Cyanotoxin

Cyanotoxins are toxins produced by cyanobacteria (also known as blue-green algae). Cyanobacteria are found almost everywhere, but particularly in lakes and in the ocean where, under high concentration of phosphorus conditions, they reproduce exponentially to form blooms. Blooming cyanobacteria can produce cyanotoxins in such concentrations that they can poison and even kill animals and humans. Cyanotoxins can also accumulate in other animals such as fish and shellfish, and cause poisonings such as shellfish poisoning.
Some of the most powerful natural poisons known are cyanotoxins. They include potent neurotoxins, hepatotoxins, cytotoxins, and endotoxins. The cyano in the term cyanobacteria refers to its colour, not to its relation to cyanides, though cyanobacteria can catabolize hydrogen cyanide during nitrogen fixation.[1]
Exposure to cyanobacteria can result in gastro-intestinal and hayfever symptoms or pruritic skin rashes.[2] Exposure to the cyanobacteria neurotoxin BMAA may be an environmental cause of neurodegenerative diseases such as amyotrophic lateral sclerosis (ALS), Parkinson's disease, and Alzheimer's disease.[3] There is also an interest in the military potential of biological neurotoxins such as cyanotoxins, which "have gained increasing significance as potential candidates for weaponization."[4]
The first published report that blue-green algae or cyanobacteria could have lethal effects appeared in Nature in 1878. George Francis described the algal bloom he observed in the estuary of the Murray River in Australia, as "a thick scum like green oil paint, some two to six inches thick." Wildlife which drank the water died rapidly and terribly.[5] Most reported incidents of poisoning by microalgal toxins have occurred in freshwater environments, and they are becoming more common and widespread. For example, thousands of ducks and geese died drinking contaminated water in the midwestern United States.[6] In 2010, for the first time, marine mammals were reported to have died from ingesting cyanotoxins.[7]
Background
[edit]
Cyanobacteria are ecologically one of the most prolific groups of phototrophic prokaryotes in both marine and freshwater habitats. Both the beneficial and detrimental aspects of cyanobacteria are of considerable significance. They are important primary producers as well as an immense source of several secondary products, including an array of toxic compounds known as cyanotoxins. Abundant growth of cyanobacteria in freshwater, estuarine, and coastal ecosystems due to increased anthropogenic eutrophication and global climate change has created serious concern toward harmful bloom formation and surface water contamination.[8]
Cyanobacteria are considered the most primitive groups of photosynthetic prokaryotes [9] and possibly appeared on the Earth about 3.5 billion years ago.[10] They are ubiquitous in nature and thrive in a variety of ecological niches ranging from desert to hot springs and ice-cold water. Cyanobacteria are an immense source of several secondary natural products with applications in the food, pharmaceuticals, cosmetics, agriculture, and energy sectors.[11] Moreover, some species of cyanobacteria grow vigorously and form a dominant microflora in terms of their biomass and productivity in specific ecosystems. Bloom formations due to excessive growth of certain cyanobacteria followed by the production of toxic compounds have been reported in many eutrophic to hypertrophic lakes, ponds, and rivers throughout the world.[12][8]
A range of toxic secondary compounds, called cyanotoxins, have been reported from cyanobacteria inhabiting freshwater and marine ecosystems. These toxic compounds are highly detrimental for survival of several aquatic organisms, wild and/or domestic animals, and humans. Aquatic organisms, including plants and animals, as well as phytoplankton and zooplankton inhabiting under toxic bloom rich ecosystems, are directly exposed to the harmful effects of different cyanotoxins. The intoxication occurring in wild and/or domestic animals and humans is either due to direct ingestion of cells of toxin producing cyanobacteria or the consumption of drinking water contaminated with cyanotoxins.[12] The toxicity of different cyanotoxins is directly proportional to the growth of cyanobacteria and the extent of their toxin production. It has been shown that the growth of different cyanobacteria and their toxin biosynthesis is greatly influenced by different abiotic factors such as light intensity, temperature, short wavelength radiations, pH, and nutrients.[13][14][12] Global warming and temperature gradients can significantly change species composition and favor blooms of toxic phytoplanktons.[15][16][8]
It has been assumed that cyanotoxins play an important role in chemical defense mechanisms giving survival advantages to the cyanobacteria over other microbes or deterring predation by higher trophic levels.[17][18] Cyanotoxins may also take part in chemical signalling.[8]
Cyanotoxins are produced by cyanobacteria, a phylum of bacteria that obtain their energy through photosynthesis. The prefix cyan comes from the Greek κύανoς meaning "a dark blue substance",[19] and usually indicates any of a number of colours in the blue/green range of the spectrum. Cyanobacteria are commonly referred to as blue-green algae. Traditionally they were thought of as a form of algae, and were introduced as such in older textbooks. However modern sources tend to regard this as outdated;[20] they are now considered to be more closely related to bacteria,[21] and the term for true algae is restricted to eukaryotic organisms.[22] Like true algae, cyanobacteria are photosynthetic and contain photosynthetic pigments, which is why they are usually green or blue.
Cyanobacteria are found almost everywhere; in oceans, lakes and rivers as well as on land. They flourish in Arctic and Antarctic lakes,[23] hotsprings[24] and wastewater treatment plants.[25] They even inhabit the fur of polar bears, to which they impart a greenish tinge.[26] Cyanobacteria produce potent toxins, but they also produce helpful bioactive compounds, including substances with antitumour, antiviral, anticancer, antibiotic and antifungal activity, UV protectants and specific inhibitors of enzymes.[27][28]
Harmful algal blooms
[edit]
Cyanotoxins are often implicated in what are commonly called red tides or harmful algal blooms. Lakes and oceans contain many single-celled organisms called phytoplankton. Under certain conditions, particularly when nutrient concentrations are high, these organisms reproduce exponentially. The resulting dense swarm of phytoplankton is called an algal bloom; these can cover hundreds of square kilometres and can be easily seen in satellite images. Individual phytoplankton rarely live more than a few days, but blooms can last weeks.[29][30]
While some of these blooms are harmless, others fall into the category of harmful algal blooms, or HABs. HABs can contain toxins or pathogens which result in fish kill and can also be fatal to humans.[30] In marine environments, HABs are mostly caused by dinoflagellates,[31] though species of other algae taxa can also cause HABs (diatoms, flagellates, haptophytes and raphidophytes).[32] Marine dinoflagellate species are often toxic, but freshwater species are not known to be toxic. Neither are diatoms known to be toxic, at least to humans.[33]
In freshwater ecosystems, algal blooms are most commonly caused by high levels of nutrients (eutrophication). The blooms can look like foam, scum or mats or like paint floating on the surface of the water, but they are not always visible. Nor are the blooms always green; they can be blue, and some cyanobacteria species are coloured brownish-red. The water can smell bad when the cyanobacteria in the bloom die.[30]
Strong cyanobacterial blooms reduce visibility to one or two centimetres. Species which are not reliant on sight (such as cyanobacteria themselves) survive, but species which need to see to find food and partners are compromised. During the day blooming cyanobacteria saturate the water with oxygen. At night respiring aquatic organisms can deplete the oxygen to the point where sensitive species, such as certain fish, die. This is more likely to happen near the sea floor or a thermocline. Water acidity also cycles daily during a bloom, with the pH reaching 9 or more during the day and dropping to low values at night, further stressing the ecosystem. In addition, many cyanobacteria species produce potent cyanotoxins which concentrate during a bloom to the point where they become lethal to nearby aquatic organisms and any other animals in direct contact with the bloom, including birds, livestock, domestic animals and sometimes humans.[33]
In 1991 a harmful cyanobacterial bloom affected 1,000 km of the Darling-Barwon River in Australia[34] at an economic cost of $10M AUD.[35]
Chemical structure
[edit]Cyanotoxins usually target the nervous system (neurotoxins), the liver (hepatotoxins) or the skin (dermatoxins).[28] The chemical structure of cyanotoxins falls into three broad groups: cyclic peptides, alkaloids and lipopolysaccharides (endotoxins).[36]
| Structure | Cyanotoxin | Primary target organ in mammals | Cyanobacteria genera |
|---|---|---|---|
| Cyclic peptides | Microcystins | Liver | Microcystis, Anabaena, Planktothrix (Oscillatoria), Nostoc, Hapalosiphon, Anabaenopsis |
| Nodularins | Liver | Nodularia | |
| Alkaloids | Anatoxin-a | Nerve synapse | Anabaena, Planktothrix (Oscillatoria), Aphanizomenon |
| Guanitoxin | Nerve synapse | Anabaena | |
| Cylindrospermopsins | Liver | Cylindrospermopsis, Aphanizomenon, Umezakia | |
| Lyngbyatoxin-a | Skin, gastro-intestinal tract | Lyngbya | |
| Saxitoxin | Nerve synapse | Anabaena, Aphanizomenon, Lyngbya, Cylindrospermopsis | |
| Aetokthonotoxin | white matter of the brain; toxicity to mammals not yet confirmed | Aetokthonos | |
| Lipopolysaccharides | Potential irritant; affects any exposed tissue | All | |
| Polyketides | Aplysiatoxins | Skin | Lyngbya, Schizothrix, Planktothrix (Oscillatoria) |
| Amino Acid | BMAA | Nervous system | All |
Most cyanotoxins have a number of variants (analogues). As of 1999, altogether over 84 cyanotoxins were known and only a small number have been well studied.[28]
Cyclic peptides
[edit]A peptide is a short polymer of amino acids linked by peptide bonds. They have the same chemical structure as proteins, except they are shorter. In a cyclic peptide, the ends link to form a stable circular chain. In mammals this stability makes them resistant to the process of digestion and they can bioaccumulate in the liver. Of all the cyanotoxins, the cyclic peptides are of most concern to human health. The microcystins and nodularins poison the liver, and exposure to high doses can cause death. Exposure to low doses in drinking water over a long period of time may promote liver and other tumours.[36]
Microcystins
[edit]
As with other cyanotoxins, microcystins were named after the first organism discovered to produce them, Microcystis aeruginosa. However it was later found other cyanobacterial genera also produced them.[36] There are about 60 known variants of microcystin, and several of these can be produced during a bloom. The most reported variant is microcystin-LR, possibly because the earliest commercially available chemical standard analysis was for microcystin-LR.[36]
Blooms containing microcystin are a problem worldwide in freshwater ecosystems.[37] Microcystins are cyclic peptides and can be very toxic for plants and animals including humans. They bioaccumulate in the liver of fish, in the hepatopancreas of mussels, and in zooplankton. They are hepatotoxic and can cause serious damage to the liver in humans.[36] In this way they are similar to the nodularins (below), and together the microcystins and nodularins account for most of the toxic cyanobacterial blooms in fresh and brackish waters.[28] In 2010, a number of sea otters were poisoned by microcystin. Marine bivalves were the likely source of hepatotoxic shellfish poisoning. This was the first confirmed example of a marine mammal dying from ingesting a cyanotoxin.[7]
Nodularins
[edit]
The first nodularin variant to be identified was nodularin-R, produced by the cyanobacterium Nodularia spumigena.[38] This cyanobacterium blooms in water bodies throughout the world. In the Baltic Sea, marine blooms of Nodularia spumigena are among some of the largest cyanobacterial mass events in the world.[39] (Parts of nine industrialized countries drain into the Baltic Sea, which has little water exchange with the North Sea and Atlantic Ocean. It is consequently one of the more polluted bodies of water in the world (nutrient-rich, from the perspective of cyanobacteria).)
Globally, the most common toxins present in cyanobacterial blooms in fresh and brackish waters are the cyclic peptide toxins of the nodularin family. Like the microcystin family (above), nodularins are potent hepatotoxins and can cause serious damage to the liver. They present health risks for wild and domestic animals as well as humans, and in many areas pose major challenges for the provision of safe drinking water.[28]
Alkaloids
[edit]Alkaloids are a group of naturally occurring chemical compounds which mostly contain basic nitrogen atoms. They are produced by a large variety of organisms, including cyanobacteria, and are part of the group of natural products, also called secondary metabolites. Alkaloids act on diverse metabolic systems in humans and other animals, often with psychotropic or toxic effects. Almost uniformly, they are bitter tasting.[40]
Anatoxin-a
[edit]
Investigations into anatoxin-a, also known as "Very Fast Death Factor", began in 1961 following the deaths of cows that drank from a lake containing an algal bloom in Saskatchewan, Canada.[41][42] The toxin is produced by at least four different genera of cyanobacteria and has been reported in North America, Europe, Africa, Asia, and New Zealand.[43]
Toxic effects from anatoxin-a progress very rapidly because it acts directly on the nerve cells (neurons) as a neurotoxin. The progressive symptoms of anatoxin-a exposure are loss of coordination, twitching, convulsions and rapid death by respiratory paralysis. The nerve tissues which communicate with muscles contain a receptor called the nicotinic acetylcholine receptor. Stimulation of these receptors causes a muscular contraction. The anatoxin-a molecule is shaped so it fits this receptor, and in this way it mimics the natural neurotransmitter normally used by the receptor, acetylcholine. Once it has triggered a contraction, anatoxin-a does not allow the neurons to return to their resting state, because it is not degraded by cholinesterase which normally performs this function. As a result, the muscle cells contract permanently, the communication between the brain and the muscles is disrupted and breathing stops.[44][45]
| External videos | |
|---|---|
University of Nottingham |
The toxin was called the Very Fast Death Factor because it induced tremors, paralysis and death within a few minutes when injected into the body cavity of mice. In 1977, the structure of VFDF was determined as a secondary, bicyclic amine alkaloid, and it was renamed anatoxin-a.[46][47] Structurally, it is similar to cocaine.[48] There is continued interest in anatoxin-a because of the dangers it presents to recreational and drinking waters, and because it is a particularly useful molecule for investigating acetylcholine receptors in the nervous system.[49] The deadliness of the toxin means that it has a high military potential as a toxin weapon.[4]
Cylindrospermopsins
[edit]
Cylindrospermopsin (abbreviated to CYN or CYL) was first discovered after an outbreak of a mystery disease on Palm Island in Australia.[50] The outbreak was traced back to a bloom of Cylindrospermopsis raciborskii in the local drinking water supply, and the toxin was subsequently identified. Analysis of the toxin led to a proposed chemical structure in 1992, which was revised after synthesis was achieved in 2000. Several variants of cylindrospermopsin, both toxic and non-toxic, have been isolated or synthesised.[51]
Cylindrospermopsin is toxic to liver and kidney tissue and is thought to inhibit protein synthesis and to covalently modify DNA and/or RNA. There is concern about the way cylindrospermopsin bioaccumulates in freshwater organisms.[52] Toxic blooms of genera which produce cylindrospermopsin are most commonly found in tropical, subtropical and arid zone water bodies, and have recently been found in Australia, Europe, Israel, Japan and the USA.[36]
Saxitoxins
[edit]
Saxitoxin (STX) is one of the most potent natural neurotoxins known. The term saxitoxin originates from the species name of the butter clam (Saxidomus giganteus) whereby it was first recognized. Saxitoxin is produced by the cyanobacteria Anabaena spp., some Aphanizomenon spp., Cylindrospermopsis sp., Lyngbya sp. and Planktothrix sp., among others).[53] Puffer fish and some marine dinoflagellates also produce saxitoxin.[54][55] Saxitoxins bioaccumulate in shellfish and certain finfish. Ingestion of saxitoxin, usually through shellfish contaminated by toxic algal blooms, can result in paralytic shellfish poisoning.[28]
Saxitoxin has been used in molecular biology to establish the function of the sodium channel. It acts on the voltage-gated sodium channels of nerve cells, preventing normal cellular function and leading to paralysis. The blocking of neuronal sodium channels which occurs in paralytic shellfish poisoning produces a flaccid paralysis that leaves its victim calm and conscious through the progression of symptoms. Death often occurs from respiratory failure.[56] Saxitoxin was originally isolated and described by the United States military, who assigned it the chemical weapon designation "TZ". Saxitoxin is listed in schedule 1 of the Chemical Weapons Convention.[57] According to the book Spycraft, U-2 spyplane pilots were provided with needles containing saxitoxin to be used for suicide in the event escape was impossible.[58]
Aetokthonotoxin
[edit]
Aetokthonotoxin (abbreviated to AETX) was discovered in 2021 as the cyanobacterial neurotoxin causing vacuolar myelinopathy (VM).[59] As the biosynthesis of aetokthonotoxin depends on the availability of bromide in freshwater systems and requires an interplay between the toxin-producing cyanobacterium Aetokthonos hydrillicola and the host plant it epiphytically grows on (most importantly hydrilla), it took > 25 years to discover aetokthonotoxin as the VM-inducing toxin after the disease has first been diagnosed in bald eagles in 1994.[60] The toxin cascades through the food-chain: Among other animals, it affects fish and waterfowl such as coots or ducks which feed on hydrilla colonized with the cyanobacterium. Aetokthonotoxin is transmitted to raptors, such as the bald eagle, that prey on these affected animals.[61]
Vacuolar myelinopathy is characterized by widespread vacuolization of the myelinated axons (intramyelinic edema) in the white matter of the brain and spinal cord. Clinical signs of the intoxication include the severe loss of motor functions and sight. Affected birds fly into objects, lack coordination in swimming, flying and walking, develop tremors of the head and lose their responsiveness. As the toxin has been shown to bioaccumulate, there is concern that it might also be a threat to human health.[59] However, toxicity to mammals has yet to be confirmed experimentally.

Lipopolysaccharides
[edit]Lipopolysaccharides are present in all cyanobacteria. Though not as potent as other cyanotoxins, some researchers have claimed that all lipopolysaccharides in cyanobacteria can irritate the skin, while other researchers doubt the toxic effects are that generalized.[62]
Amino acids
[edit]BMAA
[edit]The non-proteinogenic amino acid beta-Methylamino-L-alanine (BMAA) is ubiquitously produced by cyanobacteria in marine, freshwater, brackish, and terrestrial environments.[63][64] The exact mechanisms of BMAA toxicity on neuron cells is being investigated. Research suggests both acute and chronic mechanisms of toxicity.[65][66] BMAA is being investigated as a potential environmental risk factor for neurodegenerative diseases, including ALS, Parkinson's disease and Alzheimer's disease.[67]
Gallery
[edit]Other cyanotoxins:
-
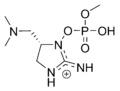
Guanitoxin
-
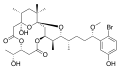
Aplysiatoxin
See also
[edit]References
[edit]- ^ Panou, Manthos; Gkelis, Spyros (2020-01-06), Cyano-assassins: Widespread cyanogenic production from cyanobacteria, doi:10.1101/2020.01.04.894782
- ^ Stewart I, Webb PM, Schluter PJ, Shaw GR (2006). "Recreational and occupational field exposure to freshwater cyanobacteria – a review of anecdotal and case reports, epidemiological studies and the challenges for epidemiologic assessment". Environmental Health. 5 (1): 6. doi:10.1186/1476-069X-5-6. PMC 1513208. PMID 16563159.
- ^ Holtcamp, W. (2012). "The emerging science of BMAA: do cyanobacteria contribute to neurodegenerative disease?". Environmental Health Perspectives. 120 (3): a110–a116. doi:10.1289/ehp.120-a110. PMC 3295368. PMID 22382274.
- ^ a b Dixit A, Dhaked RK, Alam SI, Singh L (2005). "Military potential of biological neurotoxins". Toxin Reviews. 24 (2): 175–207. doi:10.1081/TXR-200057850. S2CID 85651107.
- ^ Francis G (1878). "Poisonous Australian Lake". Nature. 18 (444): 11–12. Bibcode:1878Natur..18...11F. doi:10.1038/018011d0. S2CID 46276288.
- ^ Anatoxin Neil Edwards, University of Sussex at Brighton. Updated 1 September 1999. Retrieved 19 January 2011.
- ^ a b Miller MA, Kudela RM, Mekebri A, Crane D, Oates SC, et al. (2010). Thompson R (ed.). "Evidence for a Novel Marine Harmful Algal Bloom: Cyanotoxin (Microcystin) Transfer from Land to Sea Otters". PLOS ONE. 5 (9): e12576. Bibcode:2010PLoSO...512576M. doi:10.1371/journal.pone.0012576. PMC 2936937. PMID 20844747.
- ^ a b c d e Rastogi, Rajesh P.; Madamwar, Datta; Incharoensakdi, Aran (2015). "Bloom Dynamics of Cyanobacteria and Their Toxins: Environmental Health Impacts and Mitigation Strategies". Frontiers in Microbiology. 6: 1254. doi:10.3389/fmicb.2015.01254. PMC 4646972. PMID 26635737.
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License.
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License.
- ^ Bullerjahn, George S.; Post, Anton F. (2014). "Physiology and molecular biology of aquatic cyanobacteria". Frontiers in Microbiology. 5: 359. doi:10.3389/fmicb.2014.00359. PMC 4099938. PMID 25076944.
- ^ Tomitani, A.; Knoll, A. H.; Cavanaugh, C. M.; Ohno, T. (2006). "The evolutionary diversification of cyanobacteria: Molecular-phylogenetic and paleontological perspectives". Proceedings of the National Academy of Sciences. 103 (14): 5442–5447. Bibcode:2006PNAS..103.5442T. doi:10.1073/pnas.0600999103. PMC 1459374. PMID 16569695.
- ^ Rastogi, Rajesh P.; Sinha, Rajeshwar P. (2009). "Biotechnological and industrial significance of cyanobacterial secondary metabolites". Biotechnology Advances. 27 (4): 521–539. doi:10.1016/j.biotechadv.2009.04.009. PMID 19393308.
- ^ a b c Rastogi, Rajesh P.; Sinha, Rajeshwar P.; Incharoensakdi, Aran (2014). "The cyanotoxin-microcystins: Current overview". Reviews in Environmental Science and Bio/Technology. 13 (2): 215–249. doi:10.1007/s11157-014-9334-6. S2CID 84452003.
- ^ Neilan, Brett A.; Pearson, Leanne A.; Muenchhoff, Julia; Moffitt, Michelle C.; Dittmann, Elke (2013). "Environmental conditions that influence toxin biosynthesis in cyanobacteria". Environmental Microbiology. 15 (5): 1239–1253. doi:10.1111/j.1462-2920.2012.02729.x. PMID 22429476.
- ^ Häder, Donat-P.; Villafañe, Virginia E.; Helbling, E. Walter (2014). "Productivity of aquatic primary producers under global climate change". Photochem. Photobiol. Sci. 13 (10): 1370–1392. doi:10.1039/c3pp50418b. hdl:11336/24725. PMID 25191675.
- ^ El-Shehawy, Rehab; Gorokhova, Elena; Fernández-Piñas, Francisca; Del Campo, Francisca F. (2012). "Global warming and hepatotoxin production by cyanobacteria: What can we learn from experiments?". Water Research. 46 (5): 1420–1429. Bibcode:2012WatRe..46.1420E. doi:10.1016/j.watres.2011.11.021. PMID 22178305.
- ^ Hã¤Der, Donat-P.; Gao, Kunshan (2015). "Interactions of anthropogenic stress factors on marine phytoplankton". Frontiers in Environmental Science. 3. doi:10.3389/fenvs.2015.00014.
- ^ Jang, Min-Ho; Ha, Kyong; Takamura, Noriko (2007). "Reciprocal allelopathic responses between toxic cyanobacteria (Microcystis aeruginosa) and duckweed (Lemna japonica)". Toxicon. 49 (5): 727–733. doi:10.1016/j.toxicon.2006.11.017. PMID 17207510.
- ^ Berry, John P.; Gantar, M.; Perez, M. H.; Berry, G.; Noriega, F. G. (2008). "Cyanobacterial Toxins as Allelochemicals with Potential Applications as Algaecides, Herbicides and Insecticides". Marine Drugs. 6 (2): 117–146. doi:10.3390/md20080007. PMC 2525484. PMID 18728763.
- ^ κύανος, Henry George Liddell, Robert Scott, A Greek-English Lexicon, on Perseus
- ^ Nabors, Murray W. (2004). Introduction to Botany. San Francisco, CA: Pearson Education, Inc. ISBN 978-0-8053-4416-5.
- ^ Ed. Guiry, M.D., John, D.M., Rindi, F and McCarthy, T.K. 2007. New Survey of Clare Island Volume 6: The Freshwater and Terrestrial Algae. Royal Irish Academy. ISBN 978-1-904890-31-7
- ^ Allaby M, ed. (1992). "Algae". The Concise Dictionary of Botany. Oxford: Oxford University Press.
- ^ Skulberg OM (1996) "Terrestrial and limnic algae and cyanobacteria". In: A Catalogue of Svalvard Plants, Fungi, Algae and Cyanobacteria, Part 9, A Elvebakk and P Prestud (eds.) Norsk Polarinstitutt Skrifter, 198: 383-395.
- ^ Castenholz, R. A. (1973). "Ecology of blue-green algae in hotsprings". In Carr, N. G.; Whitton, B. A. (eds.). The Biology of Blue-green algae. Oxford: Blackwell. pp. 379–414. ISBN 0-632-09040-5.
- ^ Vasconcelos VM, Pereira E (2001). "Cyanobacteria diversity and toxicity in a Wastewater Treatment Plant (Portugal)". Water Research. 35 (5): 1354–1357. Bibcode:2001WatRe..35.1354V. doi:10.1016/S0043-1354(00)00512-1. PMID 11268858.
- ^ Gerald Karp (19 October 2009). Cell and Molecular Biology: Concepts and Experiments. John Wiley and Sons. pp. 14–. ISBN 978-0-470-48337-4. Retrieved 26 January 2011.
- ^ Herrero, Antonia; Flores, Enrique (2008). The Cyanobacteria: Molecular Biology, Genomics and Evolution. Caister Academic Press. ISBN 978-1-904455-15-8.
- ^ a b c d e f Sivonen K and Jones G (1999) "Cyanobacterial Toxins" Archived 2007-01-24 at the Wayback Machine In Toxic Cyanobacteria in Water. Chorus I and Bartram J (eds): 41-111. WHO, Geneva. ISBN 0419239308.
- ^ Lindsey R and Scott M (2010) What are phytoplankton NASA Earth Observatory.
- ^ a b c Harmful algal blooms event response Archived 2016-03-04 at the Wayback Machine NOAA, Center of Excellence for Great Lakes and Human Health. Accessed 6 August 2014.
- ^ Stewart I and Falconer IR (2008) "Cyanobacteria and cyanobacterial toxins" Pages 271–296 in Oceans and human health: risks and remedies from the seas, Eds: Walsh PJ, Smith SL and Fleming LE. Academic Press, ISBN 0-12-372584-4.
- ^ Moestrup Ø, Akselman R, Cronberg G, Elbraechter M, Fraga S, Halim Y, Hansen G, Hoppenrath M, Larsen J, Lundholm N, Nguyen LN and Zingone A. "IOC-UNESCO Taxonomic Reference List of Harmful Micro Algae (HABs)" Accessed 21 January 2011.
- ^ a b Vasconcelos V (2006). "Eutrophication, toxic cyanobacteria and cyanotoxins: when ecosystems cry for help" (PDF). Limnetica. 25 (1–2): 425–432. doi:10.23818/limn.25.30. S2CID 59434407. Archived from the original (PDF) on 2011-07-23. Retrieved 2011-01-26.
- ^ Forc, N.S.W.B.G.A.T. (1992). "Final report of the NSW Blue-Green Algae Task Force". Parramatta: NSW Department of Water Resources.
- ^ Herath, G. (1995). "The algal bloom problem in Australian waterways: an economic appraisal". Review of Marketing and Agricultural Economics. 63 (1): 77–86.
- ^ a b c d e f g Chorus, Ingrid; Welker, Martin (2021-03-07). Toxic Cyanobacteria in Water: A Guide to Their Public Health Consequences, Monitoring and Management (2 ed.). London: CRC Press. doi:10.1201/9781003081449. ISBN 978-1-003-08144-9.
- ^ Pelaez, Miguel; Antoniou, Maria G.; He, Xuexiang; Dionysiou, Dionysios D.; de la Cruz, Armah A.; Tsimeli, Katerina; et al. (2010). "Sources and Occurrence of Cyanotoxins Worldwide". Xenobiotics in the Urban Water Cycle. Environmental Pollution. Vol. 16. pp. 101–127. doi:10.1007/978-90-481-3509-7_6. ISBN 978-90-481-3508-0. ISSN 1566-0745.
- ^ Sivonen K, Kononen K, Carmichael WW, Dahlem AM, Rinehart KL, Kiviranta J, Niemela SI (1989). "Occurrence of the hepatotoxic cyanobacterium Nodularia spumigena in the Baltic Sea and structure of the toxin". Appl. Environ. Microbiol. 55 (8): 1990–5. Bibcode:1989ApEnM..55.1990S. doi:10.1128/aem.55.8.1990-1995.1989. PMC 202992. PMID 2506812.
- ^ David P. Fewer DP; Köykkä K; Halinen K; Jokela J; Lyra C; Sivonen K (2009). "Culture-independent evidence for the persistent presence and genetic diversity of microcystin-producing Anabaena (Cyanobacteria) in the Gulf of Finland". Environmental Microbiology. 11 (4): 855–866. doi:10.1111/j.1462-2920.2008.01806.x. PMID 19128321.
- ^ Rhoades, David F (1979). "Evolution of Plant Chemical Defense against Herbivores". In Rosenthal, Gerald A.; Janzen, Daniel H (eds.). Herbivores: Their Interaction with Secondary Plant Metabolites. New York: Academic Press. p. 41. ISBN 978-0-12-597180-5.
- ^ Carmichael WW, Gorham PR (1978). "Anatoxins from clones of Anabaena flos-aquae isolated from lakes of western Canada". Mitt. Infernal. Verein. Limnol. 21: 285–295.
- ^ Carmichael WW, Biggs DF, Gorham PR (1975). "Toxicology and pharmacological action of Anabaena flos-aquae toxin". Science. 187 (4176): 542–544. Bibcode:1975Sci...187..542C. doi:10.1126/science.803708. PMID 803708.
- ^ Yang, X (2007) Occurrence of the cyanobacterial neurotoxin, anatoxin-a, in New York State waters ProQuest Dissertations and Theses. ISBN 978-0-549-35451-2.
- ^ Wood S. A.; Rasmussen J. P.; Holland P. T.; Campbell R.; Crowe A. L. M. (2007). "First Report of the Cyanotoxin Anatoxin-A from Aphanizomenon issatschenkoi (cyanobacteria)". Journal of Phycology. 43 (2): 356–365. doi:10.1111/j.1529-8817.2007.00318.x. S2CID 84284928.
- ^ National Center for Environmental Assessment. "Toxicological Reviews of Cyanobacterial Toxins: Anatoxin-a" NCEA-C-1743
- ^ Devlin JP, Edwards OE, Gorham PR, Hunter NR, Pike RK, Stavric B (1977). "Anatoxin-a, a toxic alkaloid from Anabaena flos-aquae NRC-44h". Can. J. Chem. 55 (8): 1367–1371. doi:10.1139/v77-189.
- ^ Moore RE (1977). "Toxins from blue-green algae". BioScience. 27 (12): 797–802. doi:10.2307/1297756. JSTOR 1297756.
- ^ Metcalf, James S.; Codd, Geoffrey A. (2009). "Cyanobacteria, neurotoxins and water resources: Are there implications for human neurodegenerative disease?". Amyotrophic Lateral Sclerosis. 10: 74–78. doi:10.3109/17482960903272942. PMID 19929737. S2CID 41880444.
- ^ Stewart I, Seawright AA, Shaw GR (2008). "Cyanobacterial poisoning in livestock, wild mammals and birds – an overview" (PDF). Cyanobacterial Harmful Algal Blooms: State of the Science and Research Needs. Advances in Experimental Medicine and Biology. Vol. 619. pp. 613–637. doi:10.1007/978-0-387-75865-7_28. ISBN 978-0-387-75864-0. PMID 18461786. Archived from the original (PDF) on 2013-10-23.
- ^ Byth S (July 1980). "Palm Island mystery disease". The Medical Journal of Australia. 2 (1): 40–42. doi:10.5694/j.1326-5377.1980.tb131814.x. PMID 7432268. S2CID 273293.
- ^ Griffiths DJ, Saker ML (2003). "The Palm Island mystery disease 20 years on: a review of research on the cyanotoxin cylindrospermopsin". Environ Toxicol. 18 (2): 78–93. Bibcode:2003EnTox..18...78G. doi:10.1002/tox.10103. PMID 12635096. S2CID 25219655.
- ^ Kinnear S (2010). "Cylindrospermopsin: A Decade of Progress on Bioaccumulation Research". Marine Drugs. 8 (3): 542–564. doi:10.3390/md8030542. PMC 2857366. PMID 20411114.
- ^ Clark RF, Williams SR, Nordt SP, Manoguerra AS (1999). "A review of selected seafood poisonings". Undersea Hyperb Med. 26 (3): 175–84. PMID 10485519. Archived from the original on 2011-08-11. Retrieved 2008-08-12.
{{cite journal}}: CS1 maint: unfit URL (link) - ^ Nakamuraa M, Oshimaa Y, Yasumoto T (1984). "Occurrence of saxitoxin in puffer fish". Toxicon. 22 (3): 381–385. doi:10.1016/0041-0101(84)90082-5. PMID 6474491.
- ^ Landsberg JH (2002). "The effects of harmful algal blooms on aquatic organisms". Reviews in Fisheries Science. 10 (2): 113–390. doi:10.1080/20026491051695. S2CID 86185142.
- ^ Kao CY and Levinson SR (1986) Tetrodotoxin, saxitoxin, and the molecular biology of the sodium channel New York Academy of Sciences. ISBN 0-89766-354-3.
- ^ Chemical Weapons Convention: Schedule 1 Archived 2013-06-07 at the Wayback Machine Organisation for the Prohibition of Chemical Weapons, The Hague, Netherlands. Retrieved 26 January 2011.
- ^ Wallace R, Melton HK and Schlesinger HR (2009) Spycraft: the secret history of the CIA's spytechs from communism to Al-Qaeda. Penguin Group USA, ISBN 0-452-29547-5.
- ^ a b Breinlinger, Steffen; Phillips, Tabitha J.; Haram, Brigette N.; Mareš, Jan; Yerena, José A. Martínez; Hrouzek, Pavel; Sobotka, Roman; Henderson, W. Matthew; Schmieder, Peter; Williams, Susan M.; Lauderdale, James D. (2021-03-26). "Hunting the eagle killer: A cyanobacterial neurotoxin causes vacuolar myelinopathy". Science. 371 (6536): eaax9050. doi:10.1126/science.aax9050. ISSN 0036-8075. PMC 8318203. PMID 33766860.
- ^ "Avian vacuolar myelinopathy". USGS National Wildlife Health Center. Archived from the original on 6 October 2014. Retrieved 24 October 2013.
- ^ Birrenkott, A. H.; S. B Wilde; J. J. Hains; J. R. Fisher; T. M. Murphy; C. P. Hope; P. G. Parnell; W. W. Bowerman (2004). "Establishing a food-chain link between aquatic plant material and avian vacuolar myelinopathy in mallards (Anas platyrhynchos)". Journal of Wildlife Diseases. 40 (3): 485–492. doi:10.7589/0090-3558-40.3.485. PMID 15465716.
- ^ Stewart I, Schluter PJ, Shaw GR (2006). "Cyanobacterial lipopolysaccharides and human health - a review". Environ Health. 5 (1): 7. doi:10.1186/1476-069X-5-7. PMC 1489932. PMID 16563160.
- ^ Cox, PA; Banack, SA; Murch, SJ; Rasmussen, U; Tien, G; Bidigare, RR; Metcalf, JS; Morrison, LF; Codd, GA; Bergman, B. (2005). "Diverse taxa of cyanobacteria produce b-N-methylamino-L-alanine, a neurotoxic amino acid". PNAS. 102 (14): 5074–5078. Bibcode:2005PNAS..102.5074C. doi:10.1073/pnas.0501526102. PMC 555964. PMID 15809446.
- ^ Esterhuizen, M; Downing, TG. (2008). "β-N-methylamino-L-alanine (BMAA) in novel South African cyanobacterial isolates". Ecotoxicology and Environmental Safety. 71 (2): 309–313. doi:10.1016/j.ecoenv.2008.04.010. PMID 18538391.
- ^ Weiss JH, Koh JY, Choi DW (1989). "Neurotoxicity of β -N-methylamino-L-alanine (BMAA) and β-N-oxalylamino-L-alanine (BOAA) on cultured cortical neurons". Brain Research. 497 (1): 64–71. doi:10.1016/0006-8993(89)90970-0. PMID 2551452. S2CID 140209787.
- ^ Lobner, D; Piana, PM; Salous, AK; Peoples, RW. (2007). "β-N-methylamino-L-alanine enhances neurotoxicity through multiple mechanisms". Neurobiology of Disease. 25 (2): 360–366. doi:10.1016/j.nbd.2006.10.002. PMC 3959771. PMID 17098435.
- ^ Cox PA, Davis DA, Mash DC, Metcalf JS, Banack SA (2015). "Dietary exposure to an environmental toxin triggers neurofibrillary tangles and amyloid deposits in the brain". Proceedings of the Royal Society B. 283 (1823): 20152397. doi:10.1098/rspb.2015.2397. PMC 4795023. PMID 26791617.
External links
[edit]- Cyanosite - A Webserver for Cyanobacterial Research, Purdue University.
- Dangers of toxic algae Environment Canterbury Updated 31 October 2009. Retrieved 23 January 2011.