Pemetrexed
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Alimta, Pemfexy, Ciambra, others |
| AHFS/Drugs.com | Monograph |
| License data |
|
| Pregnancy category |
|
| Routes of administration | Intravenous |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | NA |
| Protein binding | 81% |
| Metabolism | Negligible |
| Elimination half-life | 3.5 hours |
| Excretion | Kidney |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.205.735 |
| Chemical and physical data | |
| Formula | C20H21N5O6 |
| Molar mass | 427.417 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Pemetrexed, sold under the brand name Alimta among others, is a chemotherapy medication for the treatment of pleural mesothelioma and non-small cell lung cancer (NSCLC).[2]
It is available as a generic medication.[6][7]
Medical use
[edit]In February 2004, the U.S. Food and Drug Administration (FDA) approved pemetrexed for treatment of malignant pleural mesothelioma, a type of tumor of the mesothelium, the thin layer of tissue that covers many of the internal organs, in combination with cisplatin[8] for patients whose disease is either unresectable or who are not otherwise candidates for curative surgery.[9] In September 2008, the FDA granted approval as a first-line treatment, in combination with cisplatin, against locally advanced and metastatic non-small cell lung cancer (NSCLC) in patients with non-squamous histology.[10][11][2]
Carboplatin
[edit]Pemetrexed is also recommended in combination with carboplatin and pembrolizumab for the first-line treatment of advanced non-small cell lung cancer.[12][13] However, the relative efficacy or toxicity of pemetrexed-cisplatin versus pemetrexed-carboplatin has not been established beyond what is generally thought about cisplatin or carboplatin doublet drug therapy.[14]
Supplementation
[edit]Patients are recommended to take folic acid and vitamin B12 supplement even if levels are normal when they are on pemetrexed therapy.[15][2] (In clinical trials for mesothelioma, folic acid and B12 supplementation reduced the frequency of adverse events.) It is also recommended for patients to be on a glucocorticoid (e.g. dexamethasone) on the day prior, day of, and day after pemetrexed infusion to avoid skin rashes.[2]
Drug interaction
[edit]The administration of cisplatin and vitamin B12 concomitantly does not modify the pharmacokinetics of the pemetrexed. It was recently shown that pemetrexed may play a role in cisplatin resistance in lung cancer by increasing the expression of Orai3 calcium channels as well as the expression of certain ABC transporters like MDR1 and MRP-5 responsible for cisplatin efflux and therefore a reduction of the effect of cisplatin[16] As current therapies are based on the co-administration of pemetrexed and cisplatin, there may be interactions between pemetrexed and cisplatin, including a reduction in the therapeutic effects of cisplatin caused by pemetrexed.
Mechanism of action
[edit]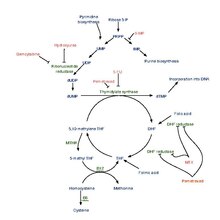
Pemetrexed is chemically similar to folic acid and is in the class of chemotherapy drugs called folate antimetabolites. It works by inhibiting three enzymes used in purine and pyrimidine synthesis—thymidylate synthase (TS), dihydrofolate reductase (DHFR), and glycinamide ribonucleotide formyltransferase[17][18] (GARFT). By inhibiting the formation of precursor purine and pyrimidine nucleotides, pemetrexed prevents the formation of DNA and RNA, which are required for the growth and survival of both normal cells and cancer cells.
Society and culture
[edit]Economics
[edit]In the United States, as of 2015[update], each vial of the medication costs between US$2,623 and US$3,100.[19]
Brand names
[edit]In February 2020, Pemfexy was approved for use in the United States.[20]
Research
[edit]A Phase III study showed benefits of maintenance use of pemetrexed for non-squamous NSCLC.[21] Activity has been shown in malignant peritoneal mesothelioma.[22]
References
[edit]- ^ "Prescription medicines: registration of new generic medicines and biosimilar medicines, 2017". Therapeutic Goods Administration (TGA). 21 June 2022. Retrieved 30 March 2024.
- ^ a b c d e "Alimta- pemetrexed disodium injection, powder, lyophilized, for solution". DailyMed. 25 March 2020. Archived from the original on 20 October 2020. Retrieved 20 October 2020.
- ^ "Pemfexy- pemetrexed injection". DailyMed. 20 January 2022. Archived from the original on 21 May 2022. Retrieved 20 May 2022.
- ^ "Alimta EPAR". European Medicines Agency. 17 September 2018. Archived from the original on 18 January 2021. Retrieved 14 October 2022.
- ^ "Pemetrexed Baxter: Pending EC decision". European Medicines Agency. 14 October 2022. Archived from the original on 14 October 2022. Retrieved 14 October 2022.
- ^ "2022 First Generic Drug Approvals". U.S. Food and Drug Administration (FDA). 3 March 2023. Archived from the original on 30 June 2023. Retrieved 30 June 2023.
- ^ "Competitive Generic Therapy Approvals". U.S. Food and Drug Administration (FDA). 29 June 2023. Archived from the original on 29 June 2023. Retrieved 29 June 2023.
- ^ Manegold C (August 2003). "Pemetrexed (Alimta, MTA, multitargeted antifolate, LY231514) for malignant pleural mesothelioma". Seminars in Oncology. 30 (4 Suppl 10): 32–36. doi:10.1016/S0093-7754(03)00283-5. PMID 12917819.
- ^ National Cancer Institute: FDA Approval for Pemetrexed Disodium Archived 6 April 2015 at the Wayback Machine
- ^ Cohen MH, Justice R, Pazdur R (September 2009). "Approval summary: pemetrexed in the initial treatment of advanced/metastatic non-small cell lung cancer". The Oncologist. 14 (9): 930–935. doi:10.1634/theoncologist.2009-0092. PMID 19737998. S2CID 28209589.
- ^ Rossi A, Ricciardi S, Maione P, de Marinis F, Gridelli C (November 2009). "Pemetrexed in the treatment of advanced non-squamous lung cancer". Lung Cancer. 66 (2): 141–149. doi:10.1016/j.lungcan.2009.06.006. PMID 19577816.
- ^ Ettinger DS, et al. "Non-small Cell Lung Cancer V.1.2007" (PDF). NCCN Clinical Practice Guidelines in Oncology. National Comprehensive Cancer Network (NCCN). Archived from the original (PDF) on 23 March 2007.
- ^ Gandhi L, Rodríguez-Abreu D, Gadgeel S, Esteban E, Felip E, De Angelis F, et al. (KEYNOTE-189 Investigators) (May 2018). "Pembrolizumab plus Chemotherapy in Metastatic Non-Small-Cell Lung Cancer". The New England Journal of Medicine. 378 (22): 2078–2092. doi:10.1056/NEJMoa1801005. hdl:10138/298862. PMID 29658856.
- ^ Azzoli CG, Kris MG, Pfister DG (June 2007). "Cisplatin versus carboplatin for patients with metastatic non-small-cell lung cancer--an old rivalry renewed". Journal of the National Cancer Institute. 99 (11): 828–829. doi:10.1093/jnci/djk222. PMID 17551137.
- ^ Hazarika M, White RM, Johnson JR, Pazdur R (2004). "FDA drug approval summaries: pemetrexed (Alimta)". The Oncologist. 9 (5): 482–488. doi:10.1634/theoncologist.9-5-482. PMID 15477632. S2CID 11444611.
- ^ Daoudi, Redoane (26 September 2024). "Characterization and implication of the Orai3 channel and ABC type transporters in the phenomenon of chemoresistance to cisplatin and pemetrexed in lung cancer". bioRxiv 10.1101/2024.09.22.613742.
- ^ McLeod HL, Cassidy J, Powrie RH, Priest DG, Zorbas MA, Synold TW, et al. (July 2000). "Pharmacokinetic and pharmacodynamic evaluation of the glycinamide ribonucleotide formyltransferase inhibitor AG2034". Clinical Cancer Research. 6 (7): 2677–2684. PMID 10914709. Archived from the original on 28 July 2009. Retrieved 2 December 2008.
- ^ Avendano C, Menendez JC (April 2008). Medicinal Chemistry of Anticancer Drugs. Amsterdam: Elsevier. p. 37. ISBN 978-0-444-52824-7.
- ^ Langreth R (29 June 2016). "Decoding Big Pharma's Secret Drug Pricing Practices". Bloomberg. Archived from the original on 13 July 2016. Retrieved 15 July 2016.
- ^ "Pemfexy: FDA-Approved Drugs". U.S. Food and Drug Administration (FDA). Archived from the original on 19 October 2020. Retrieved 13 February 2020.
- ^ Belani CP, Brodowicz T, Ciuleanu T, Kim JH, Krzakowski M, Laack E, et al. (June 2009). "Maintenance pemetrexed (Pem) plus best supportive care (BSC) versus placebo (Plac) plus BSC: A randomized phase III study in advanced non-small cell lung cancer (NSCLC)". Journal of Clinical Oncology. 27 (18 suppl). American Society of Clinical Oncology: CRA8000. doi:10.1200/jco.2009.27.18_suppl.cra8000. Archived from the original on 16 June 2009. Retrieved 22 July 2009.
- ^ Carteni G, Manegold C, Garcia GM, Siena S, Zielinski CC, Amadori D, et al. (May 2009). "Malignant peritoneal mesothelioma-Results from the International Expanded Access Program using pemetrexed alone or in combination with a platinum agent". Lung Cancer. 64 (2): 211–218. doi:10.1016/j.lungcan.2008.08.013. PMID 19042053.
External links
[edit]- "Pemetrexed disodium". NCI Drug Dictionary. National Cancer Institute.
- "Pemetrexed disodium". National Cancer Institute. 5 October 2006.
