Digital pathology
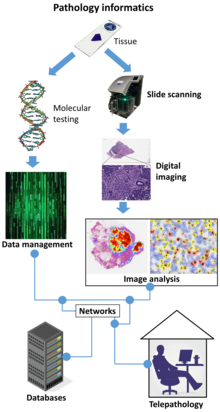
Digital pathology is a sub-field of pathology that focuses on managing and analyzing information generated from digitized specimen slides. It utilizes computer-based technology and virtual microscopy to view, manage, share, and analyze digital slides on computer monitors.[1] This field has applications in diagnostic medicine and aims to achieve more efficient and cost-effective diagnoses, prognoses, and disease predictions through advancements in machine learning and artificial intelligence in healthcare.[2][3]
History
[edit]The roots of digital pathology trace back to the 1960s with early telepathology experiments. The concept of virtual microscopy emerged in the 1990s across various areas of life science research.[4] At the turn of the century the scientific community more and more agreed on the term "digital pathology" to denote digitization efforts in pathology. However, in 2000, the technical requirements (scanner, storage, network) were still a limiting factor for a broad dissemination of digital pathology concepts. This changed as new powerful and affordable scanner technology as well as mass / cloud storage technologies appeared on the market. The field of radiology has undergone the digital transformation almost 15 years ago, not because radiology is more advanced, but there are fundamental differences between digital images in radiology and digital pathology: The image source in radiology is the (alive) patient, and today in most cases, the image is even primarily captured in digital format. In pathology the scanning is done from preserved and processed specimens, for retrospective studies even from slides stored in a biobank. Besides this difference in pre-analytics and metadata content, the required storage in digital pathology is two to three orders of magnitude higher than in radiology. However, the advantages anticipated through digital pathology are similar to those in radiology:
- Capability to transmit digital slides over distances quickly, which enables telepathology scenarios.
- Capability to access past specimen from the same patients and similar cases for comparison and review, with much less effort than retrieving slides from the archive shelves.
- Capability to compare different areas of multiple slides simultaneously (slide by slide mode) with the help of a virtual microscope.
- Capability to annotate areas directly in the slide and share this for teaching and research.
Digital pathology is today widely used for educational purposes[5] in telepathology and teleconsultation as well as in research projects. Digital pathology allows to share and annotate slides in a much easier way and to download annotated lecture sets generates new opportunities for e-learning and knowledge sharing in pathology. Digital pathology in diagnostics is an emerging and upcoming field.
Environment
[edit]Scan
[edit]

Digital slides are created from glass slides using specialized scanning machines. All high quality scans must be free of dust, scratches, and other obstructions. There are two common methods for digital slide scanning, tile-based scanning and line-based scanning.[6] Both technologies use an integrated camera and a motorized stage to move the slide around while parts of the tissue are imaged. Tile scanners capture square field-of-view images covering the entire tissue area on the slide, while line-scanners capture images of the tissue in long, uninterrupted stripes rather than tiles. In both cases, software associated with the scanner stitch the tiles or lines together into a single, seamless image.
Z-stacking is the scanning of a slide at multiple focal planes along the vertical z-axis.[7]
View
[edit]Digital slides are accessible for viewing via a computer monitor and viewing software either locally or remotely via the Internet. An example of an open-source, web-based viewer for this purpose implemented in pure JavaScript, for desktop and mobile, is the OpenSeadragon[8] viewer. QuPath[9] is another such open source software, which is often used for digital pathology applications because it offers a powerful set of tools for working with whole slide images. OpenSlide,[10] on the other hand is a C library (Python and Java bindings are also available) that provides a simple interface to read and view whole-slide images.
Manage
[edit]Digital slides are maintained in an information management system that allows for archival and intelligent retrieval.
Network
[edit]Digital slides are often stored and delivered over the Internet or private networks, for viewing and consultation.
Analyze
[edit]Image analysis tools are used to derive objective quantification measures from digital slides. Image segmentation and classification algorithms, often implemented using deep neural networks, are used to identify medically significant regions and objects on digital slides. A GPU acceleration software for pathology imaging analysis, cross-comparing spatial boundaries of a huge amount of segmented micro-anatomic objects has been developed.[11] The core algorithm of PixelBox in this software has been adopted in Fixstars' Geometric Performance Primitives (GPP) library[12] as a part of NVIDIA Developer, which is a production geometry engine for advanced graphical information systems, electronic design automation, computer vision and motion planning solutions.[13]
-
Tissue segmentation for digital calculation of bone marrow cellularity in QuPath: The system is trained on the appearance of immune cells versus other tissue, and uses this to give an overall percentage of each type.
-
Breast cancer prediction by AI.[15]
 |
 |
Integrate
[edit]Digital pathology workflow is integrated into the institution's overall operational environment. Slide digitization is expected to reduce the number of routine, manually reviewed slides, maximizing workload efficiency.
Sharing
[edit]Digital pathology also allows internet information sharing for education, diagnostics, publication and research. This may take the form of publicly available datasets or open source access to machine learning algorithms.
Challenges
[edit]

Digital pathology has been approved by the FDA for primary diagnosis.[16] The approval was based on a multi-center study of 1,992 cases in which whole-slide imaging (WSI) was shown to be non-inferior to microscopy across a wide range of surgical pathology specimens, sample types and stains.[17] While there are advantages to WSI when creating digital data from glass slides, when it comes to real-time telepathology applications, WSI is not a strong choice for discussion and collaboration between multiple remote pathologists.[18] Furthermore, unlike digital radiology where the elimination of film made return on investment (ROI) clear, the ROI on digital pathology equipment is less obvious. The strongest ROI justification includes improved quality of healthcare, increased efficiency for pathologists, and reduced costs in handling glass slides.[19]
Validation
[edit]Validation of a digital microscopy workflow in a specific environment (see above) is important to ensure high diagnostic performance of pathologists when evaluating digital whole-slide images. There are different methods that can be used for this validation process.[20] The College of American Pathologists has published a guideline with minimal requirements for validation of whole slide imaging systems for diagnostic purposes in human pathology.[21]
Potential
[edit]Trained pathologists traditionally view tissue slides under a microscope. These tissue slides may be stained to highlight cellular structures. When slides are digitized, they are able to be shared through tele-pathology and are numerically analyzed using computer algorithms. Algorithms can be used to automate the manual counting of structures, or for classifying the condition of tissue such as is used in grading tumors. They can additionally be used for feature detection of mitotic figures, epithelial cells, or tissue specific structures such as lung cancer nodules, glomeruli, or vessels, or estimation of molecular biomarkers such as mutated genes, tumor mutational burden, or transcriptional changes.[22][23][24] This has the potential to reduce human error and improve accuracy of diagnoses. Digital slides can be easily shared, increasing the potential for data usage in education as well as in consultations between expert pathologists. Multiplexed imaging (staining multiple markers on the same slide) allows pathologists to understand finer distribution of cell-types and their relative locations.[25] An understanding of the spatial distribution of cell-types or markers and pathways they express, can allow for prescription of targeted drugs or build combinational therapies in a personalized manner.
See also
[edit]References
[edit]- ^ Pantanowitz L (2018). "Twenty Years of Digital Pathology: An Overview of the Road Travelled, What is on the Horizon, and the Emergence of Vendor-Neutral Archives". Journal of Pathology Informatics. 9: 40. doi:10.4103/jpi.jpi_69_18. PMC 6289005. PMID 30607307.
- ^ "Whole Slide Imaging | MBF Bioscience". www.mbfbioscience.com. Retrieved 2019-12-02.
- ^ Saillard, Charlie; Dubois, Rémy; Tchita, Oussama; Loiseau, Nicolas; Garcia, Thierry; Adriansen, Aurélie; Carpentier, Séverine; Reyre, Joelle; Enea, Diana; von Loga, Katharina; Kamoun, Aurélie; Rossat, Stéphane; Wiscart, Corentin; Sefta, Meriem; Auffret, Michaël (2023-11-06). "Validation of MSIntuit as an AI-based pre-screening tool for MSI detection from colorectal cancer histology slides". Nature Communications. 14 (1): 6695. doi:10.1038/s41467-023-42453-6. ISSN 2041-1723. PMC 10628260. PMID 37932267.
- ^ Ferreira, R; Moon, J; Humphries, J; Sussman, A; Saltz, J; Miller, R; Demarzo, A (1997). "The virtual microscope". Romanian Journal of Morphology and Embryology. 45: 449–453. PMC 2233368. PMID 9357666.
- ^ Hamilton, Peter W.; Wang, Yinhai; McCullough, Stephen J.; Sussman (2012). "Virtual microscopy and digital pathology in training and education". APMIS. 120 (4): 305–315. doi:10.1111/j.1600-0463.2011.02869.x. PMID 22429213. S2CID 20599493.
- ^ "Informatics, digital & computational pathology".
- ^ Evans AJ, Salama ME, Henricks WH, Pantanowitz L (2017). "Implementation of Whole Slide Imaging for Clinical Purposes: Issues to Consider From the Perspective of Early Adopters". Arch Pathol Lab Med. 141 (7): 944–959. doi:10.5858/arpa.2016-0074-OA. PMID 28440660.
- ^ OpenSeadragon on GitHub
- ^ QuPath on GitHub
- ^ OpenSlide website
- ^ Kaibo Wang; Yin Huai; Rubao Lee; Fusheng Wang; Xiaodong Zhang; Joel H. Saltz (2012). "Accelerating Pathology Image Data Cross-Comparison on CPU-GPU Hybrid Systems". Proceedings VLDB Endowment. Proceedings of the VLDB Endowment. International Conference on Very Large Data Bases. Vol. 5, no. 11. pp. 1543–1554. PMC 3553551. PMID 23355955.
- ^ "Geometric Performance Primitives (GPP)". NVIDIA Developer.
- ^ "Letter to Kaibo Wang" (PDF).
- ^ Lourenço BC, Guimarães-Teixeira C, Flores BCT, Miranda-Gonçalves V, Guimarães R, Cantante M, et al. (2022). "Ki67 and LSD1 Expression in Testicular Germ Cell Tumors Is Not Associated with Patient Outcome: Investigation Using a Digital Pathology Algorithm". Life. 12 (2): 264. doi:10.3390/life12020264. PMC 8875543. PMID 35207551.
- Figure 2 - available via license: Creative Commons Attribution 4.0 International - ^ Cruz-Roa A, Gilmore H, Basavanhally A, Feldman M, Ganesan S, Shih NNC, et al. (2017). "Accurate and reproducible invasive breast cancer detection in whole-slide images: A Deep Learning approach for quantifying tumor extent". Sci Rep. 7: 46450. doi:10.1038/srep46450. PMC 5394452. PMID 28418027.
"This work is licensed under a Creative Commons Attribution 4.0 International License." - ^ "FDA allows marketing of first whole slide imaging system for digital pathology" (Press release). FDA. April 12, 2017. Retrieved May 24, 2017.
- ^ Mukhopadhyay, Sanjay; Feldman, Michael; Abels, Esther (2017). "Whole slide imaging versus microscopy for primary diagnosis in surgical pathology: a multicenter randomized blinded noninferiority study of 1992 cases (pivotal study)". American Journal of Surgical Pathology. 42 (1): 39–52. doi:10.1097/PAS.0000000000000948. PMC 5737464. PMID 28961557.
- ^ Siegel, Gabriel; Regelman, Dan; Maronpot, Robert; Rosenstock, Moti; Hayashi, Shim-mo; Nyska, Abraham (Oct 2018). "Utilizing novel telepathology system in preclinical studies and peer review". Journal of Toxicologic Pathology. 31 (4): 315–319. doi:10.1293/tox.2018-0032. PMC 6206289. PMID 30393436.
- ^ "How to Build a Business Case to Justify the Investment in Digital Pathology". Sectra Medical Systems. Retrieved April 26, 2015.
- ^ Bertram, Christof A; Stathonikos, Nikolas; Donovan, Taryn A; Bartel, Alexander; Fuchs-Baumgartinger, Andrea; Lipnik, Karoline; can Diest, Paul J; Bonsembiante, Federico; Klopfleisch, Robert (September 2021). "Validation of digital microscopy: Review of validation methods and sources of bias". Veterinary Pathology. 59 (1): 26–38. doi:10.1177/03009858211040476. PMC 8761960. PMID 34433345.
- ^ Evans, Andrew J; Brown, Richard; Bui, Marilyn (2021). "Validating whole slide imaging systems for diagnostic purposes in pathology: guideline update from the College of American Pathologists in collaboration with the American Society for Clinical Pathology and the Association for Pathology Informatics". Arch Pathol Lab Med. 146 (4): 440–450. doi:10.5858/arpa.2020-0723-CP. PMID 34003251.
- ^ Aeffner, Famke; Zarella, Mark D.; Buchbinder, Nathan; Bui, Marilyn M.; Goodman, Matthew R.; Hartman, Douglas J.; Lujan, Giovanni M.; Molani, Mariam A.; Parwani, Anil V.; Lillard, Kate; Turner, Oliver C. (2019-03-08). "Introduction to Digital Image Analysis in Whole-slide Imaging: A White Paper from the Digital Pathology Association". Journal of Pathology Informatics. 10: 9. doi:10.4103/jpi.jpi_82_18. ISSN 2229-5089. PMC 6437786. PMID 30984469.
- ^ Jain, Mika S.; Massoud, Tarik F. (2020). "Predicting tumour mutational burden from histopathological images using multiscale deep learning". Nature Machine Intelligence. 2 (6): 356–362. doi:10.1038/s42256-020-0190-5. ISSN 2522-5839. S2CID 220510782.
- ^ Serag, Ahmed; Ion-Margineanu, Adrian; Qureshi, Hammad; McMillan, Ryan; Saint Martin, Marie-Judith; Diamond, Jim; O'Reilly, Paul; Hamilton, Peter (2019). "Translational AI and Deep Learning in Diagnostic Pathology". Frontiers in Medicine. 6: 185. doi:10.3389/fmed.2019.00185. ISSN 2296-858X. PMC 6779702. PMID 31632973.
- ^ Nirmal, Ajit J.; Maliga, Zoltan; Vallius, Tuulia; Quattrochi, Brian; Chen, Alyce A.; Jacobson, Connor A.; Pelletier, Roxanne J.; Yapp, Clarence; Arias-Camison, Raquel; Chen, Yu-An; Lian, Christine G. (2022-04-11). "The spatial landscape of progression and immunoediting in primary melanoma at single cell resolution". Cancer Discovery. 12 (6): 1518–1541. doi:10.1158/2159-8290.CD-21-1357. ISSN 2159-8290. PMC 9167783. PMID 35404441.
Further reading
[edit]- Kayser, K; Kayser, G; Radziszowski, D; Oehmann, A (1999). "From telepathology to virtual pathology institution: The new world of digital pathology" (PDF). Romanian Journal of Morphology and Embryology. 45: 3–9. PMID 15847374.
- McCullough, Bruce; Ying, Xiaoyou; Monticello, Thomas; Bonnefoi, Marc (2004). "Digital Microscopy Imaging and New Approaches in Toxicologic Pathology". Toxicologic Pathology. 32 (5): 49–58. doi:10.1080/01926230490451734. PMID 15503664.
- Schlangen, David; Stede, Manfred; Bontas, Elena Paslaru (2004). "Feeding OWL: Extracting and Representing the Content of Pathology Reports". NLPXML '04 Proceedings of the Workshop on NLP and XML. Nlpxml '04: 43–50.
- Cruz-Roa, Angel; Díaz, Gloria; Romero, Eduardo; González, Fabio (2011). "Automatic Annotation of Histopathological Images Using a Latent Topic Model Based On Non-negative Matrix Factorization". Journal of Pathology Informatics. 2 (4): 4. doi:10.4103/2153-3539.92031. PMC 3312710. PMID 22811960.
- "E-Health and Telemedicine". International Journal of Computer Assisted Radiology and Surgery. 1 (Supplement 1): 119–35. 2006. doi:10.1007/s11548-006-0012-1. S2CID 5642084.
- Fine, Jeffrey L.; Grzybicki, Dana M.; Silowash, Russell; Ho, Jonhan; Gilbertson, John R.; Anthony, Leslie; Wilson, Robb; Parwani, Anil V.; et al. (2008). "Evaluation of whole slide image immunohistochemistry interpretation in challenging prostate needle biopsies". Human Pathology. 39 (4): 564–72. doi:10.1016/j.humpath.2007.08.007. PMID 18234276.
- Kayser, Klaus; Kayser, Gian; Radziszowski, Dominik; Oehmann, Alexander (2004). "New Developments in Digital Pathology: from Telepathology to Virtual Pathology Laboratory". In Duplaga, Mariusz; Zieliński, Krzysztof; Ingram, David (eds.). Transformation of Healthcare with Information Technologies. Studies in Health Technology and Informatics. Vol. 105. IOS Press. pp. 61–9. ISBN 978-1-58603-438-2. ISSN 0926-9630. PMID 15718595.
- Tolksdorf, Robert; Bontas, Elena Paslaru (2004). "Organizing Knowledge in a Semantic Web for Pathology". Object-Oriented and Internet-Based Technologies. Lecture Notes in Computer Science. Vol. 3263. pp. 115–56. doi:10.1007/978-3-540-30196-7_4. ISBN 978-3-540-23201-8. S2CID 18006838.
- Potts, Steven J. (2009). "Digital pathology in drug discovery and development: Multisite integration". Drug Discovery Today. 14 (19–20): 935–41. doi:10.1016/j.drudis.2009.06.013. PMID 19596461.
- Potts, Steven J.; Young, G. David; Voelker, Frank A. (2010). "The role and impact of quantitative discovery pathology". Drug Discovery Today. 15 (21–22): 943–50. doi:10.1016/j.drudis.2010.09.001. PMID 20946967.
- Zwonitzer, R; Kalinski, T; Hofmann, H; Roessner, A; Bernarding, J (2007). "Digital pathology: DICOM-conform draft, testbed, and first results". Computer Methods and Programs in Biomedicine. 87 (3): 181–8. doi:10.1016/j.cmpb.2007.05.010. PMID 17618703.

![Ki67 stain calculation by QuPath in a pure seminoma, which gives a measure of the proliferation rate of the tumor. The colors represent the intensity of expression: blue-no expression, yellow-low, orange-moderate, and red-high expression.[14]](http://upload.wikimedia.org/wikipedia/commons/thumb/e/eb/Ki67_calculation_by_QuPath_in_a_pure_seminoma.png/295px-Ki67_calculation_by_QuPath_in_a_pure_seminoma.png)

![Breast cancer prediction by AI.[15]](http://upload.wikimedia.org/wikipedia/commons/thumb/4/4f/Histopathology_of_tumor_identification_by_AI.png/278px-Histopathology_of_tumor_identification_by_AI.png)