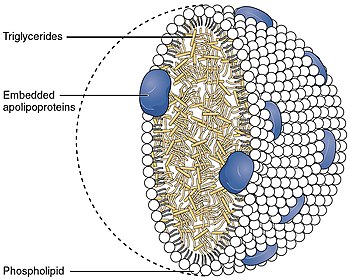
Apolipoprotein C-III
Apolipoprotein C-III also known as apo-CIII, and apolipoprotein C3, is a protein that in humans is encoded by the APOC3 gene. Apo-CIII is secreted by the liver as well as the small intestine, and is found on triglyceride-rich lipoproteins such as chylomicrons, very low density lipoprotein (VLDL), and remnant cholesterol.[5]
Structure
[edit]| ApoC-III | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| Symbol | ApoC-III | ||||||||
| Pfam | PF05778 | ||||||||
| InterPro | IPR008403 | ||||||||
| |||||||||
ApoC-III is a relatively small protein containing 79 amino acids that can be glycosylated at threonine-74.[6] The most abundant glycoforms are characterized by an O-linked disaccharide galactose linked to N-acetylgalactosamine (Gal–GalNAc), further modified with up to two sialic acid residues. Less abundant glycoforms are characterized by more complex and fucosylated glycan moieties.[7]
Function
[edit]APOC3 inhibits lipoprotein lipase and hepatic lipase; it is thought to inhibit hepatic uptake[8] of triglyceride-rich particles. The APOA1, APOC3 and APOA4 genes are closely linked in both rat and human genomes. The A-I and A-IV genes are transcribed from the same strand, while the A-1 and C-III genes are convergently transcribed. An increase in apoC-III levels induces the development of hypertriglyceridemia. Recent evidence suggests an intracellular role for Apo-CIII in promoting the assembly and secretion of triglyceride-rich VLDL particles from hepatic cells under lipid-rich conditions.[9] However, two naturally occurring point mutations in human apoC3 coding sequence, namely Ala23Thr and Lys58Glu have been shown to abolish the intracellular assembly and secretion of triglyceride-rich VLDL particles from hepatic cells.[10][11]
Clinical significance
[edit]Overexpression of Apo-CIII in humans contributes to atherosclerosis.[5] Two novel susceptibility haplotypes (specifically, P2-S2-X1 and P1-S2-X1) have been discovered in ApoAI-CIII-AIV gene cluster on chromosome 11q23; these confer approximately threefold higher risk of coronary heart disease in normal[12] as well as non-insulin diabetes mellitus.[13] In persons with type 2 diabetes, elevated plasma Apo-CIII is associated with higher plasma triglycerides and greater coronary artery calcification (a measure of subclinical atherosclerosis).[14]
Apo-CIII delays the catabolism of triglyceride rich particles. HDL cholesterol particles that bear Apo-CIII are associated with increased, rather than decreased, risk for coronary heart disease.[15]
Elevations of Apo-CIII associated with single-nucleotide polymorphisms found in genetic variation studies may predispose patients to non-alcoholic fatty liver disease,[16] although the association has been questioned[17] and may be specific to certain ethnicities[18][19] or to people without central obesity.[20]
Antisense oligonucleotides that bind APOC3 mRNA and prevent its translation have been found to reduce episodes of acute pancreatitis in people with familial chylomicronemia syndrome and lower their triglyceride levels in blood. Adverse effects include thrombocytopenia, which may be prevented by targeting hepatocellular APOC3 expression with a chemically modified oligonucleotide.[21]
Interactive pathway map
[edit]Click on genes, proteins and metabolites below to link to respective articles. [§ 1]
- ^ The interactive pathway map can be edited at WikiPathways: "Statin_Pathway_WP430".
Apolipoprotein CIII and HDL
[edit]Apolipoprotein CIII is also found on HDL particles. Formation of APOCIII-containing HDL is not a matter of simple binding of APOCII to pre-existing HDL particles but requires the lipid transported ABCA1 in a fashion similar to APOA1-containing HDL.[22] Accumulation of APOCIII on HDL is important for the maintenance of plasma triglyceride homeostasis since it prevents excessive amount of APOCIII on VLDL and other triglyceride rich lipoproteins, thus preventing APOCIII-mediated inhibition of LpL and the subsequent hydrolysis of plasma triglycerides. This may explain the hypertriglyceridemia associated with ABCA1-deficiency in patients with Tangier's disease.
References
[edit]- ^ a b c GRCh38: Ensembl release 89: ENSG00000110245 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000032081 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ a b Khetarpal SA, Zeng X, Millar JS, Vitali C, Somasundara AVH, Zanoni P, Landro JA, Barucci N, Zavadoski WJ, Sun Z, de Haard H, Toth IV, Peloso GM, Natarajan P, Cuchel M, Lund-Katz S, Phillips MC, Tall AR, Kathiresan S, DaSilva-Jardine P, Yates NA, Rader D (2017). "A human APOC3 missense variant and monoclonal antibody accelerate apoC-III clearance and lower triglyceride-rich lipoprotein levels". Nature Medicine. 23 (9): 1086–1094. doi:10.1038/nm.4390. PMC 5669375. PMID 28825717.
- ^ Vaith P, Assmann G, Uhlenbruck G (Jun 1978). "Characterization of the oligosaccharide side chain of apolipoprotein C-III from human plasma very low density lipoproteins". Biochimica et Biophysica Acta (BBA) - General Subjects. 541 (2): 234–40. doi:10.1016/0304-4165(78)90396-3. PMID 208636.
- ^ Nicolardi S, van der Burgt YE, Dragan I, Hensbergen PJ, Deelder AM (May 2013). "Identification of new apolipoprotein-CIII glycoforms with ultrahigh resolution MALDI-FTICR mass spectrometry of human sera". Journal of Proteome Research. 12 (5): 2260–68. doi:10.1021/pr400136p. PMID 23527852. S2CID 19296928.
- ^ Mendivil CO, Zheng C, Furtado J, Lel J, Sacks FM (Feb 2010). "Metabolism of very-low-density lipoprotein and low-density lipoprotein containing apolipoprotein C-III and not other small apolipoproteins". Arteriosclerosis, Thrombosis, and Vascular Biology. 30 (2): 239–45. doi:10.1161/ATVBAHA.109.197830. PMC 2818784. PMID 19910636.
- ^ Sundaram M, Zhong S, Bou Khalil M, Links PH, Zhao Y, Iqbal J, Hussain MM, Parks RJ, Wang Y, Yao Z (Jan 2010). "Expression of apolipoprotein C-III in McA-RH7777 cells enhances VLDL assembly and secretion under lipid-rich conditions". Journal of Lipid Research. 51 (1): 150–161. doi:10.1194/M900346-JLR200. PMC 2789775. PMID 19622837.
- ^ Sundaram M, Zhong S, Bou Khalil M, Zhou H, Jiang ZG, Zhao Y, Iqbal J, Hussain MM, Figeys D, Wang Y, Yao Z (Jun 2010). "Functional analysis of the missense APOC3 mutation Ala23Thr associated with human hypotriglyceridemia". Journal of Lipid Research. 51 (6): 1524–1534. doi:10.1194/jlr.M005108. PMC 3035516. PMID 20097930.
- ^ Qin W, Sundaram M, Wang Y, Zhou H, Zhong S, Chang CC, Manhas S, Yao EF, Parks RJ, McFie PJ, Stone SJ, Jiang ZG, Wang C, Figeys D, Jia W, Yao Z (Aug 2011). "Missense mutation in APOC3 within the C-terminal lipid binding domain of human ApoC-III results in impaired assembly and secretion of triacylglycerol-rich very low density lipoproteins: evidence that ApoC-III plays a major role in the formation of lipid precursors within the microsomal lumen". The Journal of Biological Chemistry. 286 (31): 27769–27780. doi:10.1074/jbc.M110.203679. PMC 3149367. PMID 21676879.
- ^ Singh P, Singh M, Kaur TP, Grewal SS (Nov 2008). "A novel haplotype in ApoAI-CIII-AIV gene region is detrimental to Northwest Indians with coronary heart disease". International Journal of Cardiology. 130 (3): e93–5. doi:10.1016/j.ijcard.2007.07.029. PMID 17825930.
- ^ Singh P, Singh M, Gaur S, Kaur T (Jun 2007). "The ApoAI-CIII-AIV gene cluster and its relation to lipid levels in type 2 diabetes mellitus and coronary heart disease: determination of a novel susceptible haplotype". Diabetes & Vascular Disease Research. 4 (2): 124–29. doi:10.3132/dvdr.2007.030. PMID 17654446. S2CID 23793589.

- ^ Qamar A, Khetarpal SA, Khera AV, Qasim A, Rader DJ, Reilly MP (2015). "Plasma apolipoprotein C-III levels, triglycerides, and coronary artery calcification in type 2 diabetics". Arteriosclerosis, Thrombosis, and Vascular Biology. 35 (8): 1880–1888. doi:10.1161/ATVBAHA.115.305415. PMC 4556282. PMID 26069232.
- ^ Sacks FM, Zheng C, Cohn JS (2011). "Complexities of plasma apolipoprotein C-III metabolism". Journal of Lipid Research. 52 (6): 1067–1070. doi:10.1194/jlr.E015701. PMC 3090227. PMID 21421846.
- ^ Petersen RF, Dufour S, Hariri A, Nelson-Williams C, Foo JN, Zhang XM, Dzjura J, Lifton RP, Shulman GI (2010). "Apoliprotein C3 Gene Variants in Nonalcoholic Fatty Liver Disease". New England Journal of Medicine. 362 (12): 1082–1089. doi:10.1056/NEJMoa0907295. PMC 2976042. PMID 20335584.
- ^ Richart C, August T, Terra X (2010). "Correspondence: Apolipoprotein C3 Gene Variants in Nonalcoholic Fatty Liver Disease". New England Journal of Medicine. 363 (2): 193–195. doi:10.1056/NEJMc1005265. PMID 20647217.
- ^ Wang J, Ye C, Fei S (2020). "Association between APOC3 polymorphisms and non-alcoholic fatty liver disease risk: a Meta-Analysis". African Health Sciences. 20 (4). Makerere Medical School: 1800–1804. doi:10.4314/ahs.v20i4.34. PMC 8351815. PMID 34394242.
- ^ Chen BF, Chien Y, Tsai PH, Perng PC, Yang YP, Hsueh KC, Liu CH, Wang YH (2021). "A PRISMA-compliant meta-analysis of apolipoprotein C3 polymorphisms and nonalcoholic fatty liver disease". Journal of the Chinese Medical Association. 84 (10): 923–929. doi:10.1097/JCMA.0000000000000564. PMID 34108427.
- ^ Peter A, Kantartzis K, Machicao F, Machann J, Wagner S, Templin S, Königsrainer I, Königsrainer A, Schick F, Fritsche A, Häring HU, Stefan N (2012). "Visceral obesity modulates the impact of apolipoprotein C3 Gene variants on liver fat content". International Journal of Obesity. 36 (6): 774–782. doi:10.1038/ijo.2011.154. PMID 21829161. S2CID 205154071.
- ^ Stroes ES, Alexander VJ, Karwatowska-Prokopczuk E, Hegele RA, Arca M, Ballantyne CM, Soran H, Prohaska TA, Xia S, Ginsberg HN, Witztum JL, Tsimikas S, et al. (Balance Investigators) (2024). "Olezarsen, Acute Pancreatitis, and Familial Chylomicronemia Syndrome". New England Journal of Medicine. 390 (19): 1781–1792. doi:10.1056/NEJMoa2400201. PMID 38587247.
- ^ Kypreos KE (2008). "ABCA1 Promotes the de Novo Biogenesis of Apolipoprotein CIII-Containing HDL Particles in Vivo and Modulates the Severity of Apolipoprotein CIII-Induced Hypertriglyceridemia". Biochemistry. 47 (39): 10491–10502. doi:10.1021/bi801249c. PMID 18767813.
External links
[edit]- Apolipoprotein+C-III at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Human APOC3 genome location and APOC3 gene details page in the UCSC Genome Browser.
Further reading
[edit]- von Eckardstein A, Holz H, Sandkamp M, Weng W, Funke H, Assmann G (May 1991). "Apolipoprotein C-III(Lys58----Glu). Identification of an apolipoprotein C-III variant in a family with hyperalphalipoproteinemia". The Journal of Clinical Investigation. 87 (5): 1724–31. doi:10.1172/JCI115190. PMC 295277. PMID 2022742.
- Karathanasis SK, Zannis VI, Breslow JL (Apr 1985). "Isolation and characterization of cDNA clones corresponding to two different human apoC-III alleles". Journal of Lipid Research. 26 (4): 451–56. doi:10.1016/S0022-2275(20)34359-5. PMID 2989400.
- Karathanasis SK, Oettgen P, Haddad IA, Antonarakis SE (Nov 1986). "Structure, evolution, and polymorphisms of the human apolipoprotein A4 gene (APOA4)". Proceedings of the National Academy of Sciences of the United States of America. 83 (22): 8457–61. Bibcode:1986PNAS...83.8457K. doi:10.1073/pnas.83.22.8457. PMC 386949. PMID 3095836.
- Maeda H, Hashimoto RK, Ogura T, Hiraga S, Uzawa H (Dec 1987). "Molecular cloning of a human apoC-III variant: Thr 74----Ala 74 mutation prevents O-glycosylation". Journal of Lipid Research. 28 (12): 1405–09. doi:10.1016/S0022-2275(20)38574-6. PMID 3123586.
- Karathanasis SK (Oct 1985). "Apolipoprotein multigene family: tandem organization of human apolipoprotein AI, CIII, and AIV genes". Proceedings of the National Academy of Sciences of the United States of America. 82 (19): 6374–78. Bibcode:1985PNAS...82.6374K. doi:10.1073/pnas.82.19.6374. PMC 390718. PMID 3931073.
- Zannis VI, Cole FS, Jackson CL, Kurnit DM, Karathanasis SK (Jul 1985). "Distribution of apolipoprotein A-I, C-II, C-III, and E mRNA in fetal human tissues. Time-dependent induction of apolipoprotein E mRNA by cultures of human monocyte-macrophages". Biochemistry. 24 (16): 4450–55. doi:10.1021/bi00337a028. PMID 3931677.
- Shelley CS, Sharpe CR, Baralle FE, Shoulders CC (Nov 1985). "Comparison of the human apolipoprotein genes. Apo AII presents a unique functional intron-exon junction". Journal of Molecular Biology. 186 (1): 43–51. doi:10.1016/0022-2836(85)90255-4. PMID 3935800.
- Hospattankar AV, Brewer HB, Ronan R, Fairwell T (Mar 1986). "Amino acid sequence of human plasma apolipoprotein C-III from normolipidemic subjects". FEBS Letters. 197 (1–2): 67–73. Bibcode:1986FEBSL.197...67H. doi:10.1016/0014-5793(86)80300-3. PMID 3949020. S2CID 83923614.
- Brewer HB, Shulman R, Herbert P, Ronan R, Wehrly K (Aug 1974). "The complete amino acid sequence of alanine apolipoprotein (apoC-3), and apolipoprotein from human plasma very low density lipoproteins". The Journal of Biological Chemistry. 249 (15): 4975–84. doi:10.1016/S0021-9258(19)42416-2. PMID 4846755.
- Karathanasis SK, McPherson J, Zannis VI, Breslow JL (1983). "Linkage of human apolipoproteins A-I and C-III genes". Nature. 304 (5924): 371–73. Bibcode:1983Natur.304..371K. doi:10.1038/304371a0. PMID 6308458. S2CID 4336695.
- Sharpe CR, Sidoli A, Shelley CS, Lucero MA, Shoulders CC, Baralle FE (May 1984). "Human apolipoproteins AI, AII, CII and CIII. cDNA sequences and mRNA abundance". Nucleic Acids Research. 12 (9): 3917–32. doi:10.1093/nar/12.9.3917. PMC 318799. PMID 6328445.
- Law SW, Gray G, Brewer HB (Apr 1983). "cDNA cloning of human apoA-I: amino acid sequence of preproapoA-I". Biochemical and Biophysical Research Communications. 112 (1): 257–64. doi:10.1016/0006-291X(83)91824-7. PMID 6404278.
- Protter AA, Levy-Wilson B, Miller J, Bencen G, White T, Seilhamer JJ (Dec 1984). "Isolation and sequence analysis of the human apolipoprotein CIII gene and the intergenic region between the apo AI and apo CIII genes". DNA. 3 (6): 449–56. doi:10.1089/dna.1.1984.3.449. PMID 6439535.
- Levy-Wilson B, Appleby V, Protter A, Auperin D, Seilhamer JJ (Oct 1984). "Isolation and DNA sequence of full-length cDNA for human preapolipoprotein CIII". DNA. 3 (5): 359–64. doi:10.1089/dna.1984.3.359. PMID 6548954.
- Dammerman M, Sandkuijl LA, Halaas JL, Chung W, Breslow JL (May 1993). "An apolipoprotein CIII haplotype protective against hypertriglyceridemia is specified by promoter and 3' untranslated region polymorphisms". Proceedings of the National Academy of Sciences of the United States of America. 90 (10): 4562–66. Bibcode:1993PNAS...90.4562D. doi:10.1073/pnas.90.10.4562. PMC 46552. PMID 8099442.
- Wu JH, Kao JT, Wen MS, Lo SK (May 2000). "DNA polymorphisms at the apolipoprotein A1-CIII loci in Taiwanese: correlation of plasma APOCIII with triglyceride level and body mass index". Journal of the Formosan Medical Association = Taiwan Yi Zhi. 99 (5): 367–74. PMID 10870325.
- Geraci MW, Moore M, Gesell T, Yeager ME, Alger L, Golpon H, Gao B, Loyd JE, Tuder RM, Voelkel NF (Mar 2001). "Gene expression patterns in the lungs of patients with primary pulmonary hypertension: a gene microarray analysis". Circulation Research. 88 (6): 555–62. doi:10.1161/01.RES.88.6.555. PMID 11282888.
- Inoue Y, Miyazaki M, Tsuji T, Sakaguchi M, Fukaya K, Huh NH, Namba M (Nov 2001). "Reactivation of liver-specific gene expression in an immortalized human hepatocyte cell line by introduction of the human HNF4alpha2 gene". International Journal of Molecular Medicine. 8 (5): 481–87. doi:10.3892/ijmm.8.5.481. PMID 11605014.
- Pastier D, Lacorte JM, Chambaz J, Cardot P, Ribeiro A (Apr 2002). "Two initiator-like elements are required for the combined activation of the human apolipoprotein C-III promoter by upstream stimulatory factor and hepatic nuclear factor-4". The Journal of Biological Chemistry. 277 (17): 15199–206. doi:10.1074/jbc.M200227200. PMID 11839757.
- Chhabra S, Narang R, Krishnan LR, Vasisht S, Agarwal DP, Srivastava LM, Manchanda SC, Das N (Jun 2002). "Apolipoprotein C3 SstI polymorphism and triglyceride levels in Asian Indians". BMC Genetics. 3: 9. doi:10.1186/1471-2156-3-9. PMC 116591. PMID 12052247.