance the rate of inactivation of clotting factors, indirectly inactivating thrombin through actions on antithrombin.[42] Heparin is a widely used anticoagulant. It is administered intravenously (into a vein) or subcutaneously (below the skin). Heparin can exert an immediate anti-clotting effect which is useful for the treatment of acute symptoms.[36] Besides, heparin therapy is indicated for anticoagulation during pregnancy as it does not cross the placenta and is not associated with fetal malformations.[43][44]
Drug overdose and management
[edit]Cardiovascular agents generally have narrow therapeutic indices, implying that small differences in dose or blood concentration may give rise to adverse drug reactions.[45] Serious acute toxicity may result from accidental, intentional or iatrogenic overdose.[46] Therefore, patients need to be aware of any unusual and serious side effects. Seek immediate medical attention if a drug overdose is suspected.

General management of acute poisoning requires stabilisation of the airway, breathing, and circulation.[citation needed] Supportive treatment to reduce further absorption of the drug is achievable by the administration of activated charcoal.[47] Antidotes can be used to reverse effects of the overdosed medication if the exact poisoning agent is identified. However, only a few antidotes are available for cardiovascular medications.
| Drug class | Medication | Antidote |
|---|---|---|
| Antihypertensive agents | Beta-blockers | High-dose glucagon[48] |
| ACE inhibitors | Naloxone[49] | |
| Calcium channel blockers (CCB) | Levosimendan[50] | |
| Blood-thinning agents | Heparin | Protamine sulfate[51] |
Table 1: antidotes for cardiovascular agent overdose
For patients taking antihyperlipidemic agents, liver function tests have to be conducted before and during the therapy to monitor the elevation of liver enzymes which may result in hepatotoxicity, especially for those undergoing statin therapy.[52] For patients taking blood-thinners, signs of severe bleeding should be monitored. The effect of aspirin can be life-threatening if taken over 150 mg/kg of body weight.[47] The medication should be discontinued at the first sign of excessive bleeding.
References
[edit]- ^ a b Siest, G; Jeannesson, E; Berrahmoune, H; Maumus, S; Marteau, J-B; Mohr, S; Visvikis, S (2004-10-01). "Pharmacogenomics and drug response in cardiovascular disorders". Pharmacogenomics. 5 (7): 779–802. doi:10.1517/14622416.5.7.779. ISSN 1462-2416. PMID 15469403.
- ^ a b Laurent, Stéphane (2017-10-27). "Antihypertensive drugs". Pharmacological Research. 124 (1): 116–125. doi:10.1016/j.phrs.2017.07.026. ISSN 1043-6618. PMID 28780421.
- ^ Roush, George C.; Sica, Domenic A. (2016-04-05). "Diuretics for Hypertension: A Review and Update". American Journal of Hypertension. 29 (10): 1130–1137. doi:10.1093/ajh/hpw030. ISSN 0895-7061. PMID 27048970.
- ^ a b c d Zinkovsky, Daniel (2021-01-01), Ray, Sidhartha D. (ed.), Chapter 20 - Side effects, ADRs & ADEs of diuretics, Side Effects of Drugs Annual, vol. 43, Elsevier, pp. 259–265, doi:10.1016/bs.seda.2021.09.018, ISBN 978-0-12-824119-6, retrieved 2024-04-03
- ^ Duncan, Richards; Aronson, Jeffrey (2005-01-01), "Diuretics: Potassium-sparing diuretics (amiloride and triamterene): BNF 2.3.2 and 2.2.4", Oxford Handbook of Practical Drug Therapy, Oxford University Press, doi:10.1093/med/1.1.med-9780198530077-div1-72 (inactive 1 November 2024), retrieved 2024-04-03
{{citation}}: CS1 maint: DOI inactive as of November 2024 (link) - ^ a b c d Brown, Nancy J.; Vaughan, Douglas E. (1998-04-14). "Angiotensin-Converting Enzyme Inhibitors". Circulation. 97 (14): 1411–1420. doi:10.1161/01.cir.97.14.1411. ISSN 0009-7322. PMID 9577953.
- ^ a b c d Montoro de Francisco, Ana; García-Luque, Amelia; Fernández, Manuel; Puerro, Miguel (2012-11-15). "Side Effects of Angiotensin Converting Enzyme Inhibitors and Angiotensin II Receptor Antagonists: Are We Facing a New Syndrome". The American Journal of Cardiology. 110 (10): 1552–1553. doi:10.1016/j.amjcard.2012.08.027. ISSN 0002-9149. PMID 23102232.
- ^ a b c d e Matchar, David B.; McCrory, Douglas C.; Orlando, Lori A.; Patel, Manesh R.; Patel, Uptal D.; Patwardhan, Meenal B.; Powers, Benjamin; Samsa, Gregory P.; Gray, Rebecca N. (2008-01-01). "Systematic Review: Comparative Effectiveness of Angiotensin-Converting Enzyme Inhibitors and Angiotensin II Receptor Blockers for Treating Essential Hypertension". Annals of Internal Medicine. 148 (1): 16–29. doi:10.7326/0003-4819-148-1-200801010-00189. ISSN 0003-4819. PMID 17984484.
- ^ Barreras, Amy; Gurk-Turner, Cheryle (2017-12-11). "Angiotensin Ii Receptor Blockers". Baylor University Medical Center Proceedings. 16 (1): 123–126. doi:10.1080/08998280.2003.11927893. ISSN 0899-8280. PMC 1200815. PMID 16278727.
- ^ Elliott, William J.; Ram, C. Venkata S. (2011-07-27). "Calcium Channel Blockers". The Journal of Clinical Hypertension. 13 (9): 687–689. doi:10.1111/j.1751-7176.2011.00513.x. ISSN 1524-6175. PMC 8108866. PMID 21896151.
- ^ Stern, Simon (2018-05-24). "The Effects of Calcium Channel Blocker Drugs on Gingival Vasculature". doi:10.26226/morressier.5ac383182afeeb00097a402c. Retrieved 2024-04-03.
{{cite journal}}: Cite journal requires|journal=(help) - ^ a b Sirker, A.; Missouris, C. G.; MacGregor, G. A. (2001-10-12). "Dihydropyridine calcium channel blockers and peripheral side effects". Journal of Human Hypertension. 15 (10): 745–746. doi:10.1038/sj.jhh.1001248. ISSN 1476-5527. PMID 11607807.
- ^ a b c d Sica, Domenic A. (2007-01-31). "Pharmacotherapy Review: Calcium Channel Blockers". The Journal of Clinical Hypertension. 8 (1): 53–56. doi:10.1111/j.1524-6175.2005.04140.x. ISSN 1524-6175. PMC 8109625. PMID 16407690.
- ^ Wiysonge, Charles S; Bradley, Hazel A; Volmink, Jimmy; Mayosi, Bongani M; Opie, Lionel H (2017-01-20). "Beta-blockers for hypertension". Cochrane Database of Systematic Reviews. 2017 (1): CD002003. doi:10.1002/14651858.cd002003.pub5. ISSN 1465-1858. PMC 5369873. PMID 28107561.
- ^ a b Bain, Amie (2018-10-02). "Beta-blocker use in cardiovascular disease". British Journal of Cardiac Nursing. 13 (10): 491–497. doi:10.12968/bjca.2018.13.10.491. ISSN 1749-6403.
- ^ a b Bangalore, Sripal; Messerli, Franz H.; Kostis, John B.; Pepine, Carl J. (2007-08-14). "Cardiovascular Protection Using Beta-Blockers". Journal of the American College of Cardiology. 50 (7): 563–572. doi:10.1016/j.jacc.2007.04.060. ISSN 0735-1097. PMID 17692739.
- ^ Berberich, Amanda J; Hegele, Robert A (2022-07-13). "A Modern Approach to Dyslipidemia". Endocrine Reviews. 43 (4): 611–653. doi:10.1210/endrev/bnab037. ISSN 0163-769X. PMC 9277652. PMID 34676866.
- ^ a b c Stancu, Camelia; Sima, Anca (2007-05-01). "Statins: mechanism of action and effects". J Cell Mol Med. 5 (4): 378–387. doi:10.1111/j.1582-4934.2001.tb00172.x. ISSN 1582-1838. PMC 6740083. PMID 12067471.
- ^ a b c d Grundy, Scott M.; Stone, Neil J.; Bailey, Alison L.; Beam, Craig; Birtcher, Kim K.; Blumenthal, Roger S.; Braun, Lynne T.; de Ferranti, Sarah; Faiella-Tommasino, Joseph; Forman, Daniel E.; Goldberg, Ronald; Heidenreich, Paul A.; Hlatky, Mark A.; Jones, Daniel W.; Lloyd-Jones, Donald (2019-06-25). "2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the Management of Blood Cholesterol". Journal of the American College of Cardiology. 73 (24): e285–e350. doi:10.1016/j.jacc.2018.11.003. hdl:20.500.12749/1738. PMID 30423393.
- ^ a b c Ruscica, Massimiliano; Ferri, Nicola; Banach, Maciej; Sirtori, Cesare R; Corsini, Alberto (2023-01-18). "Side effects of statins: from pathophysiology and epidemiology to diagnostic and therapeutic implications". Cardiovascular Research. 118 (17): 3288–3304. doi:10.1093/cvr/cvac020. ISSN 0008-6363. PMID 35238338.
- ^ Dicken, Weston; Mehta, Anurag; Karagiannis, Angelos; Jain, Vardhmaan; Vavuranakis, Michael; Sperling, Laurence; Cassimatis, Dimitri (2022-11-30). "Statin associated muscle symptoms: An update and review". Progress in Cardiovascular Diseases. 75: 40–48. doi:10.1016/j.pcad.2022.11.010. PMID 36400232.
- ^ Newman, Connie B.; Preiss, David; Tobert, Jonathan A.; Jacobson, Terry A.; Page, Robert L.; Goldstein, Larry B.; Chin, Clifford; Tannock, Lisa R.; Miller, Michael; Raghuveer, Geetha; Duell, P. Barton; Brinton, Eliot A.; Pollak, Amy; Braun, Lynne T.; Welty, Francine K. (2018-12-10). "Statin Safety and Associated Adverse Events: A Scientific Statement From the American Heart Association". Arteriosclerosis, Thrombosis, and Vascular Biology. 39 (2): e38–e81. doi:10.1161/ATV.0000000000000073. ISSN 1079-5642. PMID 30580575.
- ^ a b c d e f Mach, François; Baigent, Colin; Catapano, Alberico L; Koskinas, Konstantinos C; Casula, Manuela; Badimon, Lina; Chapman, M John; De Backer, Guy G; Delgado, Victoria; Ference, Brian A; Graham, Ian M; Halliday, Alison; Landmesser, Ulf; Mihaylova, Borislava; Pedersen, Terje R (2020-01-01). "2019 ESC/EAS Guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk". European Heart Journal. 41 (1): 111–188. doi:10.1093/eurheartj/ehz455. hdl:2434/706264. ISSN 0195-668X. PMID 31504418.
- ^ Balasubramanian, Rajkapoor; Maideen, Naina M.P. (2021-07-15). "HMG-CoA Reductase Inhibitors (Statins) and their Drug Interactions Involving CYP Enzymes, P-glycoprotein and OATP Transporters-An Overview". Current Drug Metabolism. 22 (5): 328–341. doi:10.2174/1389200222666210114122729. PMID 33459228.
- ^ a b Team C by Mimso. Ezetimibe Sandoz. [cited 2024 Mar 11]. Available from: https://www.mims.com/hongkong/drug/info/ezetimibe%20sandoz
- ^ Lee, Jeongmin; Lee, Seung-Hwan (2023-11-01). "Expanding the therapeutic landscape: ezetimibe as non-statin therapy for dyslipidemia". The Korean Journal of Internal Medicine. 38 (6): 797–809. doi:10.3904/kjim.2023.243. ISSN 1226-3303. PMC 10636547. PMID 37866817.
- ^ Duan, Yajun; Gong, Ke; Xu, Suowen; Zhang, Feng; Meng, Xianshe; Han, Jihong (2022-08-02). "Regulation of cholesterol homeostasis in health and diseases: from mechanisms to targeted therapeutics". Signal Transduction and Targeted Therapy. 7 (1): 265. doi:10.1038/s41392-022-01125-5. ISSN 2059-3635. PMC 9344793. PMID 35918332.
- ^ Kanagalingam, Tharsan; Lazarte, Julieta; Wong, David K.H.; Hegele, Robert A. (2020-09-28). "Liver Injury Associated With Ezetimibe Monotherapy". CJC Open. 3 (2): 195–197. doi:10.1016/j.cjco.2020.09.018. PMC 7893188. PMID 33644733.
- ^ a b Team C by Mimso. Ezetimibe. [cited 2024 Mar 11]. Available from: https://www.mims.com/hongkong/drug/info/ezetimibe?mtype=generic
- ^ Kim, Nam Hoon; Kim, Sin Gon (2020). "Fibrates Revisited: Potential Role in Cardiovascular Risk Reduction". Diabetes & Metabolism Journal. 44 (2): 213–221. doi:10.4093/dmj.2020.0001. ISSN 2233-6079. PMC 7188966. PMID 32347023.
- ^ a b c Team C by Mimso. Gemfibrozil. [cited 2024 Mar 11]. Available from: https://www.mims.com/hongkong/drug/info/gemfibrozil?mtype=generic
- ^ Brinton, Eliot A.; Pulipati, Vishnu Priya (2023), Jenkins, Alicia J.; Toth, Peter P. (eds.), "Fibrate Therapy: Impact on Dyslipidemia and Cardiovascular Events in Patients with Diabetes Mellitus Type 2", Lipoproteins in Diabetes Mellitus, Cham: Springer International Publishing, pp. 637–679, doi:10.1007/978-3-031-26681-2_24, ISBN 978-3-031-26680-5
- ^ Gu, Jiali; Liu, Hongrui; Huang, Xiyao; Ma, Yanxuan; Zhang, Liang (2023-05-01). "Investigation of the separate and simultaneous bindings of warfarin and fenofibrate to bovine serum albumin". International Journal of Biological Macromolecules. 236: 123978. doi:10.1016/j.ijbiomac.2023.123978. PMID 36906198.
- ^ a b c d e f Drug Office (September 2014). "Oral Anticoagulants and Antiplatelet Drugs".
- ^ a b Born, Gustav; Patrono, Carlo (2006). "Antiplatelet drugs". British Journal of Pharmacology. 147 (Suppl 1): S241-51. doi:10.1038/sj.bjp.0706401. PMC 1760725. PMID 16402110.
- ^ a b "What are blood thinners (anti-clotting medication) and how are they used?", InformedHealth.org [Internet], Institute for Quality and Efficiency in Health Care (IQWiG), 2017-10-05, retrieved 2024-03-10
- ^ Iqbal, Arshad Muhammad; Lopez, Richard A.; Hai, Ofek (2024), "Antiplatelet Medications", StatPearls, Treasure Island (FL): StatPearls Publishing, PMID 30725747, retrieved 2024-03-10
- ^ Bates, Eric R.; Lau, Wei C.; Angiolillo, Dominick J. (2011). "Clopidogrel–Drug Interactions". Journal of the American College of Cardiology. 57 (11): 1251–1263. doi:10.1016/j.jacc.2010.11.024. PMID 21392639.
- ^ Singh, Shejal; Singh, Vaibhav; Patel, Shobhit (2024-03-08). "Cosmeceuticals; The Fusion of Cosmetics and Pharmaceuticals". Journal of Community Pharmacy Practice (42): 16–27. doi:10.55529/jcpp.42.16.27. ISSN 2799-1199.
- ^ Baetz, Brooke E.; Spinler, Sarah A. (2008). "Dabigatran Etexilate: An Oral Direct Thrombin Inhibitor for Prophylaxis and Treatment of Thromboembolic Diseases". Pharmacotherapy: The Journal of Human Pharmacology and Drug Therapy. 28 (11): 1354–1373. doi:10.1592/phco.28.11.1354. PMID 18956996.
- ^ Zeng, Shan; Zheng, Yuxiang; Jiang, Jingzhou; Ma, Jianyong; Zhu, Wengen; Cai, Xingming (2022-06-24). "Effectiveness and Safety of DOACs vs. Warfarin in Patients With Atrial Fibrillation and Frailty: A Systematic Review and Meta-Analysis". Frontiers in Cardiovascular Medicine. 9. doi:10.3389/fcvm.2022.907197. ISSN 2297-055X. PMC 9263568. PMID 35811719.
- ^ Alquwaizani, Mohammed; Buckley, Leo; Adams, Christopher; Fanikos, John (2013). "Anticoagulants: A Review of the Pharmacology, Dosing, and Complications". Current Emergency and Hospital Medicine Reports. 1 (2): 83–97. doi:10.1007/s40138-013-0014-6. PMC 3654192. PMID 23687625.
- ^ Schleich, J. M.; Laurent, M.; Almange, C. (2001). "Heparin therapy in pregnant women". Archives des Maladies du Coeur et des Vaisseaux. 94 (11 Suppl): 1291–1500. PMID 11794972.
- ^ Ginsberg, J. S.; Kowalchuk, G.; Hirsh, J.; Brill-Edwards, P.; Burrows, R. (1989). "Heparin therapy during pregnancy. Risks to the fetus and mother". Archives of Internal Medicine. 149 (10): 2233–2236. doi:10.1001/archinte.1989.00390100059015. PMID 2802889.
- ^ Blix, Hege S.; Viktil, Kirsten K.; Moger, Tron A.; Reikvam, Aasmund (2010). "Drugs with narrow therapeutic index as indicators in the risk management of hospitalised patients". Pharmacy Practice. 8 (1): 50–55. doi:10.4321/s1886-36552010000100006. PMC 4140577. PMID 25152793.
- ^ Volans, Glyn N.; Holt, David W.; Ali, Christine (1984). "Management of cardiac drug overdose". Resuscitation. 11 (3–4): 207–216. doi:10.1016/0300-9572(84)90018-2.
- ^ a b Dargan, P. I. (2002). "An evidence based flowchart to guide the management of acute salicylate (Aspirin) overdose". Emergency Medicine Journal. 19 (3): 206–209. doi:10.1136/emj.19.3.206. PMC 1725844. PMID 11971828.
- ^ Shepherd, Greene (2006). "Treatment of poisoning caused by β-adrenergic and calcium-channel blockers". American Journal of Health-System Pharmacy. 63 (19): 1828–1835. doi:10.2146/ajhp060041. PMID 16990629.. (Erratum: PMID 16990629)
- ^ Trivedi, Vatsal; Glezerson, Bryan A.; Chaudhuri, Dipayan; Davidson, Morgan; Douflé, Ghislaine (2020). "Naloxone as an antidote for angiotensin converting enzyme inhibitor poisoning: A case report". Canadian Journal of Anesthesia. 67 (10): 1442–1443. doi:10.1007/s12630-020-01680-x. PMC 7223556. PMID 32342351.
- ^ Varpula, Tero; Rapola, Janne; Sallisalmi, Marko; Kurola, Jouni (2009). "Treatment of Serious Calcium Channel Blocker Overdose with Levosimendan, a Calcium Sensitizer". Anesthesia & Analgesia. 108 (3): 790–792. doi:10.1213/ane.0b013e3181931737. PMID 19224785.
- ^ Sokolowska, Emilia; Kalaska, Bartlomiej; Miklosz, Joanna; Mogielnicki, Andrzej (2016). "The toxicology of heparin reversal with protamine: Past, present and future". Expert Opinion on Drug Metabolism & Toxicology. 12 (8): 897–909. doi:10.1080/17425255.2016.1194395. PMID 27223896.
- ^ Bhardwaj, Sidharth S.; Chalasani, Naga (2007). "Lipid-Lowering Agents That Cause Drug-Induced Hepatotoxicity". Clinics in Liver Disease. 11 (3): 597–613. doi:10.1016/j.cld.2007.06.010. PMC 2048990. PMID 17723922.
