Aviculin
Appearance
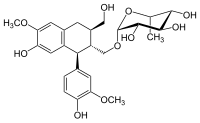
| |
| Names | |
|---|---|
| IUPAC name
[(1S,2R,3R)-7-Hydroxy-1-(4-hydroxy-3-methoxyphenyl)-3-(hydroxymethyl)-6-methoxy-1,2,3,4-tetrahydronaphthalen-2-yl]methyl α-L-rhamnopyranoside
| |
| Systematic IUPAC name
(2R,3R,4R,5R,6S)-2-{[(1S,2R,3R)-7-Hydroxy-1-(4-hydroxy-3-methoxyphenyl)-3-(hydroxymethyl)-6-methoxy-1,2,3,4-tetrahydronaphthalen-2-yl]methoxy}-6-methyloxane-3,4,5-triol | |
| Other names | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C26H34O10 | |
| Molar mass | 506.54 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Aviculin is a lignan. It is bio-active isolate of Pseudocydonia sinensis [2] or Polygonum aviculare.[3]
References
[edit]- ^ "KNApSAcK Metabolite Information - C00031615". www.knapsackfamily.com.
- ^ Gao H, Wu L, Kuroyanagi M, Harada K, Kawahara N, Nakane T, Umehara K, Hirasawa A, Nakamura Y (November 2003). "Antitumor-promoting constituents from Chaenomeles sinensis KOEHNE and their activities in JB6 mouse epidermal cells". Chem. Pharm. Bull. 51 (11): 1318–21. doi:10.1248/cpb.51.1318. PMID 14600382.
- ^ Ja Kim, Hyoung (1994). "A Novel Lignan and Flavonoids from Polygonum aviculare". Journal of Natural Products. 57 (5): 581–586. doi:10.1021/np50107a003.
