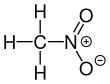
Nitromethane
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Nitromethane
| |||
| Preferred IUPAC name
Nitromethane[1] | |||
| Other names
Nitrocarbol
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.000.797 | ||
| KEGG | |||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| CH3NO2 | |||
| Molar mass | 61.04 g/mol | ||
| Appearance | colorless, oily liquid[2] | ||
| Odor | Light, fruity[2] | ||
| Density | 1.1371 g/cm3 (20 °C)[3] | ||
| Melting point | −28.7 °C (−19.7 °F; 244.5 K)[3] | ||
| Boiling point | 101.2 °C (214.2 °F; 374.3 K)[3] | ||
| Critical point (T, P) | 588 K, 6.0 MPa[4] | ||
| ca. 10 g/100 mL | |||
| Solubility | miscible in diethyl ether, acetone, ethanol, methanol[3] | ||
| Vapor pressure | 28 mmHg (20 °C)[2] | ||
| Acidity (pKa) | |||
| -21.0·10−6 cm3/mol[7] | |||
| Thermal conductivity | 0.204 W/(m·K) at 25 °C[8] | ||
Refractive index (nD)
|
1.3817 (20 °C)[3] | ||
| Viscosity | 0.63 cP at 25 °C[8] | ||
| 3.46[9] | |||
| Explosive data | |||
| Shock sensitivity | Low | ||
| Friction sensitivity | Low | ||
| Detonation velocity | 6400 m/s | ||
| Thermochemistry[10] | |||
Heat capacity (C)
|
106.6 J/(mol·K) | ||
Std molar
entropy (S⦵298) |
171.8 J/(mol·K) | ||
Std enthalpy of
formation (ΔfH⦵298) |
-112.6 kJ/mol | ||
Gibbs free energy (ΔfG⦵)
|
-14.4 kJ/mol | ||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
Flammable, health hazard | ||
| GHS labelling: | |||
   
| |||
| Danger | |||
| H203, H226, H301, H331, H351 | |||
| P210, P261, P280, P304+P340, P312, P370+P378, P403+P233 | |||
| NFPA 704 (fire diamond) | |||
| Flash point | 35[9] °C (95 °F; 308 K) | ||
| 418[9] °C (784 °F; 691 K) | |||
| Explosive limits | 7–22%[9] | ||
Threshold limit value (TLV)
|
20 ppm[9] | ||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose)
|
940 mg/kg (oral, rat) 950 mg/kg (oral, mouse)[11] | ||
LDLo (lowest published)
|
750 mg/kg (rabbit, oral) 125 mg/kg (dog, oral)[11] | ||
LCLo (lowest published)
|
7087 ppm (mouse, 2 h) 1000 ppm (monkey) 2500 ppm (rabbit, 12 h) 5000 ppm (rabbit, 6 h)[11] | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
TWA 100 ppm (250 mg/m3)[2] | ||
REL (Recommended)
|
none[2] | ||
IDLH (Immediate danger)
|
750 ppm[2] | ||
| Related compounds | |||
Related nitro compounds
|
nitroethane | ||
Related compounds
|
methyl nitrite methyl nitrate | ||
| Supplementary data page | |||
| Nitromethane (data page) | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Nitromethane, sometimes shortened to simply "nitro", is an organic compound with the chemical formula CH
3NO
2. It is the simplest organic nitro compound. It is a polar liquid commonly used as a solvent in a variety of industrial applications such as in extractions, as a reaction medium, and as a cleaning solvent. As an intermediate in organic synthesis, it is used widely in the manufacture of pesticides, explosives, fibers, and coatings.[12] Nitromethane is used as a fuel additive in various motorsports and hobbies, e.g. Top Fuel drag racing and miniature internal combustion engines in radio control, control line and free flight model aircraft.
Preparation
[edit]Nitromethane is produced industrially by combining propane and nitric acid in the gas phase at 350–450 °C (662–842 °F). This exothermic reaction produces the four industrially significant nitroalkanes: nitromethane, nitroethane, 1-nitropropane, and 2-nitropropane. The reaction involves free radicals, including the alkoxyl radicals of the type CH3CH2CH2O, which arise via homolysis of the corresponding nitrite ester. These alkoxy radicals are susceptible to C—C fragmentation reactions, which explains the formation of a mixture of products.[12]
Laboratory methods
[edit]It can be prepared in other methods that are of instructional value. The reaction of sodium chloroacetate with sodium nitrite in aqueous solution produces this compound:[13]
Uses
[edit]The dominant use of the nitromethane is as a precursor reagent. A major derivative is chloropicrin (CCl3NO2), a widely used pesticide. It condenses with formaldehyde (Henry reaction) to eventually give tris(hydroxymethyl)aminomethane ("tris"), a widely used buffer and ingredient in alkyd resins.[12]
Solvent and stabilizer
[edit]The major application is as a stabilizer in chlorinated solvents. As an organic solvent, nitromethane has an unusual combination of properties: highly polar (εr = 36 at 20 °C and μ = 3.5 Debye) but aprotic and weakly basic. This combination makes it useful for dissolving positively charged, strongly electrophilic species. It is a solvent for acrylate monomers, such as cyanoacrylates (more commonly known as "super-glues").[12]
Fuel
[edit]Although a minor application in terms of volume,[12] nitromethane also is used as a fuel or fuel additive for sports and hobby. For some application, it is mixed with methanol in racing cars, boats, and model engines.
Nitromethane is used as a fuel in motor racing, particularly drag racing, as well as for radio-controlled model power boats, cars, planes and helicopters. In this context, nitromethane is commonly referred to as "nitro fuel" or simply "nitro", and is the principal ingredient for fuel used in the "Top Fuel" category of drag racing.[14]
The oxygen content of nitromethane enables it to burn with much less atmospheric oxygen than conventional fuels.[15] During nitromethane combustion, nitric oxide (NO) is one of the major emission products along with CO2 and H2O.[16] Nitric oxide contributes to air pollution, acid rain, and ozone layer depletion. Recent (2020) studies[17] suggest the correct stoichiometric equation for the burning of nitromethane is:
- 4 CH3NO2 + 5 O2 → 4 CO2 + 6 H2O + 4 NO
The amount of air required to burn 1 kg (2.2 lb) of gasoline is 14.7 kg (32 lb), but only 1.7 kg (3.7 lb) of air is required for 1 kg of nitromethane. Since an engine's cylinder can only contain a limited amount of air on each stroke, 8.6 times as much nitromethane as gasoline can be burned in one stroke. Nitromethane, however, has a lower specific energy: gasoline provides about 42–44 MJ/kg, whereas nitromethane provides only 11.3 MJ/kg.[citation needed] This analysis indicates that nitromethane generates about 2.3 times the power of gasoline when combined with a given amount of oxygen.[citation needed]
Nitromethane can also be used as a monopropellant, i.e., a propellant that decomposes to release energy without added oxygen. It was first tested as rocket monopropellant in 1930s by Luigi Crocco fom Italian Rocket Society.[18][19] There is a renewed interest in nitromethane as safer replacement of hydrazine monopropellant.[20] The following equation describes this process:
- 2 CH3NO2 → 2 CO + 2 H2O + H2 + N2
Nitromethane has a laminar combustion velocity of approximately 0.5 m/s, somewhat higher than gasoline, thus making it suitable for high-speed engines. It also has a somewhat higher flame temperature of about 2,400 °C (4,350 °F). The high heat of vaporization of 0.56 MJ/kg together with the high fuel flow provides significant cooling of the incoming charge (about twice that of methanol), resulting in reasonably low temperatures.[citation needed]
Nitromethane is usually used with rich air–fuel mixtures because it provides power even in the absence of atmospheric oxygen. When rich air–fuel mixtures are used, hydrogen and carbon monoxide are two of the combustion products. These gases often ignite, sometimes spectacularly, as the normally very rich mixtures of the still burning fuel exits the exhaust ports. Very rich mixtures are necessary to reduce the temperature of combustion chamber hot parts in order to control pre-ignition and subsequent detonation. Operational details depend on the particular mixture and engine characteristics.[citation needed]
A small amount of hydrazine blended in nitromethane can increase the power output even further. With nitromethane, hydrazine forms an explosive salt that is again a monopropellant. This unstable mixture poses a severe safety hazard. The National Hot Rod Association and Academy of Model Aeronautics do not permit its use in competitions.[21]
In model aircraft and car glow fuel, the primary ingredient is generally methanol with some nitromethane (0% to 65%, but rarely over 30%, and 10–20% lubricants (usually castor oil and/or synthetic oil)). Even moderate amounts of nitromethane tend to increase the power created by the engine (as the limiting factor is often the air intake), making the engine easier to tune (adjust for the proper air/fuel ratio).
Former uses
[edit]It formerly was used in the explosives industry as a component in a binary explosive formulation with ammonium nitrate and in shaped charges, and it was used as a chemical stabilizer to prevent decomposition of various halogenated hydrocarbons.[22]
Other
[edit]It can be used as an explosive, when gelled with several percent of gelling agent. This type of mixture is called PLX. Other mixtures include ANNM and ANNMAl – explosive mixtures of ammonium nitrate, nitromethane and aluminium powder.
Reactions
[edit]Acid-base properties
[edit]Nitromethane is a relatively acidic carbon acid. It has a pKa of 17.2 in DMSO solution. This value indicates an aqueous pKa of about 11.[23] It is so acidic because the anion admits an alternate, stabilizing resonance structure:

The acid deprotonates only slowly. Protonation of the conjugate base O2NCH2−, which is nearly isosteric with nitrate, occurs initially at oxygen.[24]
Organic reactions
[edit]In organic synthesis nitromethane is employed as a one carbon building block.[25][26] Its acidity allows it to undergo deprotonation, enabling condensation reactions analogous to those of carbonyl compounds. Thus, under base catalysis, nitromethane adds to aldehydes in 1,2-addition in the nitroaldol reaction. Some important derivatives include the pesticides chloropicrin (Cl3CNO2), beta-nitrostyrene, and tris(hydroxymethyl)nitromethane, ((HOCH2)3CNO2). Reduction of the latter gives tris(hydroxymethyl)aminomethane, (HOCH2)3CNH2, better known as tris, a widely used buffer. In more specialized organic synthesis, nitromethane serves as a Michael donor, adding to α,β-unsaturated carbonyl compounds via 1,4-addition in the Michael reaction.
Purification
[edit]Nitromethane is a popular solvent in organic and electroanalytical chemistry. It can be purified by cooling below its freezing point, washing the solid with cold diethyl ether, followed by distillation.[27]
Safety
[edit]Nitromethane has a modest acute toxicity. LD50 (oral, rats) is 1210±322 mg/kg.[12]
Nitromethane is "reasonably anticipated to be a human carcinogen" according to a U.S. government report.[28]
Explosive properties
[edit]Nitromethane was not known to be a high explosive until a railroad tank car loaded with it exploded on June 1, 1958.[29] After much testing[citation needed], it was realized that nitromethane was a more energetic high explosive than TNT[citation needed], although TNT has a higher velocity of detonation (VoD) and brisance[citation needed]. Both of these explosives are oxygen-poor, and some benefits are gained from mixing with an oxidizer, such as ammonium nitrate. Pure nitromethane is an insensitive explosive with a VoD of approximately 6,400 m/s (21,000 ft/s), but even so inhibitors may be used to reduce the hazards. The tank car explosion was speculated[citation needed] to be due to adiabatic compression, a hazard common to all liquid explosives. This is when small entrained air bubbles compress and superheat with rapid rises in pressure. It was thought that an operator rapidly snapped shut a valve creating a "hammer-lock" pressure surge.[citation needed]
If mixed with ammonium nitrate, which is used as an oxidizer, it forms an explosive mixture known as ANNM.
Nitromethane is used as a model explosive, along with TNT. It has several advantages as a model explosive over TNT, namely its uniform density and lack of solid post-detonation species that complicate the determination of equation of state and further calculations.
Nitromethane reacts with solutions of sodium hydroxide or methoxide in alcohol to produce an insoluble salt of nitromethane. This substance is a sensitive explosive which reverts to nitromethane under acidic conditions and decomposes in water to form another explosive compound, sodium methazonate, which has a reddish-brown color:
- 2 CH3NO2 + NaOH → HON=CHCH=NO2Na + 2 H2O
Nitromethane's reaction with solid sodium hydroxide is hypergolic.
See also
[edit]- Top Fuel
- Adiabatic flame temperature, a thermodynamic calculation of the flame temperature of nitromethane
- Dinitromethane
- Model engine
- Trinitromethane
- Tetranitromethane
- RE factor
References
[edit]- ^ "Front Matter". Nomenclature of Organic Chemistry : IUPAC Recommendations and Preferred Names 2013 (Blue Book). Cambridge: The Royal Society of Chemistry. 2014. p. 662. doi:10.1039/9781849733069-FP001. ISBN 978-0-85404-182-4.
- ^ a b c d e f NIOSH Pocket Guide to Chemical Hazards. "#0457". National Institute for Occupational Safety and Health (NIOSH).
- ^ a b c d e Haynes, p. 3.414
- ^ Haynes, p. 6.69
- ^ Haynes, p. 5.94
- ^ Reich, Hans. "Bordwell pKa table: "Nitroalkanes"". University of Wisconsin Chemistry Department. Retrieved 27 January 2022.
- ^ Haynes, p. 3.576
- ^ a b Haynes, p. 6.231
- ^ a b c d e Haynes, p. 15.19
- ^ Haynes, p. 5.20
- ^ a b c "Nitromethane". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- ^ a b c d e f Markofsky, S. B. (2000). "Nitro Compounds, Aliphatic". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a17_401.pub2. ISBN 978-3527306732.
- ^ Whitmore, F. C.; Whitmore, M. G. (1941). "Nitromethane". Organic Syntheses; Collected Volumes, vol. 1, p. 401.
- ^ Carley, Larry (2013-01-06). "HPBG: The Power of Racing Fuels". Engine Builder Magazine. Retrieved 2024-05-31.
- ^ "What is Nitro Methane Fuel: Understanding High-Performance Racing's Power Source - Ran When Parked - Car, Vehicle & Truck Guides and Repair Journals". ranwhenparked.net. 2024-03-05. Retrieved 2024-05-31.
- ^ Shrestha, Krishna Prasad; Vin, Nicolas; Herbinet, Olivier; Seidel, Lars; Battin-Leclerc, Frédérique; Zeuch, Thomas; Mauss, Fabian (2020-02-01). "Insights into nitromethane combustion from detailed kinetic modeling – Pyrolysis experiments in jet-stirred and flow reactors" (PDF). Fuel. 261: 116349. Bibcode:2020Fuel..26116349S. doi:10.1016/j.fuel.2019.116349. ISSN 0016-2361. S2CID 208755285.
- ^ Shrestha, Krishna Prasad; Vin, Nicolas; Herbinet, Olivier; Seidel, Lars; Battin-Leclerc, Frédérique; Zeuch, Thomas; Mauss, Fabian (2020-02-01). "Insights into nitromethane combustion from detailed kinetic modeling – Pyrolysis experiments in jet-stirred and flow reactors" (PDF). Fuel. 261: 116349. Bibcode:2020Fuel..26116349S. doi:10.1016/j.fuel.2019.116349. ISSN 0016-2361. S2CID 208755285.
- ^ Boyer, E.; Kuo, K. (January 2006). Characteristics of Nitromethane for Propulsion Applications. 44th AIAA Aerospace Sciences Meeting and Exhibit. Reno, NV. doi:10.2514/6.2006-361. ISBN 978-1-62410-039-0. AIAA 2006-361.
- ^ Clark, J. D.; Asimov, Isaac (1972). Ignition! an informal history of liquid rocket propellants. Rutgers University Press. pp. 9-10. ISBN 978-0-8135-0725-5.
- ^ Kurilov, Maxim; Werling, Lukas; Kirchberger, Christoph (2023). Nitromethane as a Green Propellant: First Results of a Combustion Test Campaign (PDF). Aerospace Europe Conference 2023. doi:10.13009/EUCASS2023-372.
- ^ "AMA Competition Regulations 2015–2016 Part 7. Fuels" (PDF). www.modelaircraft.org. Academy of Model Aeronautics. February 15, 2016. p. 24. Retrieved April 18, 2014.
- ^ SABIC, Cas AardenGraduate University of Groningen Worked as a chemist in companies such as Wilmar Oleochemicals B. Vand. "Nitromethane: An Ultimate Guide to Properties, Uses and Synthesis". Safrole. Retrieved 2024-05-31.
- ^ Bordwell, F. G.; Satish, A. V. (1994). "Is Resonance Important in Determining the Acidities of Weak Acids or the Homolytic Bond Dissociation Enthalpies (BDEs) of Their Acidic H-A Bonds?". Journal of the American Chemical Society. 116 (20): 8885–8889. doi:10.1021/ja00099a004.
- ^ Kramarz, K. W.; Norton, J. R. (2007). "Slow Proton-Transfer Reactions in Organometallic and Bioinorganic Chemistry". Progress in Inorganic Chemistry. pp. 1–65. doi:10.1002/9780470166437.ch1. ISBN 9780470166437.
- ^ Dauben, H. J. Jr.; Ringold, H. J.; Wade, R. H.; Pearson, D. L.; Anderson, A. G. Jr.; de Boer, T. J.; Backer, H. J. (1963). "Cycloheptanone". Organic Syntheses; Collected Volumes, vol. 4, p. 221.
- ^ Noland, W. E. (1963). "2-Nitroethanol". Organic Syntheses; Collected Volumes, vol. 4, p. 833.
- ^ Coetzee, J. F.; Chang, T.-H. (1986). "Recommended Methods for the Purification of Solvents and Tests for Impurities: Nitromethane" (PDF). Pure and Applied Chemistry. 58 (11): 1541–1545. doi:10.1351/pac198658111541. S2CID 95631774.
- ^ "National Toxicology Program 15th Report on Carcinogens" (PDF). National Toxicology Program U.S. Department of Health and Human Services. December 21, 2021. Archived (PDF) from the original on October 2, 2023. Retrieved May 30, 2024.
- ^ Interstate Commerce Commission. "Accident Near Mt. Pulaski, ILL" (PDF). Ex Parte No 213. Archived from the original (PDF) on 1 November 2020.
Cited sources
[edit]- Haynes, William M., ed. (2011). CRC Handbook of Chemistry and Physics (92nd ed.). CRC Press. ISBN 978-1439855119.