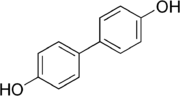
4,4'-Biphenol
| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
[1,1′-Biphenyl]-4,4′-diol | |
| Other names
4,4′-Dihydroxybiphenyl
4,4′-Diphenol 4,4′-Biphenyldiol | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.002.001 |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C12H10O2 | |
| Molar mass | 186.210 g·mol−1 |
| Appearance | colorless or white solid |
| Melting point | 283 °C (541 °F; 556 K)[1] |
| Boiling point | Sublimes |
| Insoluble in water Soluble in ethanol and ether | |
| Hazards | |
| Flash point | > 93.3 °C (199.9 °F; 366.4 K) |
| Safety data sheet (SDS) | MSDS |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
4,4′-Biphenol is an aromatic organic compound, and one of three symmetrical isomers of biphenol. It is a colourless crystalline solid with a high melting point. It is primarily used in the production of polymers, particularly liquid crystals where it imparts high thermal stability.
Synthesis
[edit]The industrial synthesis of 4,4′-biphenol was developed by Allan Hay in the 1960s.[2][3] As the direct oxidative coupling of phenol gives a mixture of isomers,[4][5] 4,4′-biphenol is instead prepared from 2,6-di-tert-butylphenol, where para-coupling is the only possibility.[3] The reaction with oxygen produces phenol-radicals which undergo rapid dimerisation, forming a diphenoquinone. This is reduced to the tetra-butyl-biphenyl derivative by a reaction with additional 2,6-di-tert-butylphenol, in an oxygen-free environment.[2] In the final step, high temperature dealkylation is performed to remove the butyl groups, producing the desired 4,4′-biphenol product.[3] If groups less bulky that t-butyl are used then polyphenylene ethers such as poly(p-phenylene oxide) can be produced.[6]
Safety
[edit]4,4'-Biphenol had actually been elucidated to have an estrogenic SAR.[7]
See also
[edit]References
[edit]- ^ Chen, Guoliang; Du, Fangyu; Zhou, Qifan; Liu, Dongdong; Fang, Ting; Shi, Yajie; Du, Yang (2018-01-31). "Dimerization of Aromatic Compounds Using Palladium-Carbon-Catalyzed Suzuki–Miyaura Cross-Coupling by One-Pot Synthesis (Supporting Information)". Synlett. 29 (6): 779–784. doi:10.1055/s-0036-1591892. ISSN 0936-5214. S2CID 104289422.
- ^ a b Hay, A.S. (January 1965). "Dehydrogenation reactions with diphenoquinones". Tetrahedron Letters. 6 (47): 4241–4243. doi:10.1016/S0040-4039(01)89114-0.
- ^ a b c Hay, Allan S. (April 1969). "p,p'-Biphenols". The Journal of Organic Chemistry. 34 (4): 1160–1161. doi:10.1021/jo01256a098.
- ^ Helmut Fiege; Heinz-Werner Voges; Toshikazu Hamamoto; et al. (2002). "Phenol Derivatives". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a19_313. ISBN 3527306730.
- ^ O’Brien, M. K.; Vanasse, B. (2004). "Vanadium(IV) Chloride". In Paquette, L. (ed.). Encyclopedia of Reagents for Organic Synthesis. New York, NY: J. Wiley & Sons. doi:10.1002/047084289X.rv001. ISBN 0471936235.
- ^ Hay, Allan S. (April 1962). "Polymerization by oxidative coupling. II. Oxidation of 2,6‐disubstituted phenols". Journal of Polymer Science. 58 (166): 581–591. doi:10.1002/pol.1962.1205816634.
- ^ Dodds, E. C.; Lawson, W. (1937). "A Simple Aromatic oestrogenic Agent with an Activity of the Same Order as that of estrone". Nature. 139 (3519): 627–628. doi:10.1038/139627b0. ISSN 0028-0836. S2CID 4119670.
