Icatibant
 | |
| Clinical data | |
|---|---|
| Trade names | Firazyr |
| Other names | Hoe 140, JE 049[1] |
| AHFS/Drugs.com | Monograph |
| License data | |
| Pregnancy category |
|
| Routes of administration | Subcutaneous |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG |
|
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
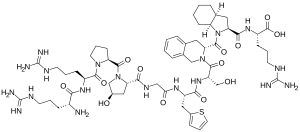
| Formula | C59H89N19O13S |
| Molar mass | 1304.54 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Icatibant, sold under the brand name Firazyr, is a medication for the symptomatic treatment of acute attacks of hereditary angioedema (HAE) in adults with C1-esterase-inhibitor deficiency.[5][3][4] It is not effective in angioedema caused by medication from the ACE inhibitor class.[6]
It is a peptidomimetic consisting of ten amino acids, which is a selective and specific antagonist of bradykinin B2 receptors.
Mechanism of action
[edit]Bradykinin is a peptide-based hormone that is formed locally in tissues, very often in response to a trauma. It increases vessel permeability, dilates blood vessels and causes smooth muscle cells to contract. Bradykinin plays an important role as the mediator of pain. Surplus bradykinin is responsible for the typical symptoms of inflammation, such as swelling, redness, overheating and pain. These symptoms are mediated by activation of bradykinin B2 receptors. Icatibant acts as a bradykinin inhibitor by blocking the binding of native bradykinin to the bradykinin B2 receptor. Little is known about the effects of icatibant on the bradykinin B1 receptor.
Society and culture
[edit]Legal status
[edit]Icatibant received orphan drug status in Australia, the EU, Switzerland, and the US for the treatment of hereditary angioedema (HAE).[7]
In the EU, the approval by the European Commission (July 2008) allows Jerini to market Firazyr in the European Union's 27 member states, as well as Switzerland, Liechtenstein and Iceland, making it the first product to be approved in all EU countries for the treatment of hereditary angioedema.[5] In the US, the drug was granted FDA approval in August 2011.[8]
References
[edit]- ^ "Icatibant: HOE 140, JE 049, JE049". Drugs in R&D. 5 (6): 343–8. 2004. doi:10.2165/00126839-200405060-00006. PMID 15563238. S2CID 25491021.
- ^ https://www.tga.gov.au/resources/prescription-medicines-registrations/icatibant-wkt-wockhardt-bio-pty-ltd
- ^ a b "Firazyr- icatibant acetate injection, solution". DailyMed. 16 December 2019. Retrieved 17 April 2020.
- ^ a b "Firazyr EPAR". European Medicines Agency (EMA). 17 September 2018. Retrieved 17 April 2020.
- ^ a b "Jerini Receives European Commission Approval for Firazyr (Icatibant) in the Treatment of HAE" (Press release). Jerini AG. 15 July 2008. Retrieved 22 July 2008.[permanent dead link]
- ^ Sinert R, Levy P, Bernstein JA, Body R, Sivilotti ML, Moellman J, et al. (September–October 2017). "Randomized Trial of Icatibant for Angiotensin-Converting Enzyme Inhibitor-Induced Upper Airway Angioedema". The Journal of Allergy and Clinical Immunology. In Practice. 5 (5): 1402–1409.e3. doi:10.1016/j.jaip.2017.03.003. PMID 28552382.
{{cite journal}}: CS1 maint: overridden setting (link) - ^ Longhurst HJ (September 2010). "Management of acute attacks of hereditary angioedema: potential role of icatibant". Vascular Health and Risk Management. 6: 795–802. doi:10.2147/vhrm.s4332. PMC 2941790. PMID 20859548.
- ^ "FDA Approves Shire's Firazyr (icatibant injection) for Acute Attacks of Hereditary Angioedema (HAE)" (Press release). Shire. Retrieved 28 August 2011.
